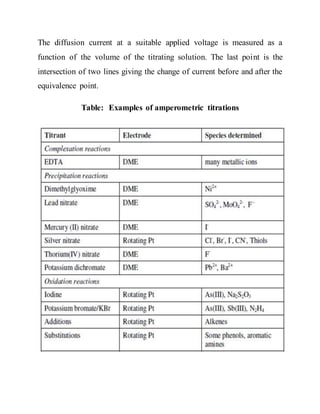
1. Complexometric titrations involve titrating metal ions with chelating agents like EDTA.
2. Applications include direct titration of metals like Cu and Zn as well as back titration, replacement titration, and indirect titration for metals that cannot be directly titrated.
3. Aquametric titrations determine the water content of samples, important for applications like soil quality analysis, food quality control, and environmental monitoring.