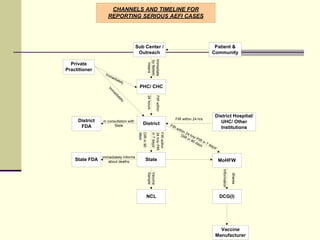
This document outlines India's guidelines for monitoring adverse events following immunization (AEFI). It discusses the importance of AEFI surveillance for ensuring vaccine safety and public confidence. It describes the steps for detecting, reporting, investigating, and responding to AEFIs at various levels of the healthcare system. All serious or unexpected health issues post-vaccination should be reported and investigated to determine the cause and prevent future occurrences. The goal is to quickly respond to AEFIs to strengthen immunization programs and ensure safe vaccination practices.