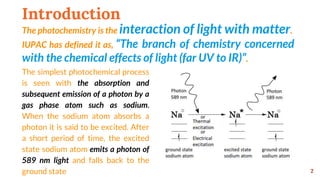
The document provides an overview of basic principles of photochemistry. It discusses key concepts like photochemical processes, importance of photochemistry, terminology used in photochemistry including charge transfer transitions, multiplicity, internal conversion and more. It also explains photochemical reaction processes like dissociation, direct reactions, isomerization, energy transfer and quenching. Laws of photochemistry, quantum yield, Jablonski diagram, Franck-Condon principle, electronic transitions, mechanisms of photosensitization and applications of photosensitization are summarized.


















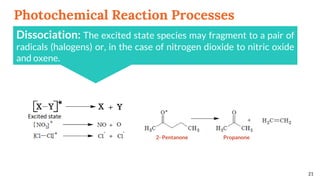
![Photochemical Reaction Processes
20
A photo-excited species, [X-Y]*, can relax (react) via a
variety of pathways as given below.](https://image.slidesharecdn.com/basicphotochemistry-200514085949/85/Basic-photochemistry-20-320.jpg)
![Photochemical Reaction Processes
22
Direct Reaction: The photo-excited state may undergo reactions
unavailable to the ground state species. E.g., a photo-excited ketone can
undergo a [2+2] cycloaddition with an alkene to give an oxetane, a
Paterno-Büchi reaction .](https://image.slidesharecdn.com/basicphotochemistry-200514085949/85/Basic-photochemistry-22-320.jpg)




![Primary Photochemical Reactions
27
Reactions initiated by 𝛑 𝛑 * state
[S1 state]
Reactions involving Carbocations
Reactions involving Carbanions
Concerted Pericyclic reactions
Electron Transfer reactions
Cis-trans Isomerization reactions
Homolytic Fragmentation
Hydrogen atom Abstraction
Addition to Unsaturated Bonds
Rearrangement of stable carbon
centred radicals
Reactions initiated by n 𝛑 * state
[T1 state]](https://image.slidesharecdn.com/basicphotochemistry-200514085949/85/Basic-photochemistry-27-320.jpg)




