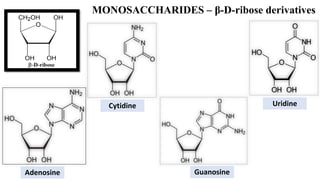
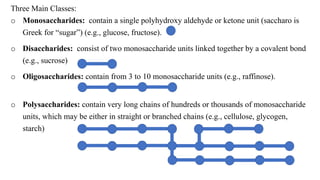
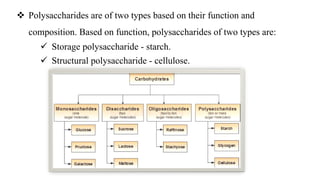
This document provides information about carbohydrates. It begins by defining carbohydrates and describing their main biological functions. It then discusses the three main classes of carbohydrates: monosaccharides, disaccharides, and polysaccharides. For each class, key examples are provided and their structures and properties are explained. The document also covers topics like stereochemistry of carbohydrates, glycosaminoglycans, and important monosaccharides and polysaccharides like starch, cellulose, and glycogen. In summary, it serves as a comprehensive overview of carbohydrate structure, classification, and functions in biological systems.





![STEREOCHEMISTRY OF CARBOHYDRATES
Optical Activity:
• A levorotatory (–) substance rotates polarized light to the left.
[E.g., l-glucose; (-)-glucose]
• A dextrorotatory (+) substance rotates polarized light to the right.
[E.g., d-glucose; (+)-glucose]
• Molecules which rotate the plane of of polarized light are optically active.
• Most biologically important molecules are chiral, and hence are optically
active. Often, living system contain only one of all of the possible
stereochemical forms of a compound. In some cases, one form of a molecule
is beneficial, and the enantiomer is a poison (e.g., thalidomide).](https://image.slidesharecdn.com/biochemistrylecturenotescarbohydrates-200427035744/85/Biochemistry-lecture-notes-carbohydrates-8-320.jpg)





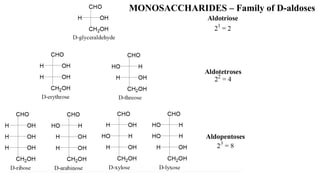
![MONOSACCHARIDES
• Monosaccharides are classified according to the number of carbon atoms they
contain:
• The presence of an aldehyde is indicated by
prefix aldo- and a ketone by the prefix keto-
ü glucose is an aldohexose (aldehyde + 6 Cs)
ü ribulose is a ketopentose (ketone + 5 Cs)]](https://image.slidesharecdn.com/biochemistrylecturenotescarbohydrates-200427035744/85/Biochemistry-lecture-notes-carbohydrates-14-320.jpg)