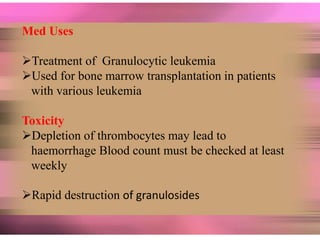
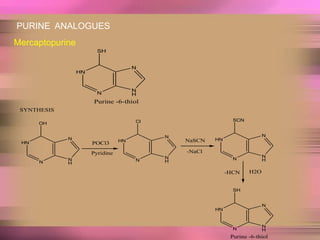
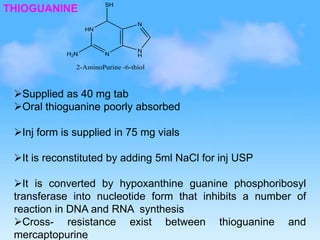
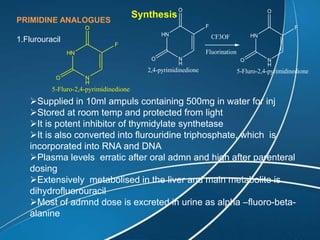
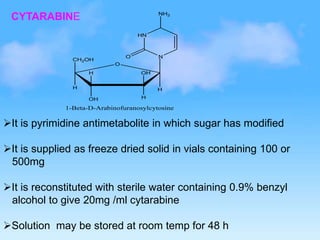
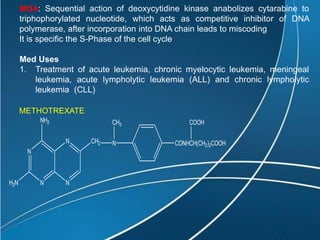
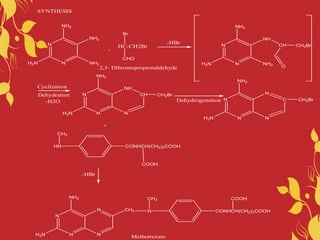
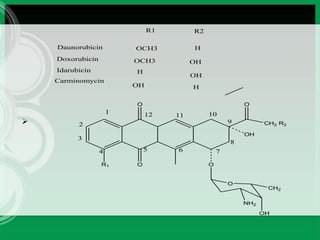
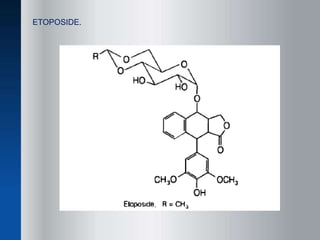
This document discusses anti-neoplastic agents used for cancer treatment. It describes how cancer is characterized by abnormal cell division and spread. Anti-cancer treatments include surgery, radiation, immunotherapy and chemotherapy using drugs that kill cancer cells. Common classes of chemotherapeutic drugs discussed include alkylating agents, antimetabolites, antibiotics, and plant products. Specific drugs like mechlorethamine, cyclophosphamide, melphalan, chlorambucil and their mechanisms of action, uses, and toxicities are explained.









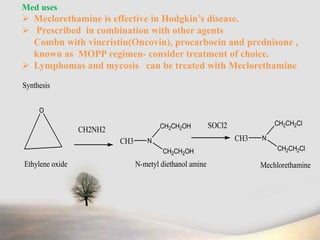




![Melphalan
N
ClH2CH2C
ClH2CH2C
COOH
NH2
Synthesis
N
ClH2CH2C
ClH2CH2C
COOH
NH2
COOH
NH2
HNO3
H2SO4
C2H5OH/HCl
COOC2H5
NH2
O2N
Phthalic anhydride [H]
COOC2H5
N
H2N
O
O
O
1.
2.SOCl2
3.HCl/NH2NH2](https://image.slidesharecdn.com/anticanceragentsfcp-final1-211225172656/85/Anticancer-agents-in-medicinal-chemistry-15-320.jpg)