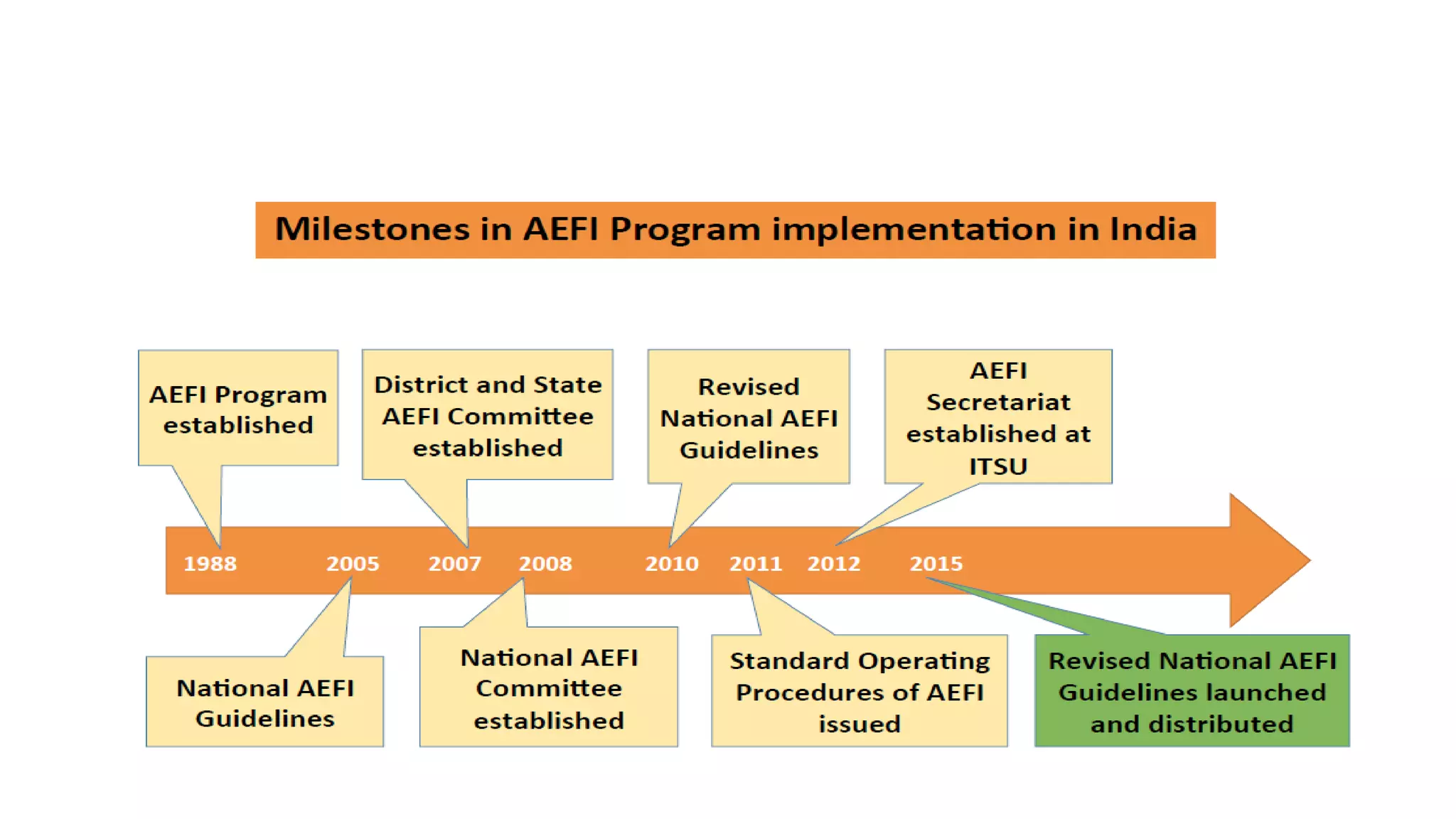
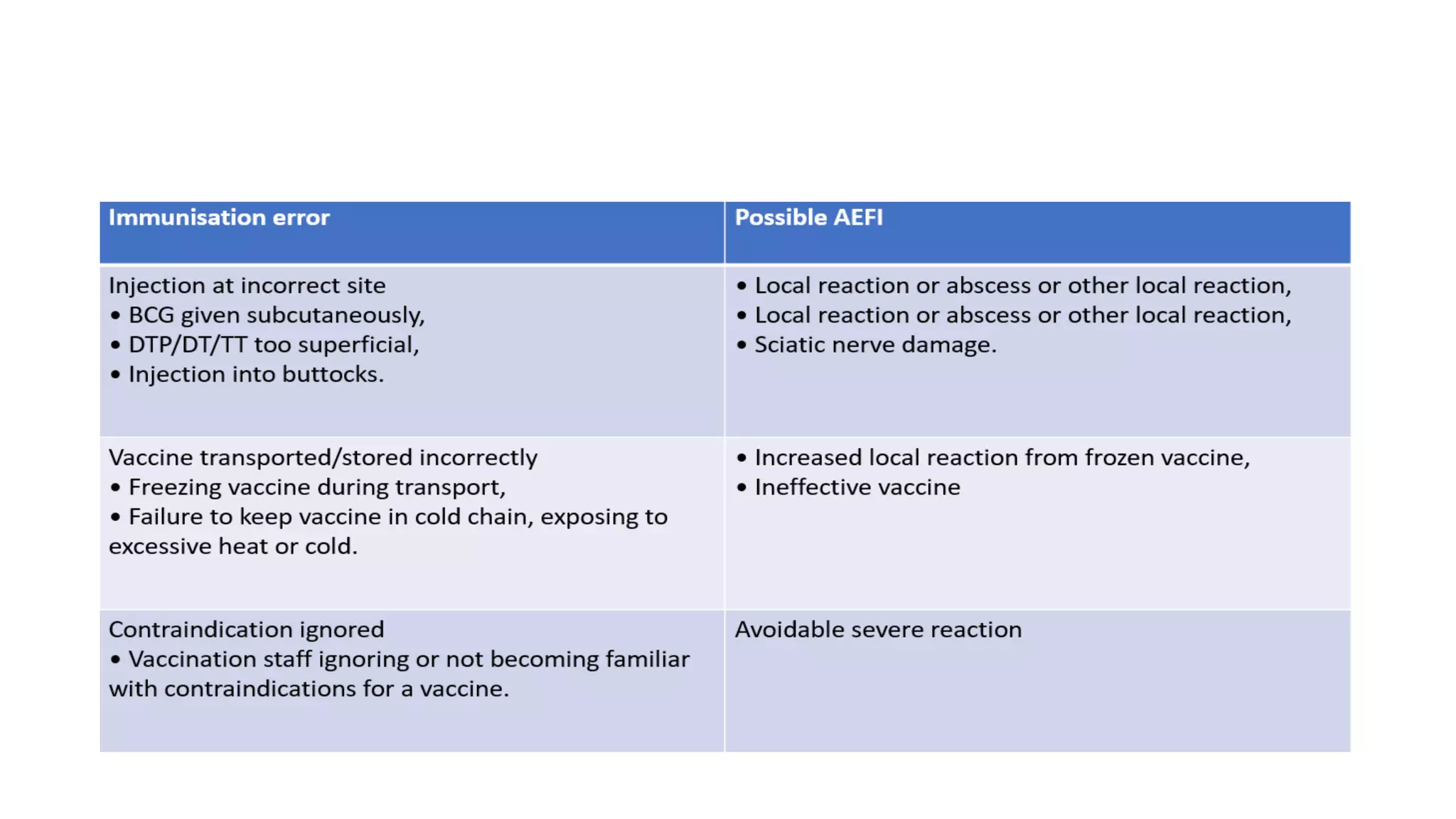
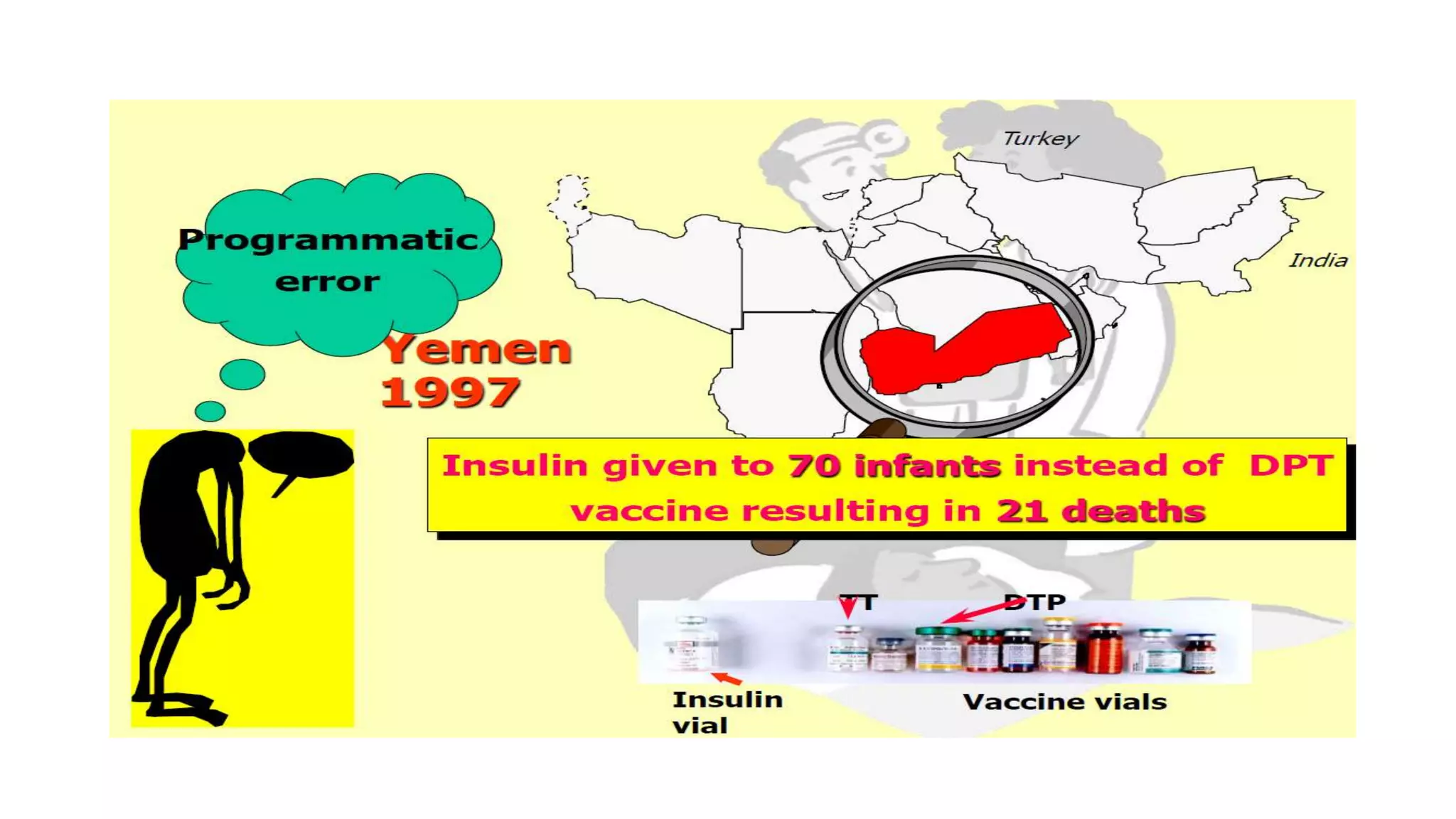
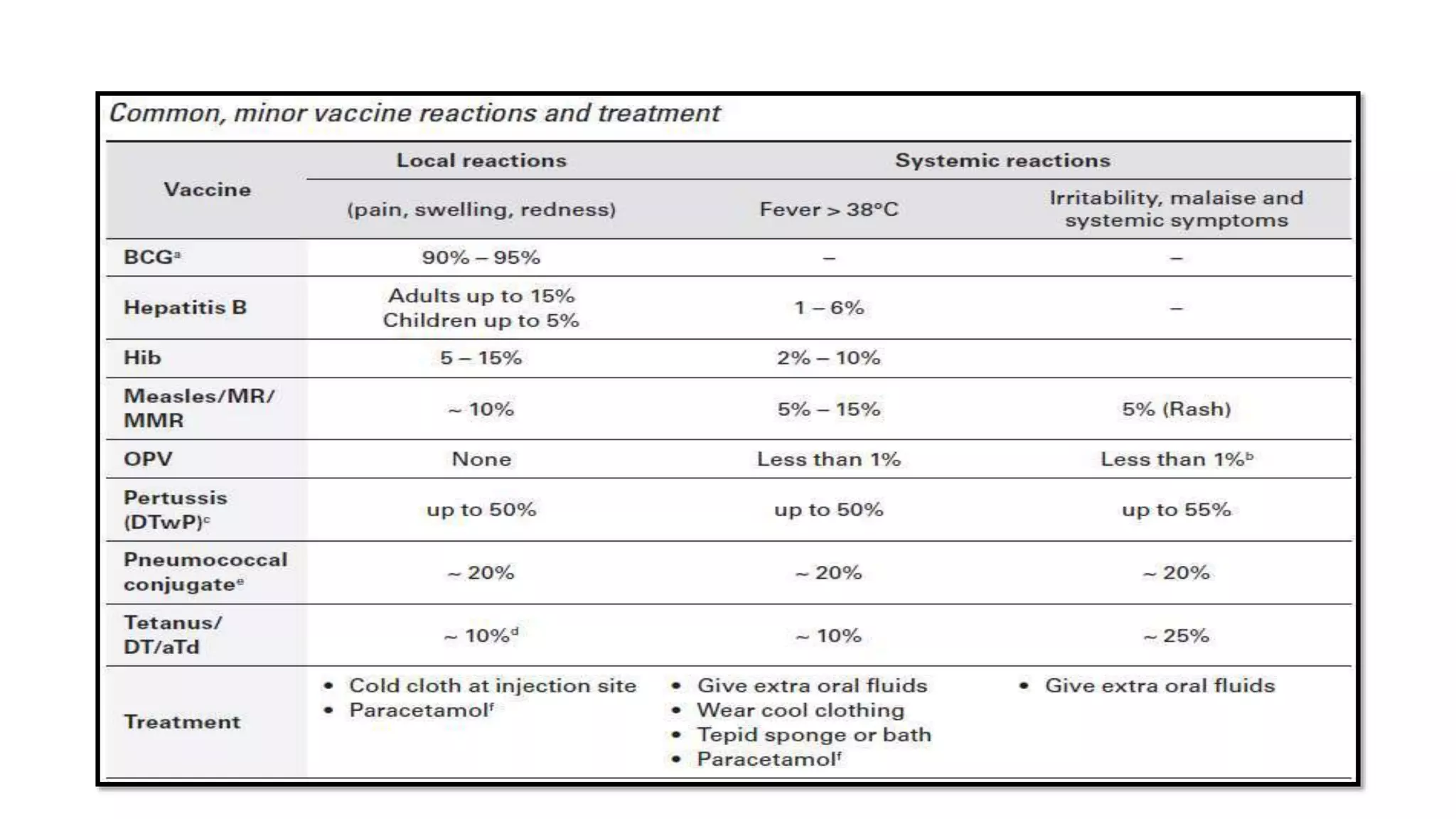
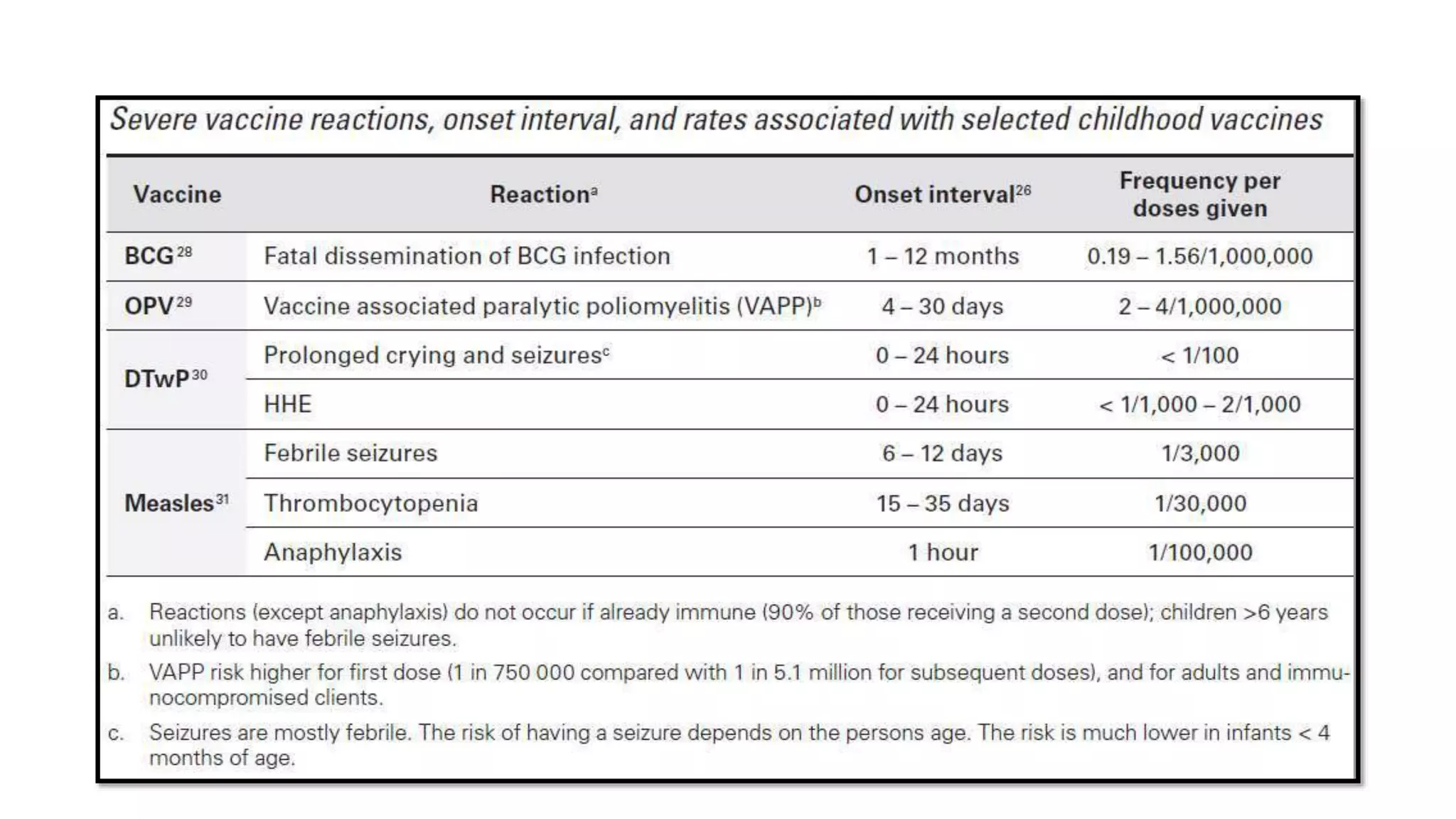
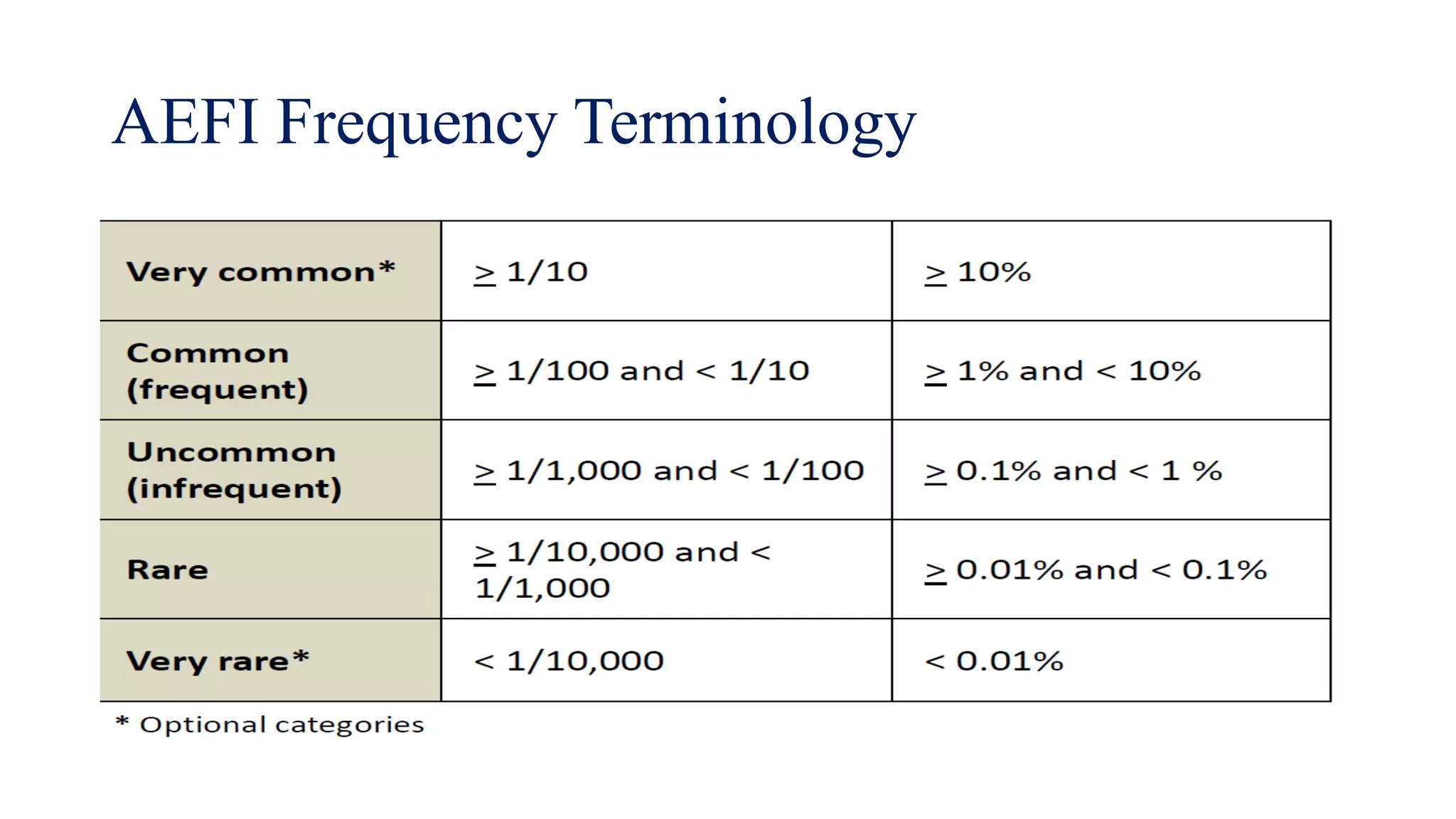
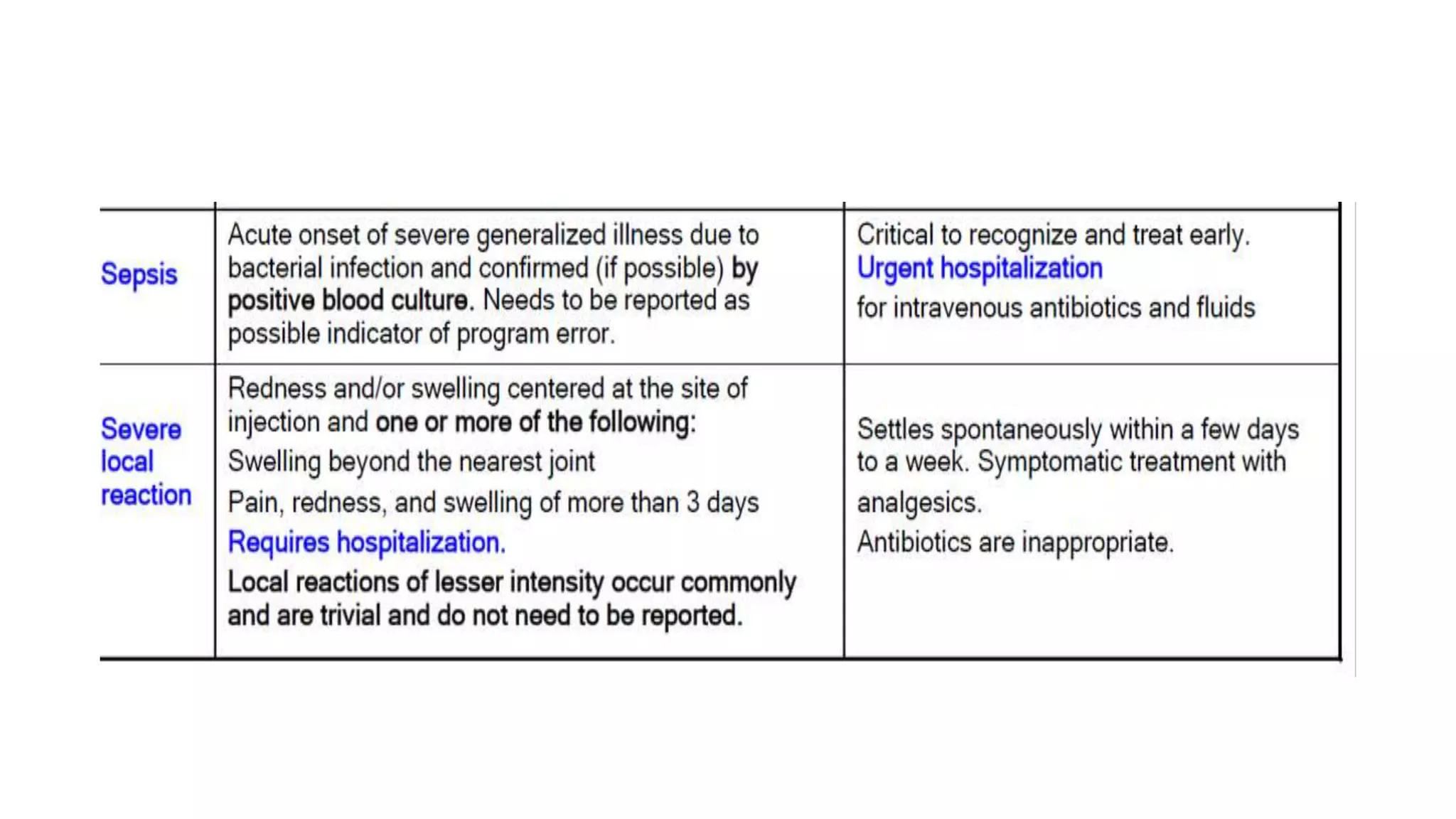
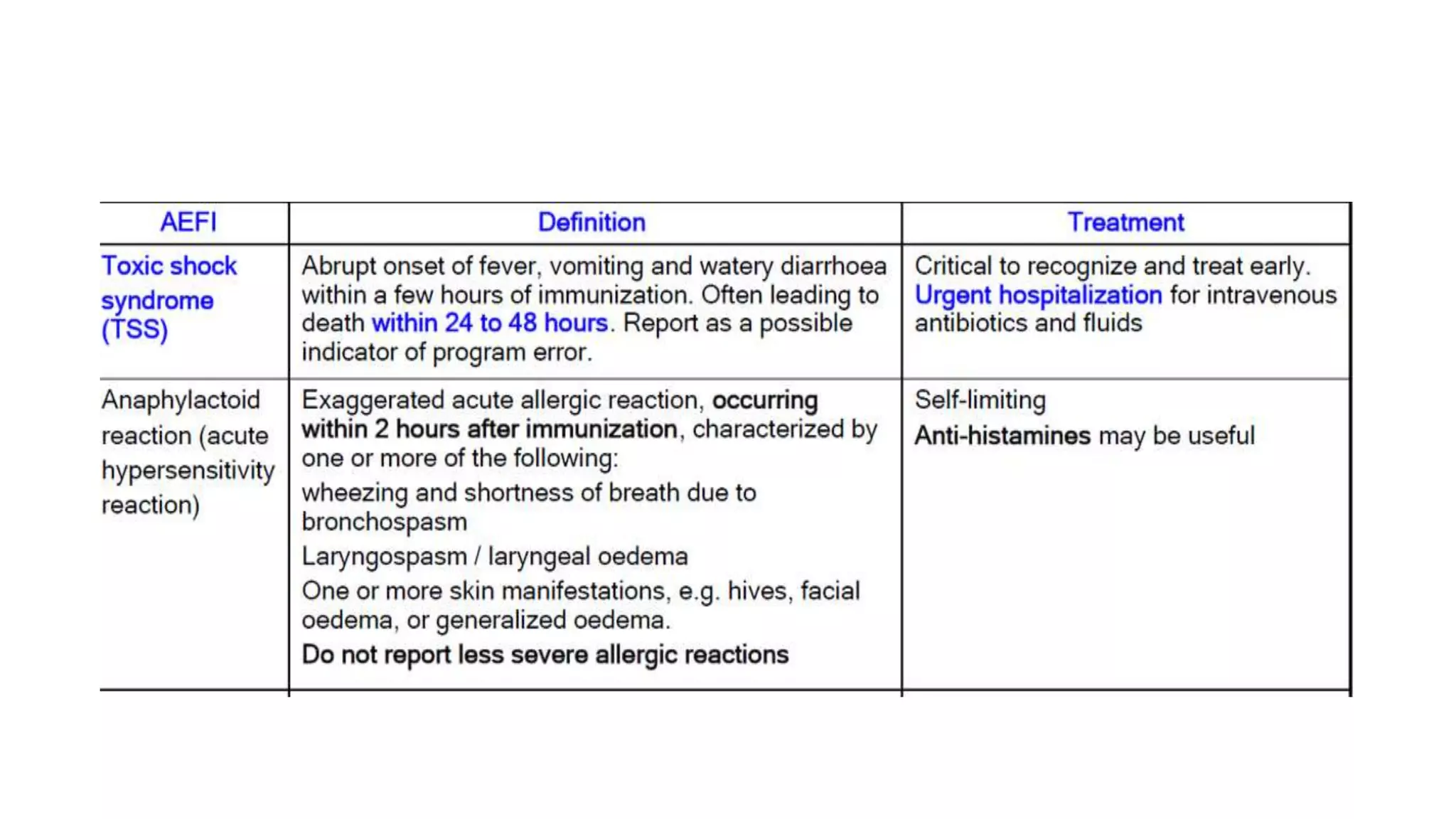
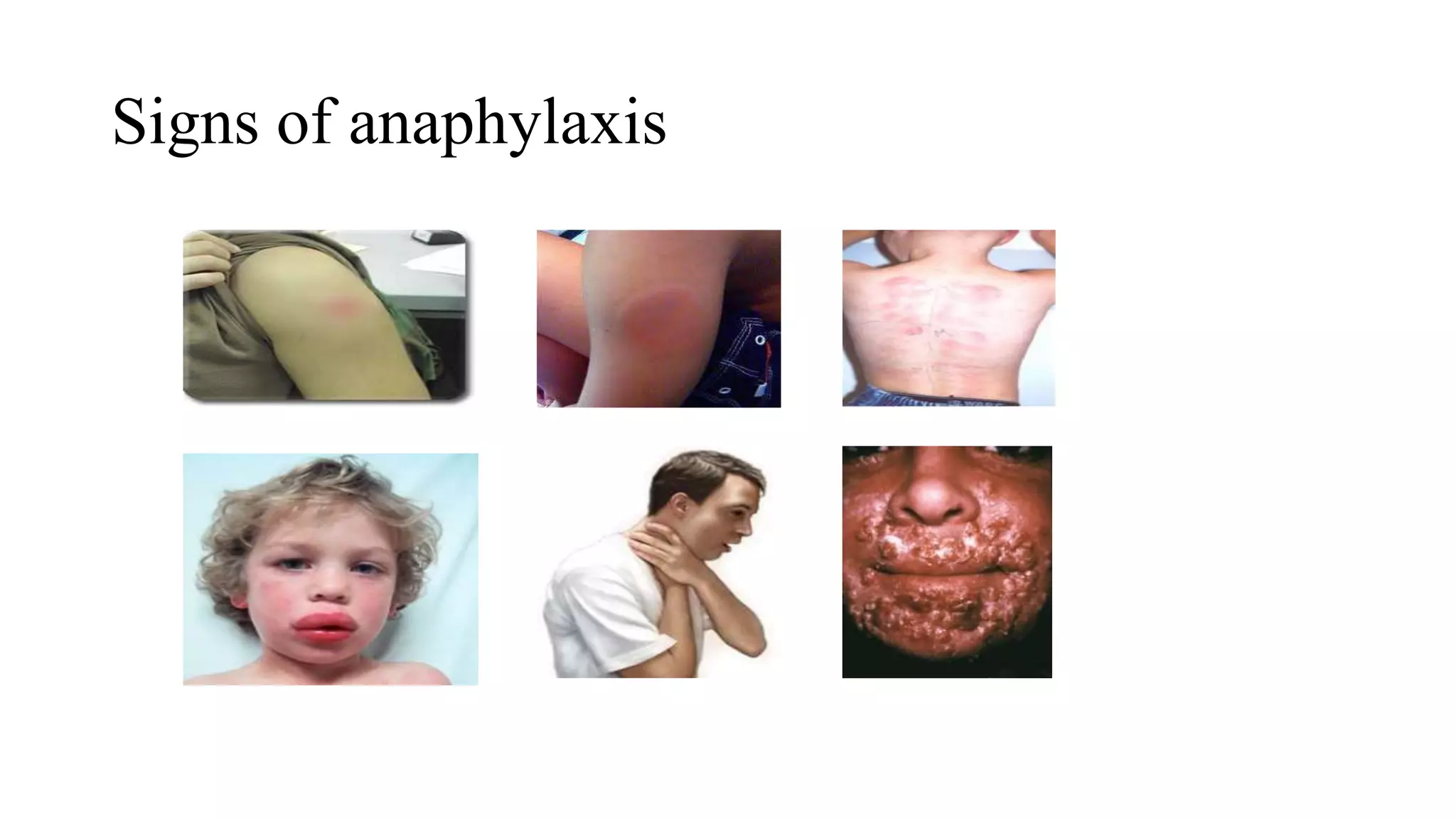
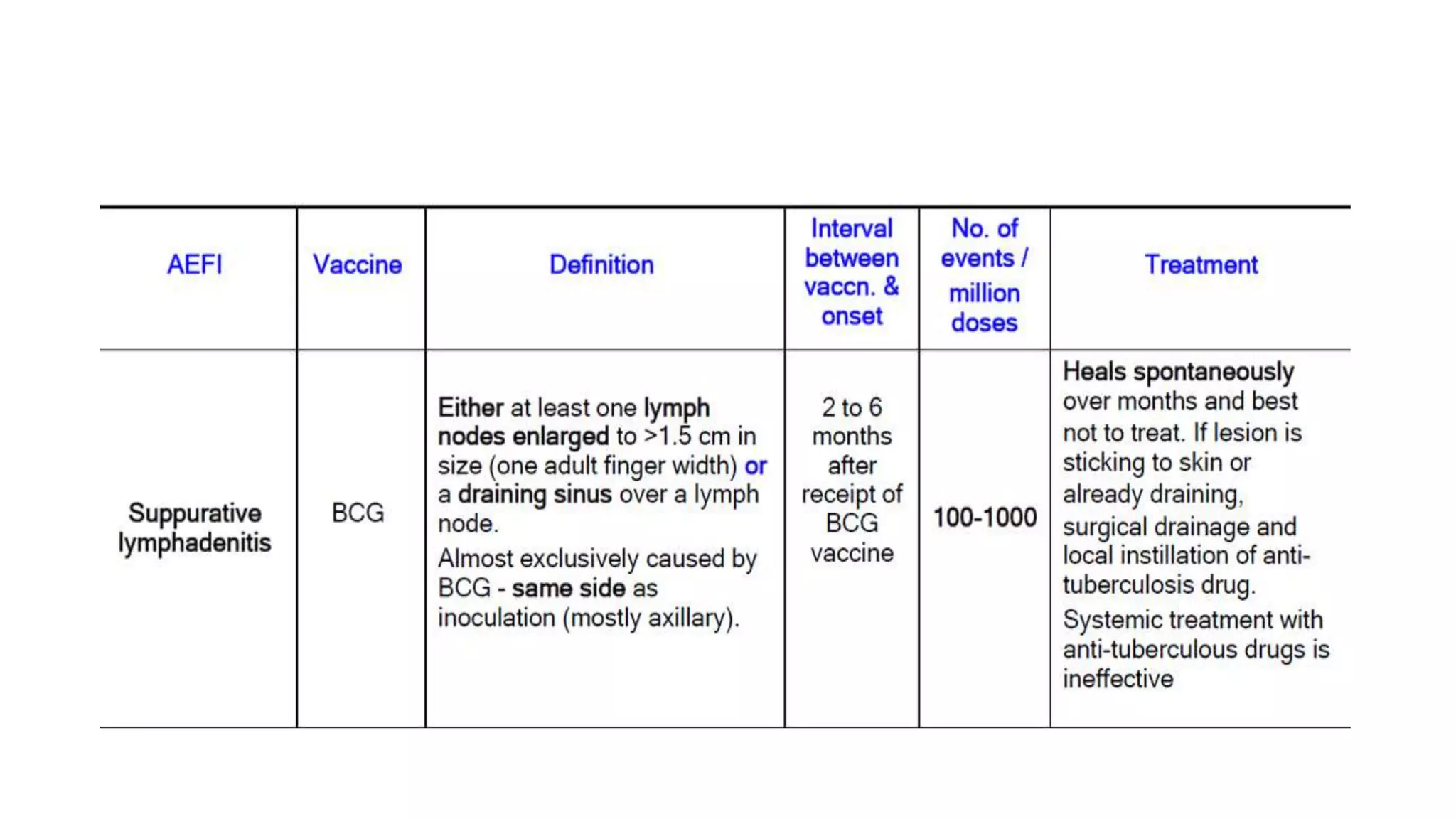
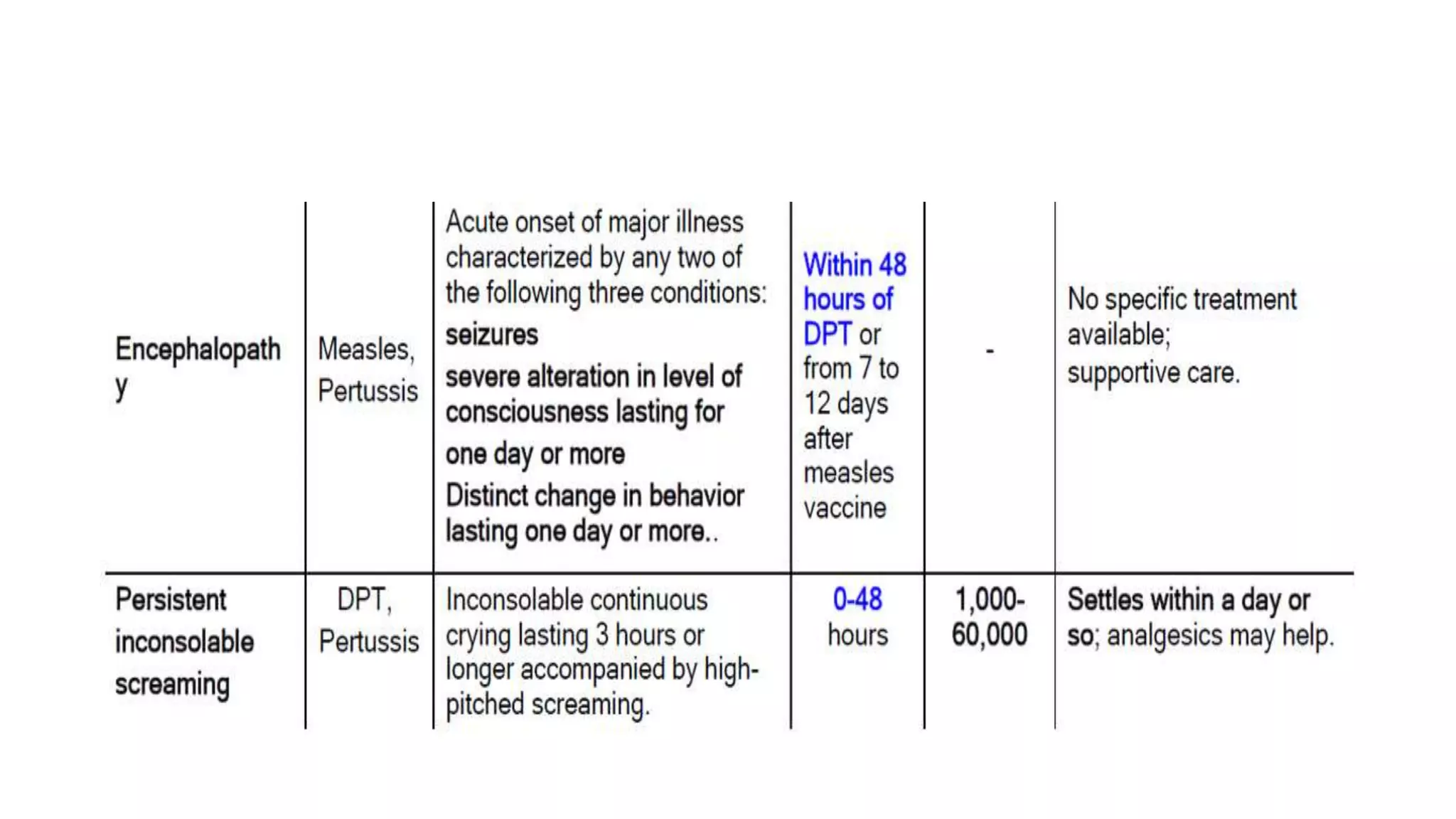
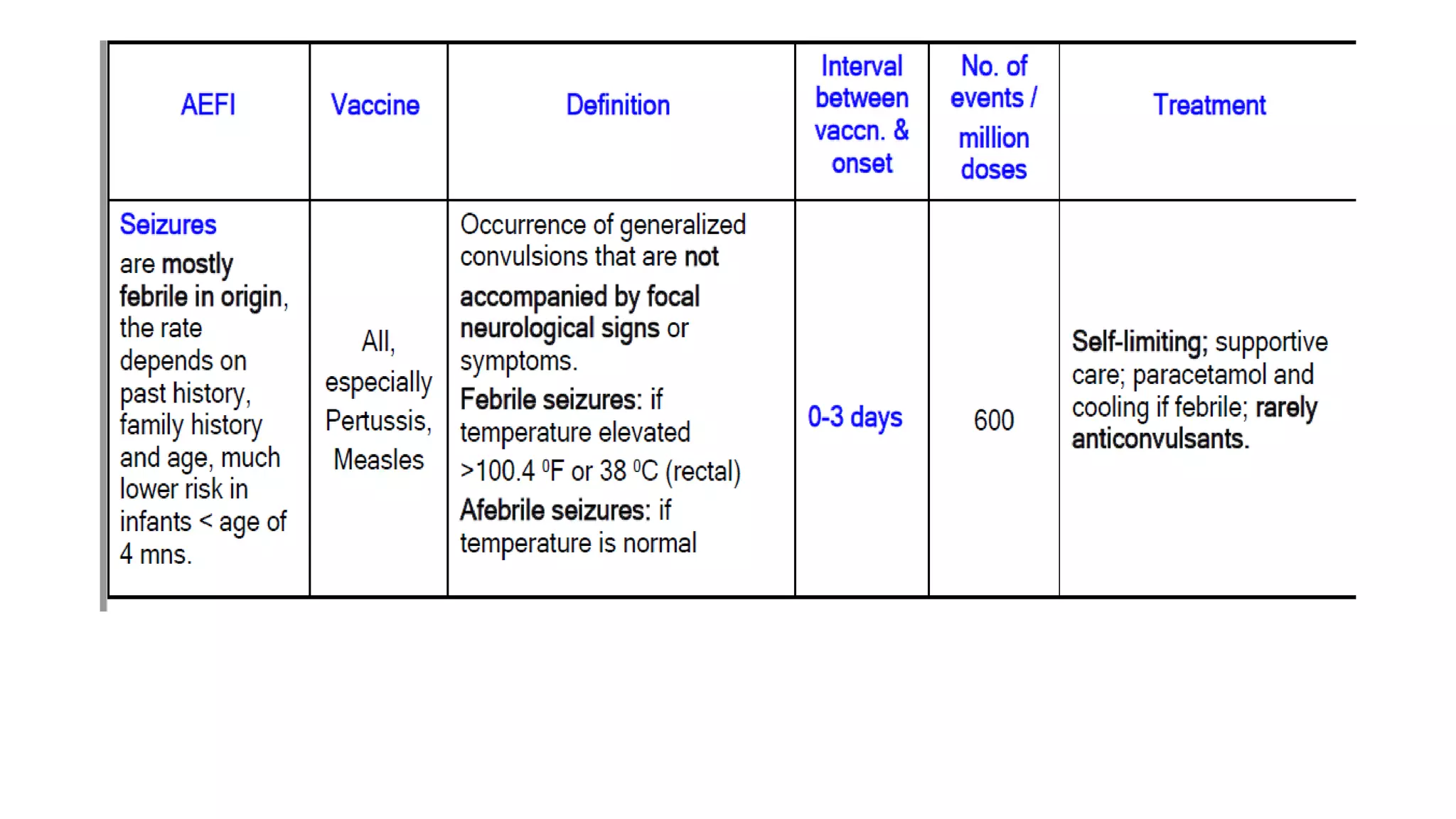
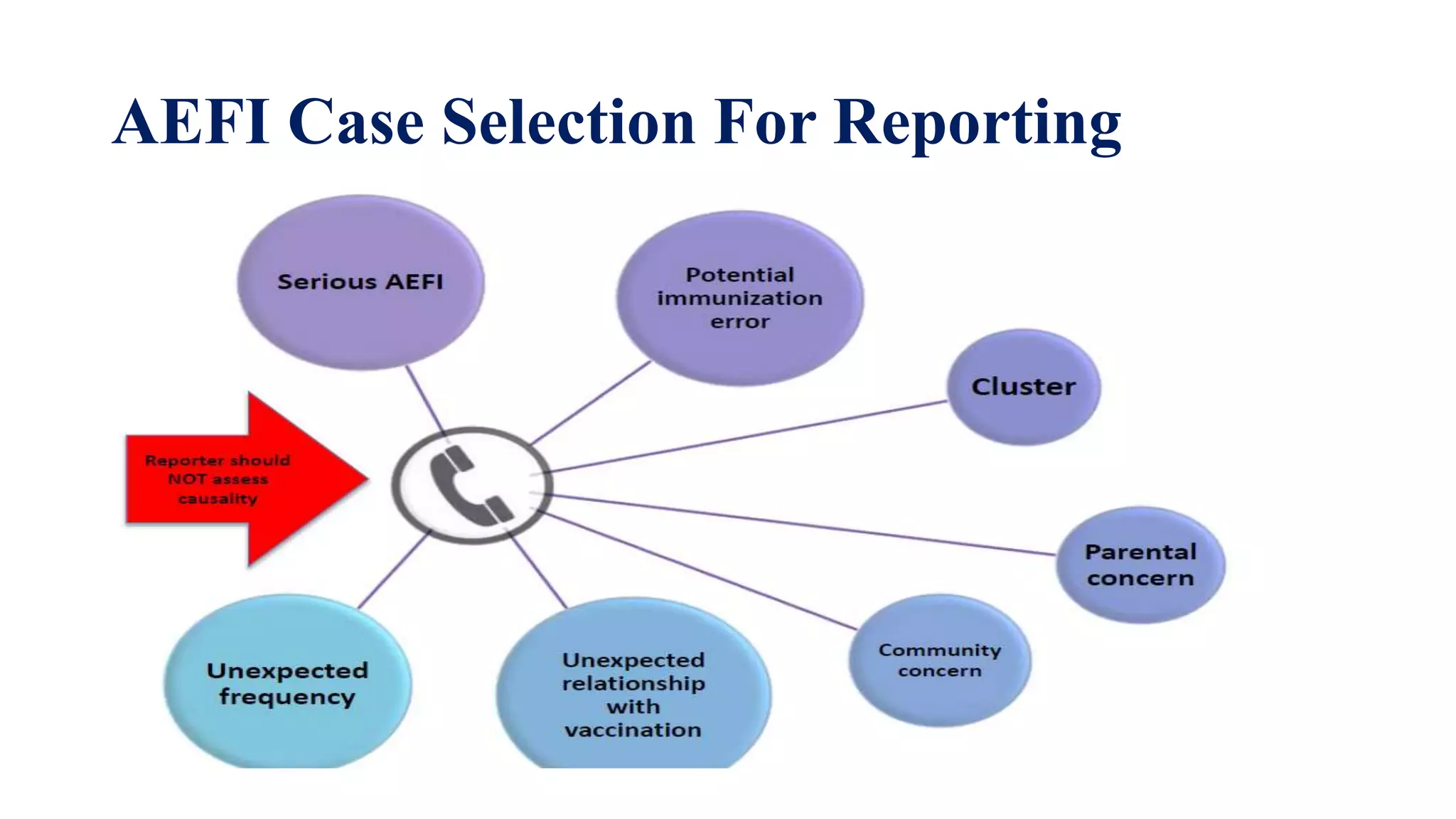
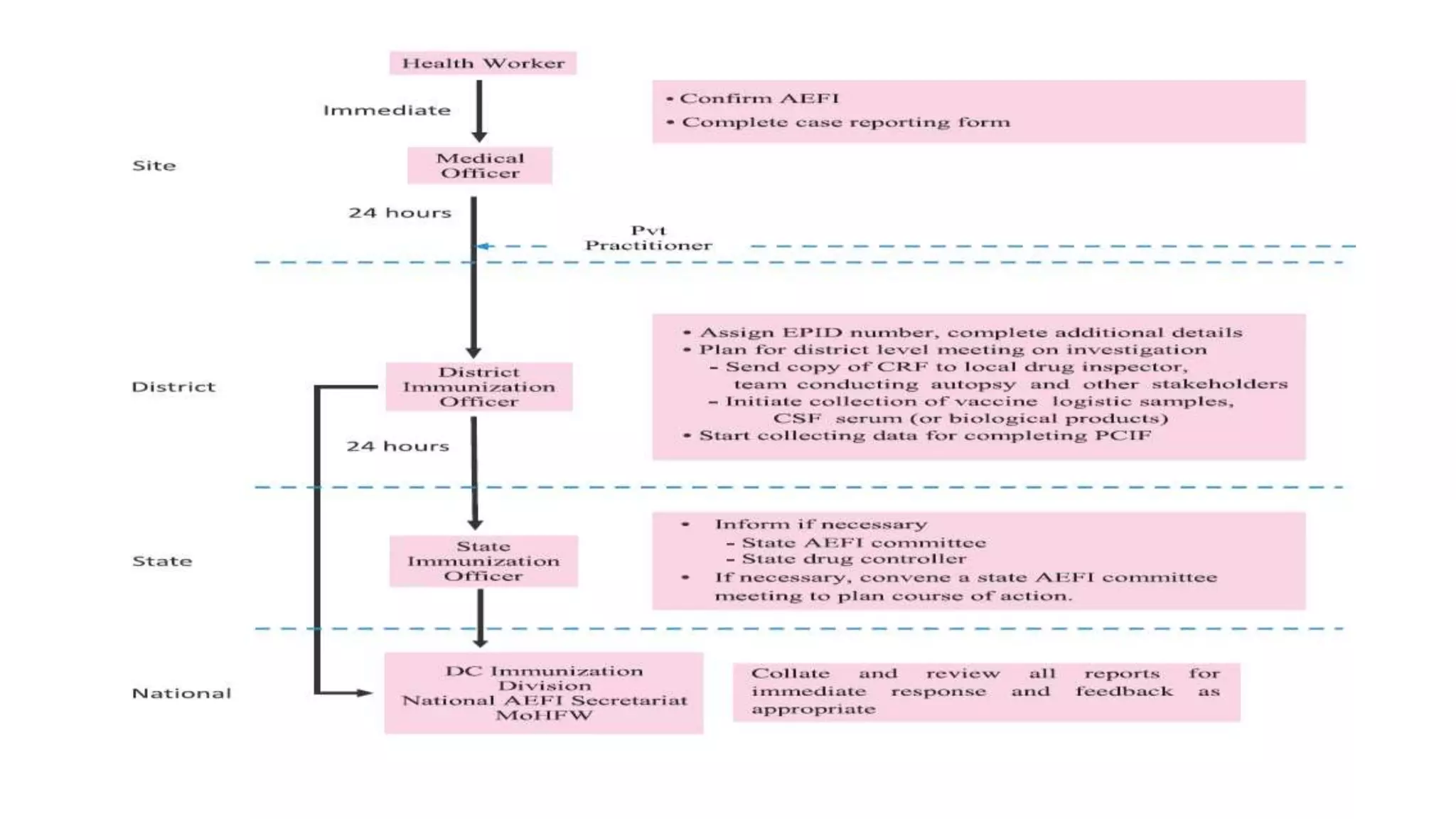
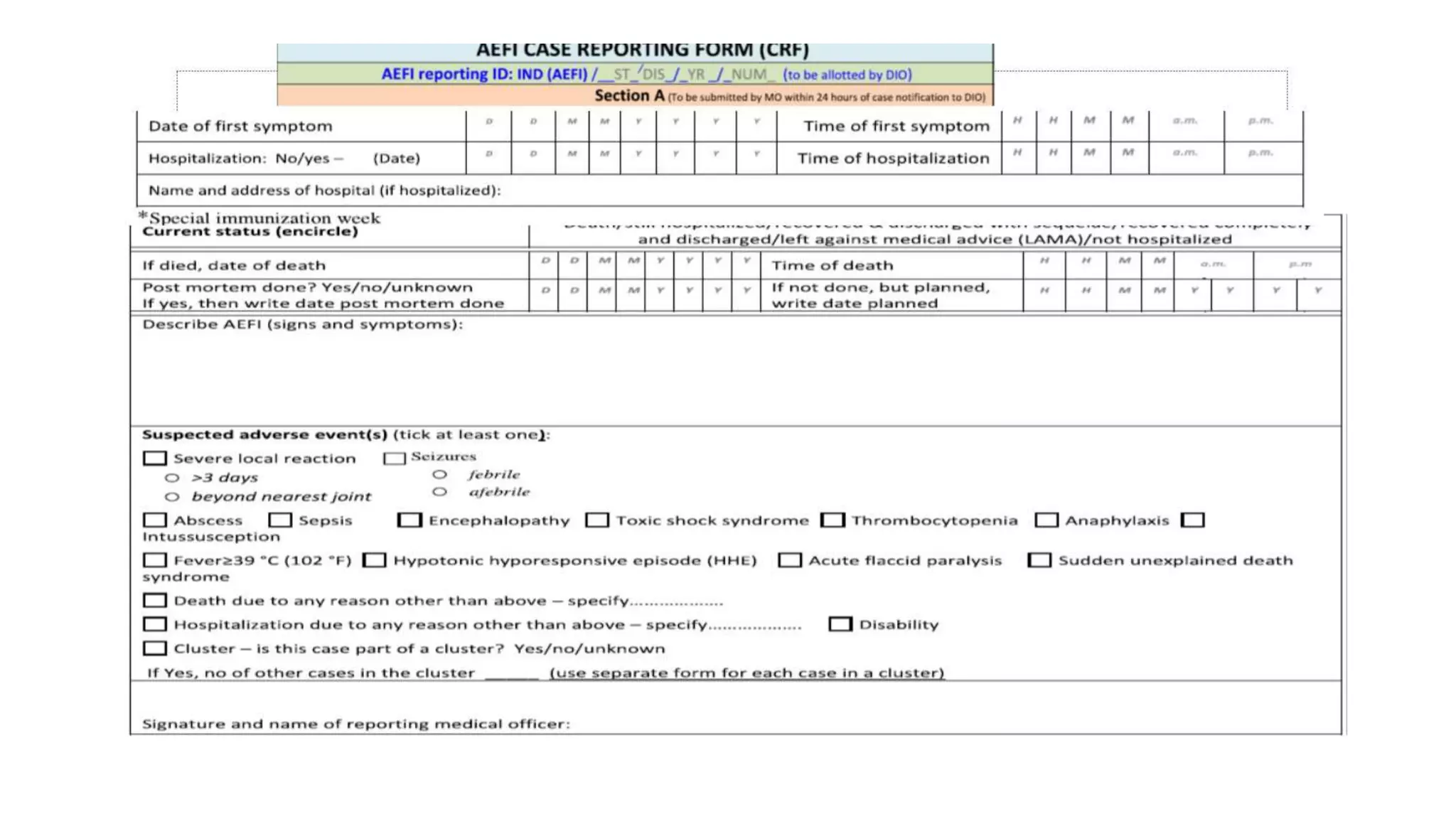
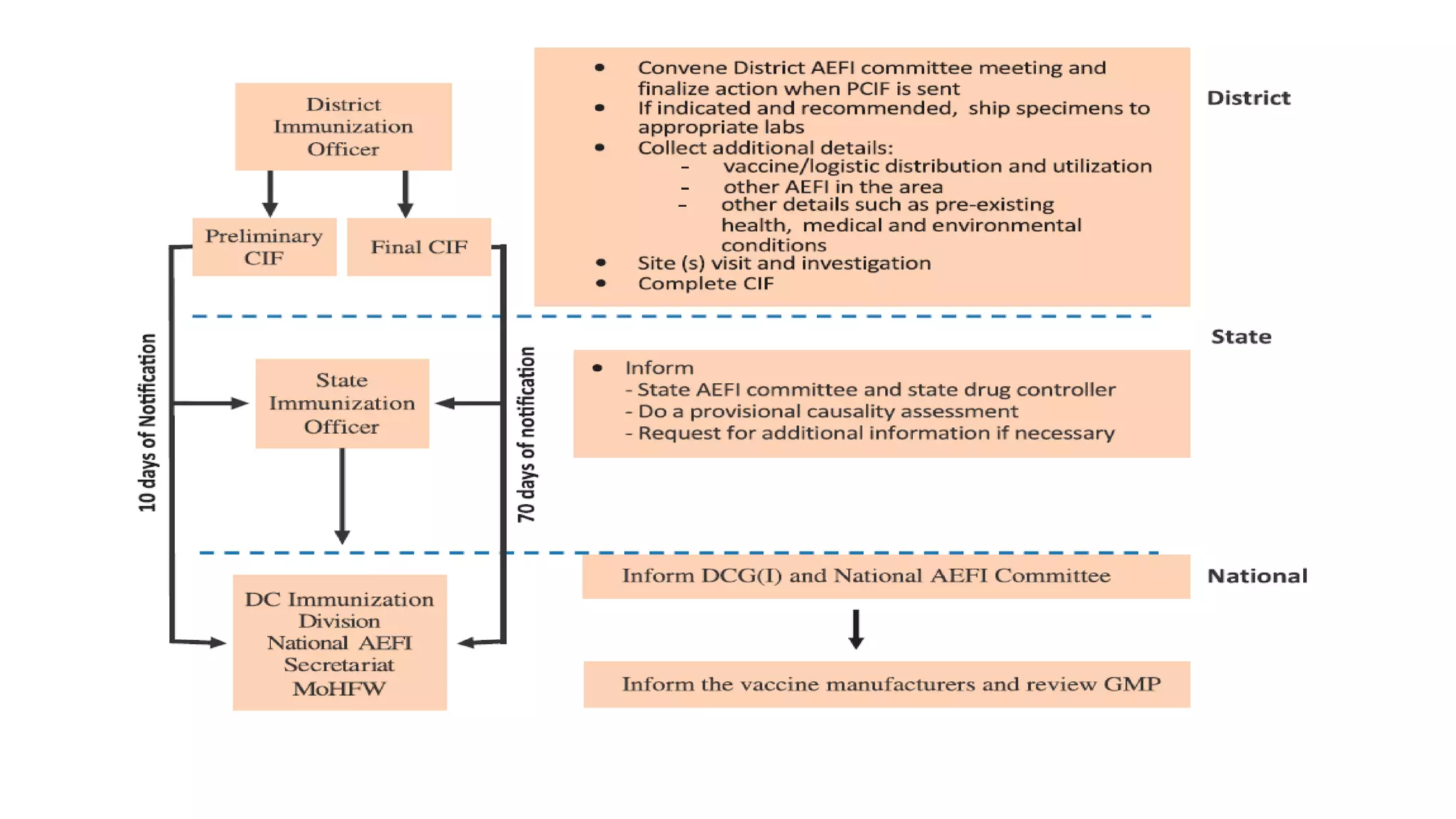
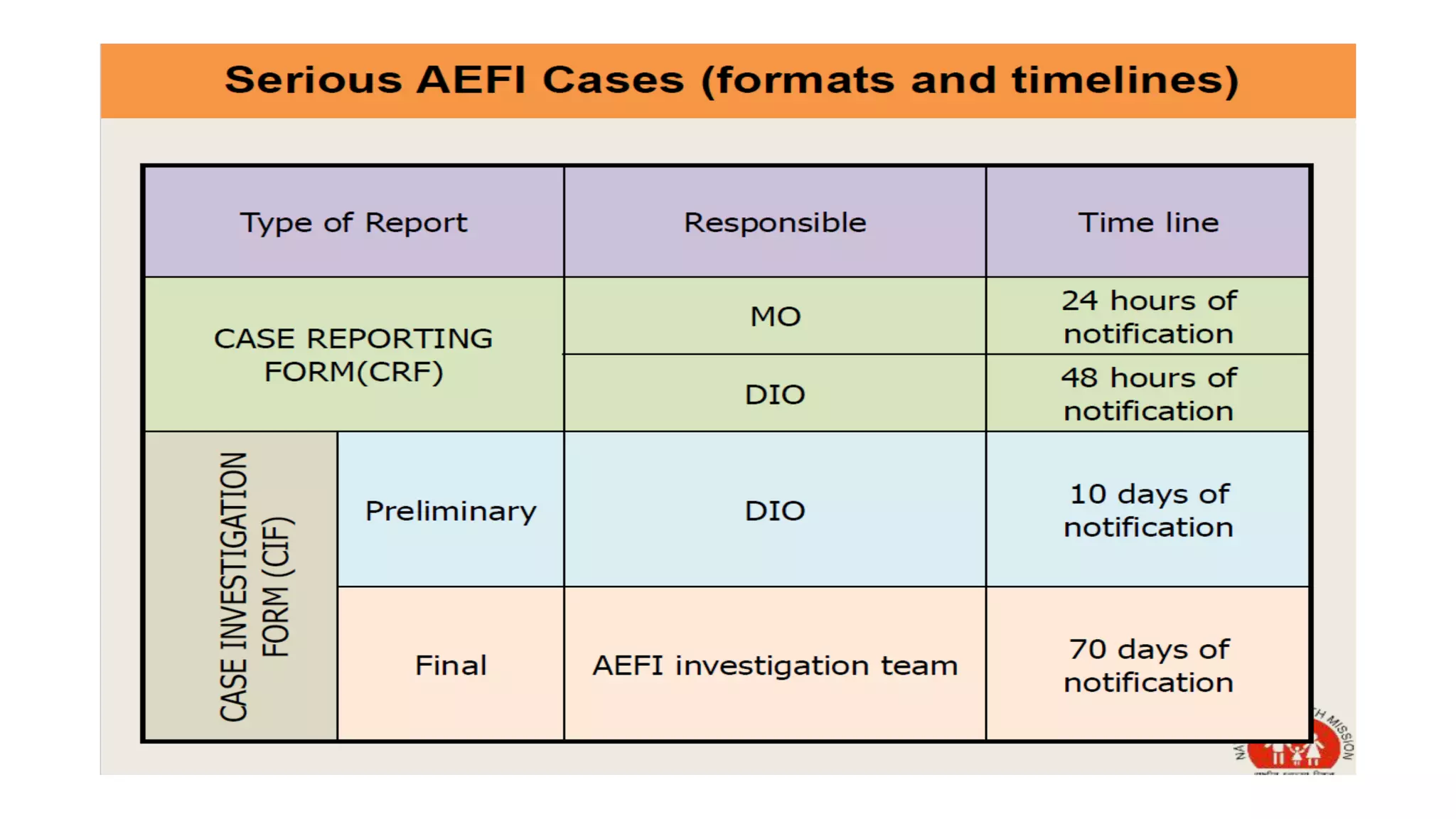
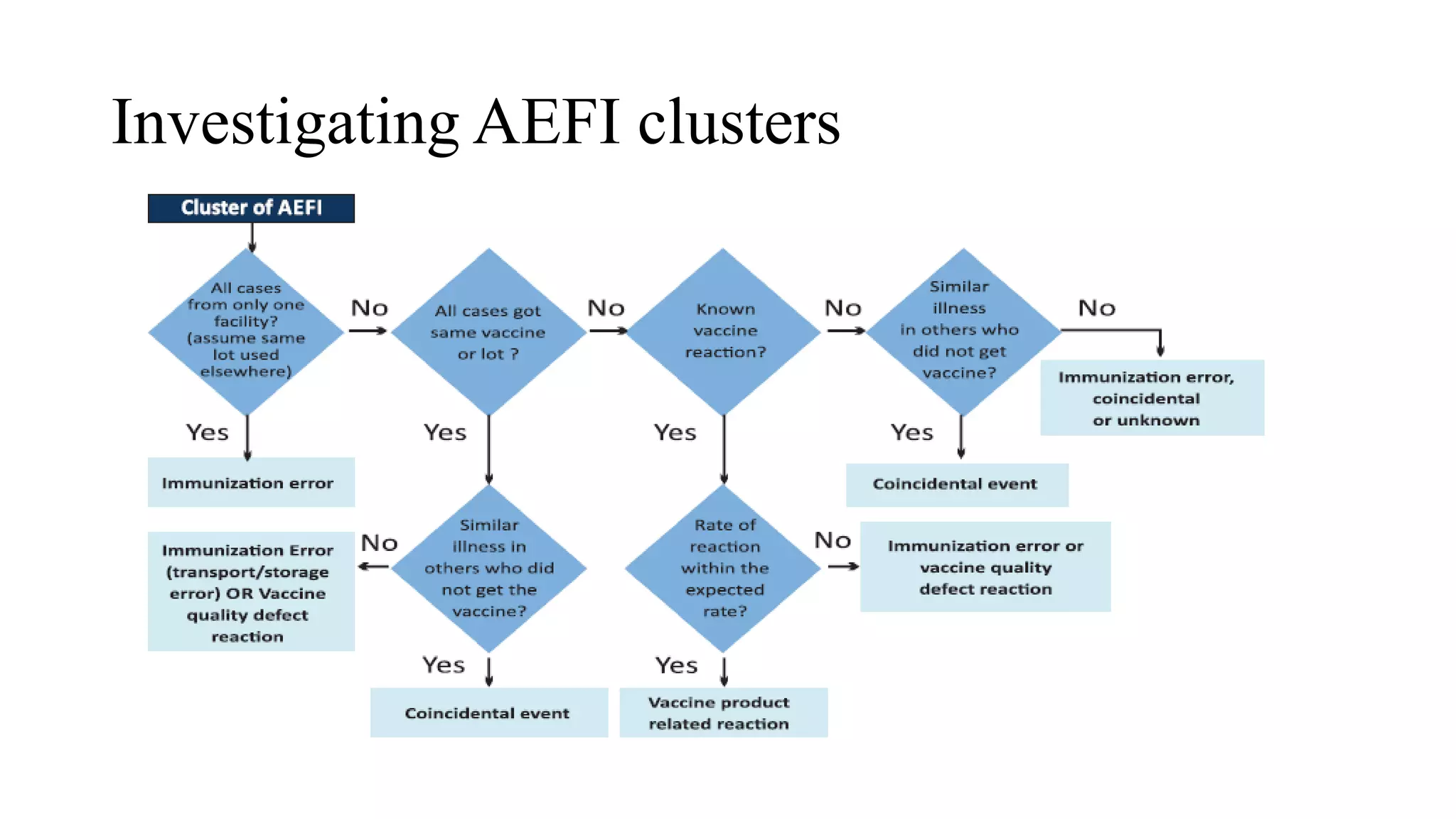
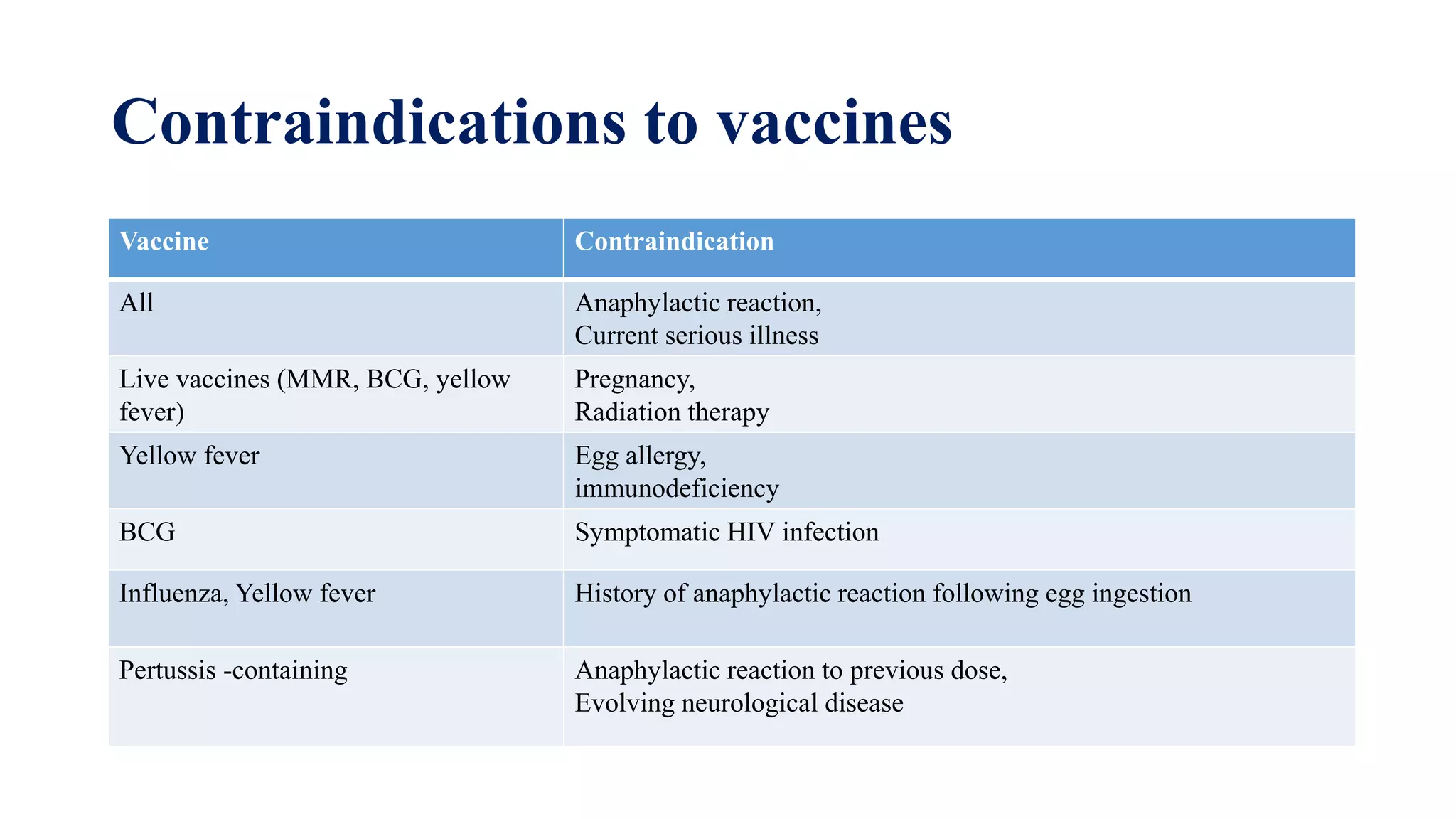
The document discusses adverse events following immunization (AEFI), detailing definitions, types, case definitions, and investigation steps while highlighting India's extensive immunization program. It emphasizes the importance of post-marketing surveillance for vaccine safety and provides guidelines for minimizing AEFI occurrences. The text also outlines contraindications for vaccines and the roles of reporting and investigating AEFIs to enhance public health safety.






![Introduction
• India is one of the largest manufacturers and exporters of vaccines in
the world.
• Apart from that, India runs one of the largest immunization programs
(Universal Immunization Program [UIP]) in the world for the
prevention of various vaccine-preventable diseases targeting
approximately 27 million newborns and 30 million pregnant women
every year.
• The detection of all possible AEs during the various phases of the
clinical trial is near impossible; hence, post marketing surveillance
(PMS) plays a major role in evaluating the safety of the vaccine.](https://image.slidesharecdn.com/aefippt-210826071923/75/AEFI-Adverse-event-following-immunization-by-Dr-Sonam-Aggarwal-7-2048.jpg)