Embed presentation
Downloaded 63 times




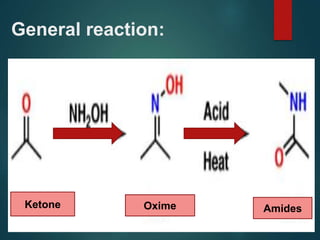
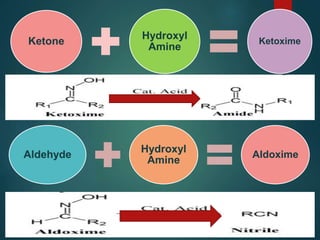
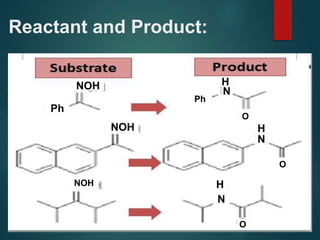
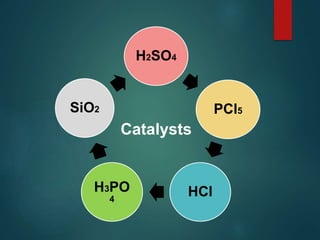
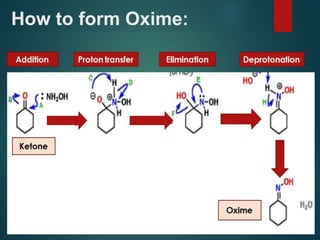
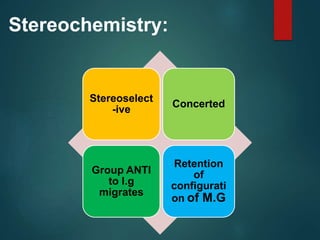

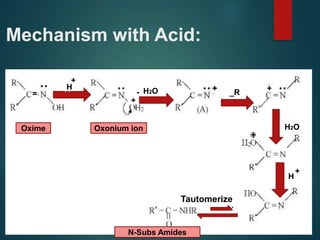
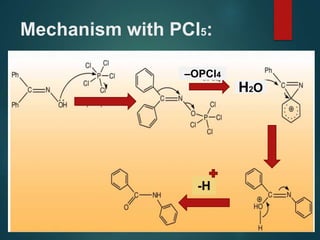
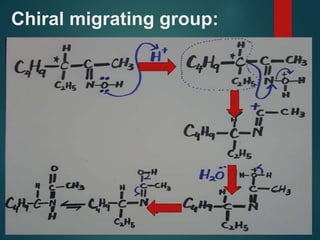
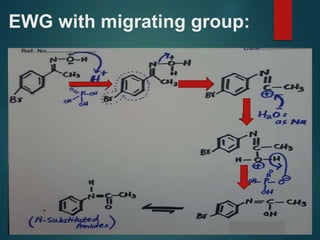
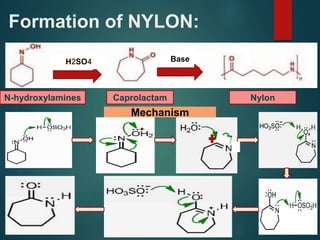
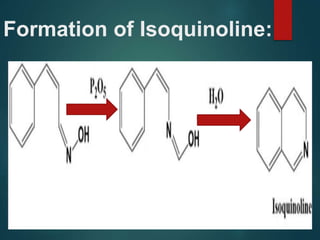

The Beckmann rearrangement is an acid-catalyzed reaction where an oxime is converted to an amide. Specifically, the reaction involves the migration of an acyl group from carbon to nitrogen on the oxime. This rearrangement allows the conversion of ketones and aldehydes to amides. Common catalysts used include sulfuric acid, phosphorus pentachloride, and hydrochloric acid. The reaction proceeds through an oxonium ion intermediate.




















