Embed presentation
Downloaded 28 times


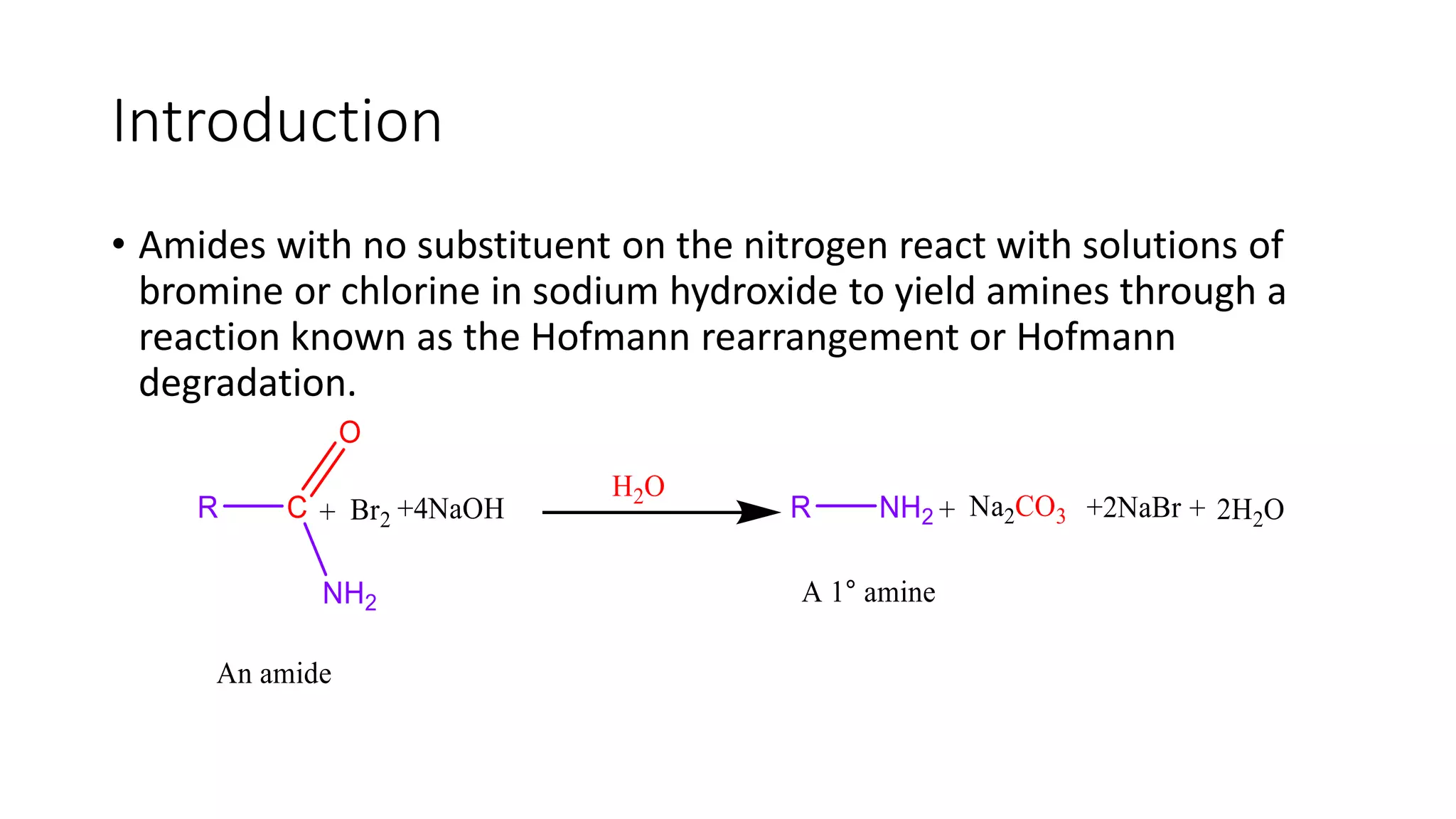
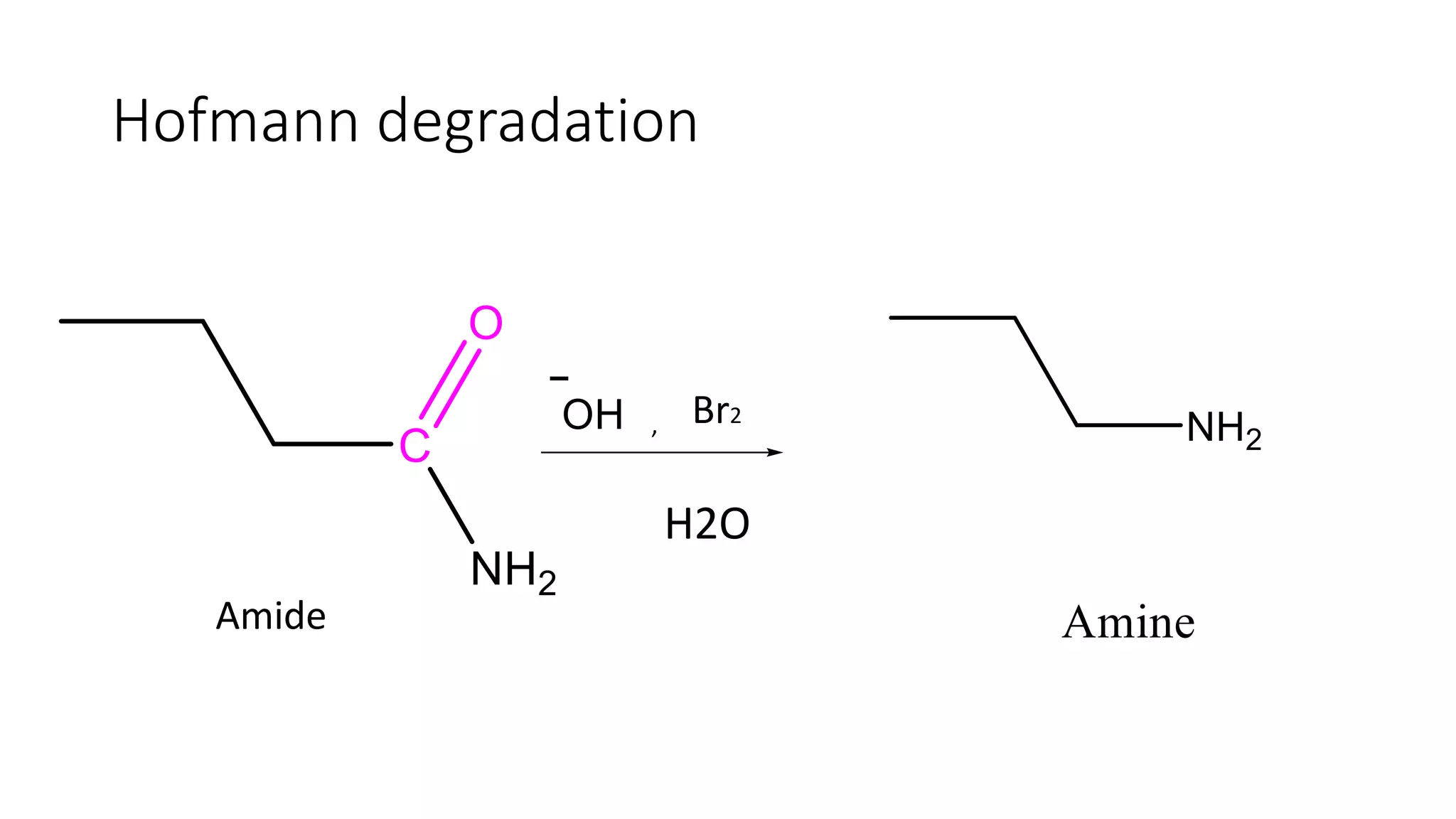
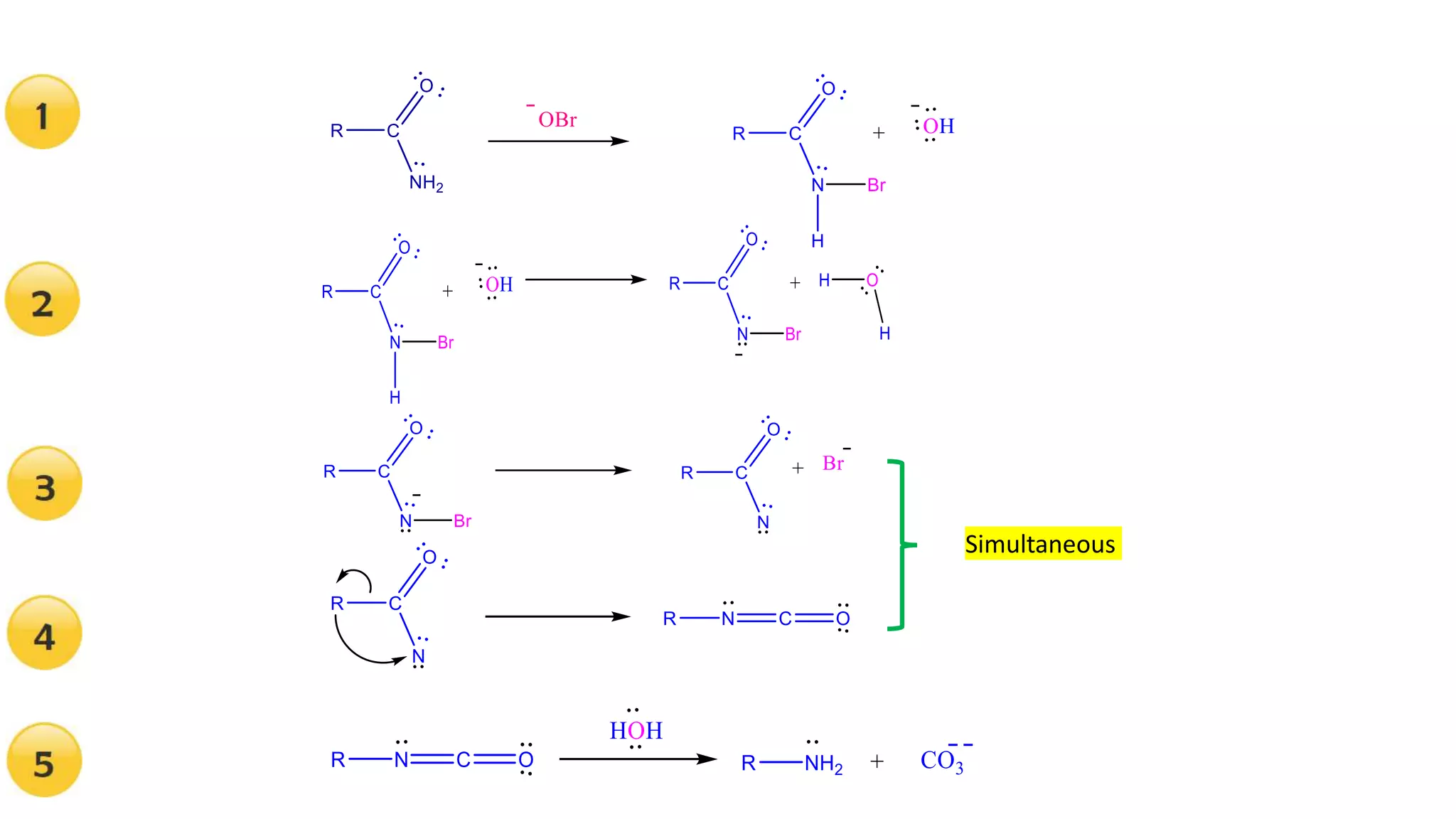
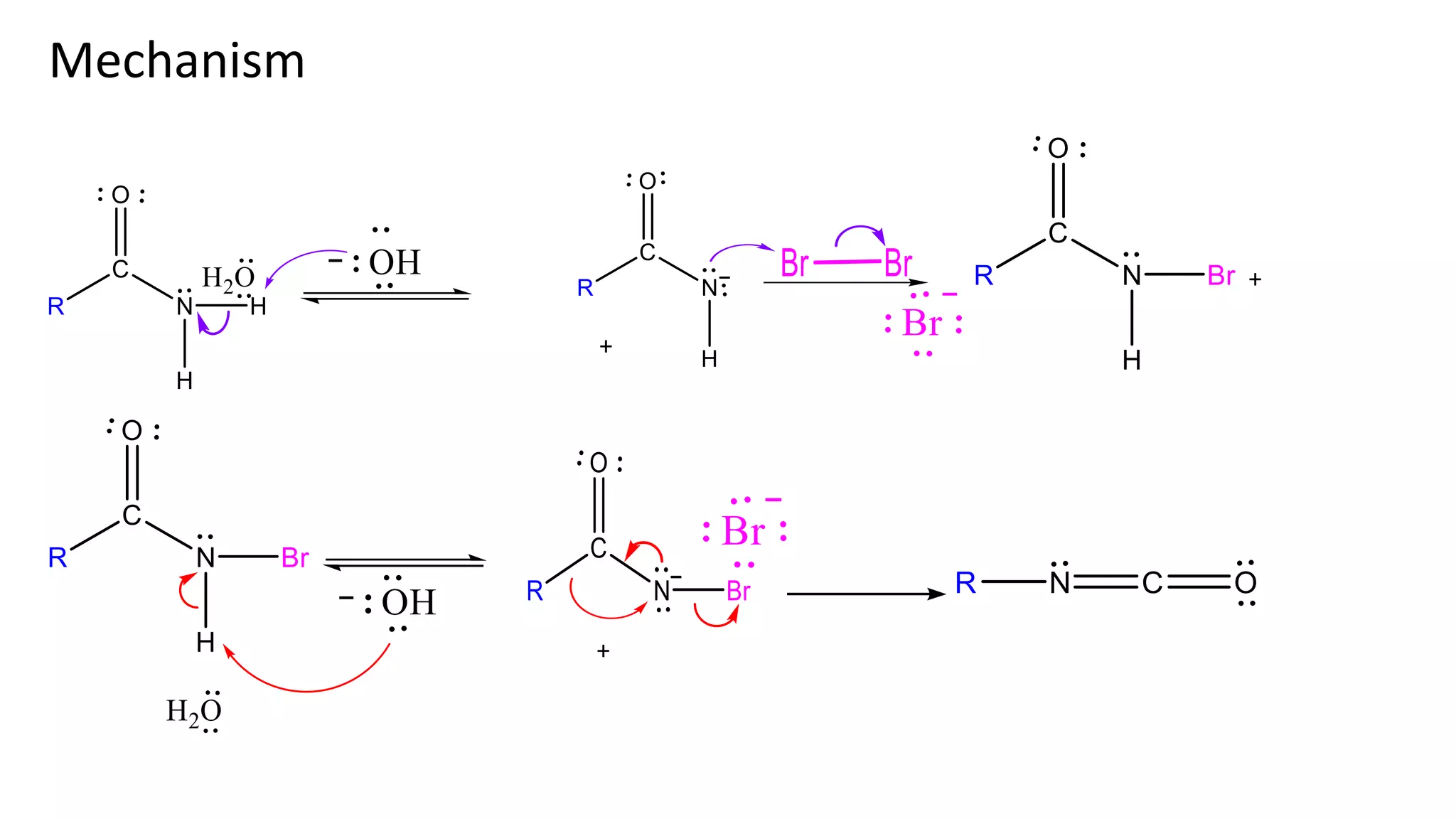


The document discusses the Hofmann rearrangement, a chemical reaction where amides without nitrogen substituents convert into amines using bromine or chlorine in sodium hydroxide. It highlights the historical context and contributions of August Wilhelm von Hofmann, as well as the mechanisms involved in the reaction. Support for the mechanism is provided through the isolation of intermediates such as N-haloamides and isocyanates.
Overview of the Hofmann Rearrangement and its discoverer, August Wilhelm von Hofmann, highlighting his contributions to chemistry.
Description of the Hofmann Rearrangement where amides react with bromine or chlorine in NaOH to form amines.
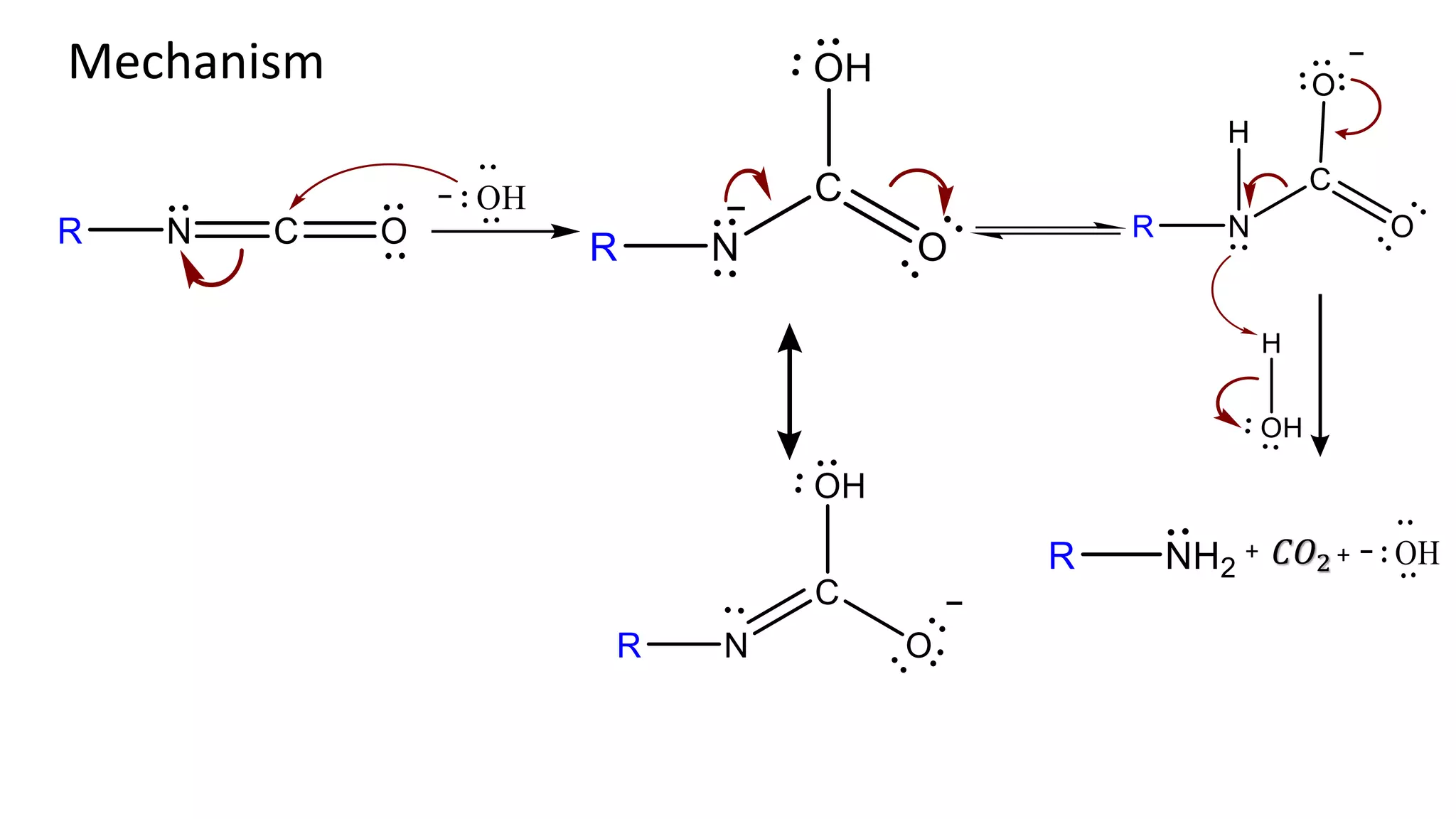
Illustrations of the reaction mechanism involved in the Hofmann degradation, showing intermediates and products.
Evidence for the mechanism includes the isolation of intermediates like N-haloamide and Isocyanate.
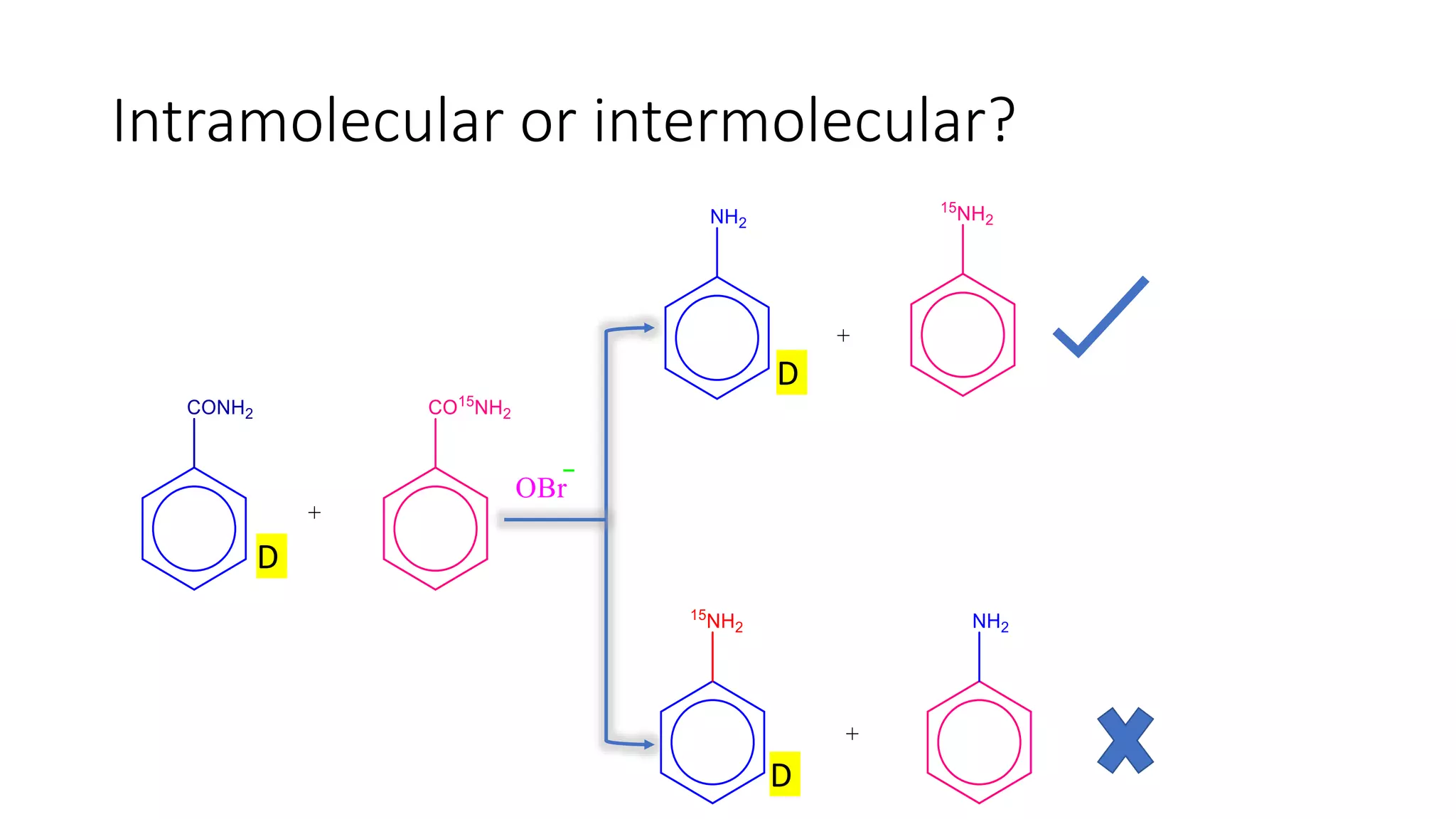
Exploration of intramolecular vs intermolecular aspects of the Hofmann Rearrangement.
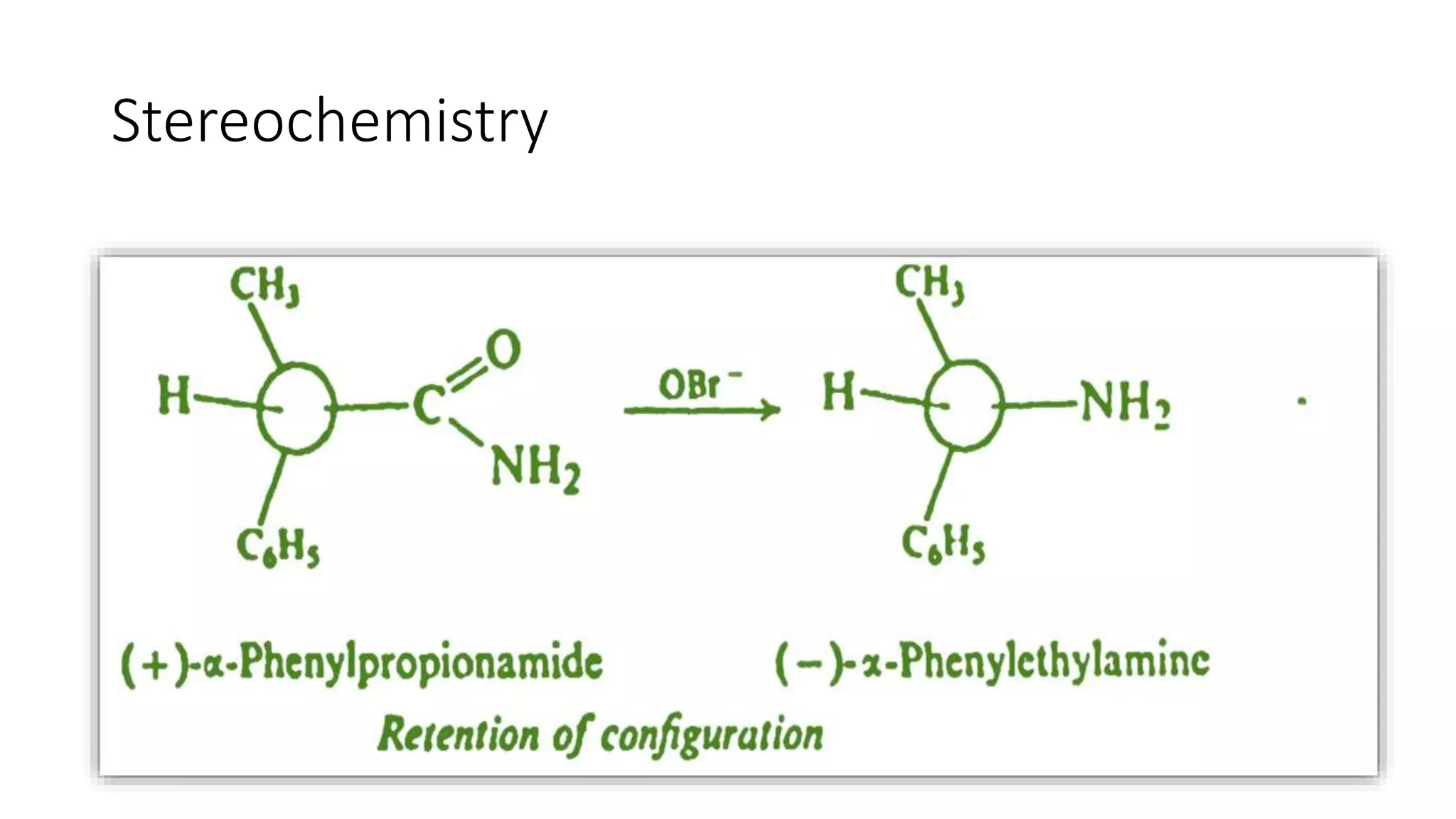
Discussion of stereochemistry associated with the Hofmann Rearrangement outcomes.









