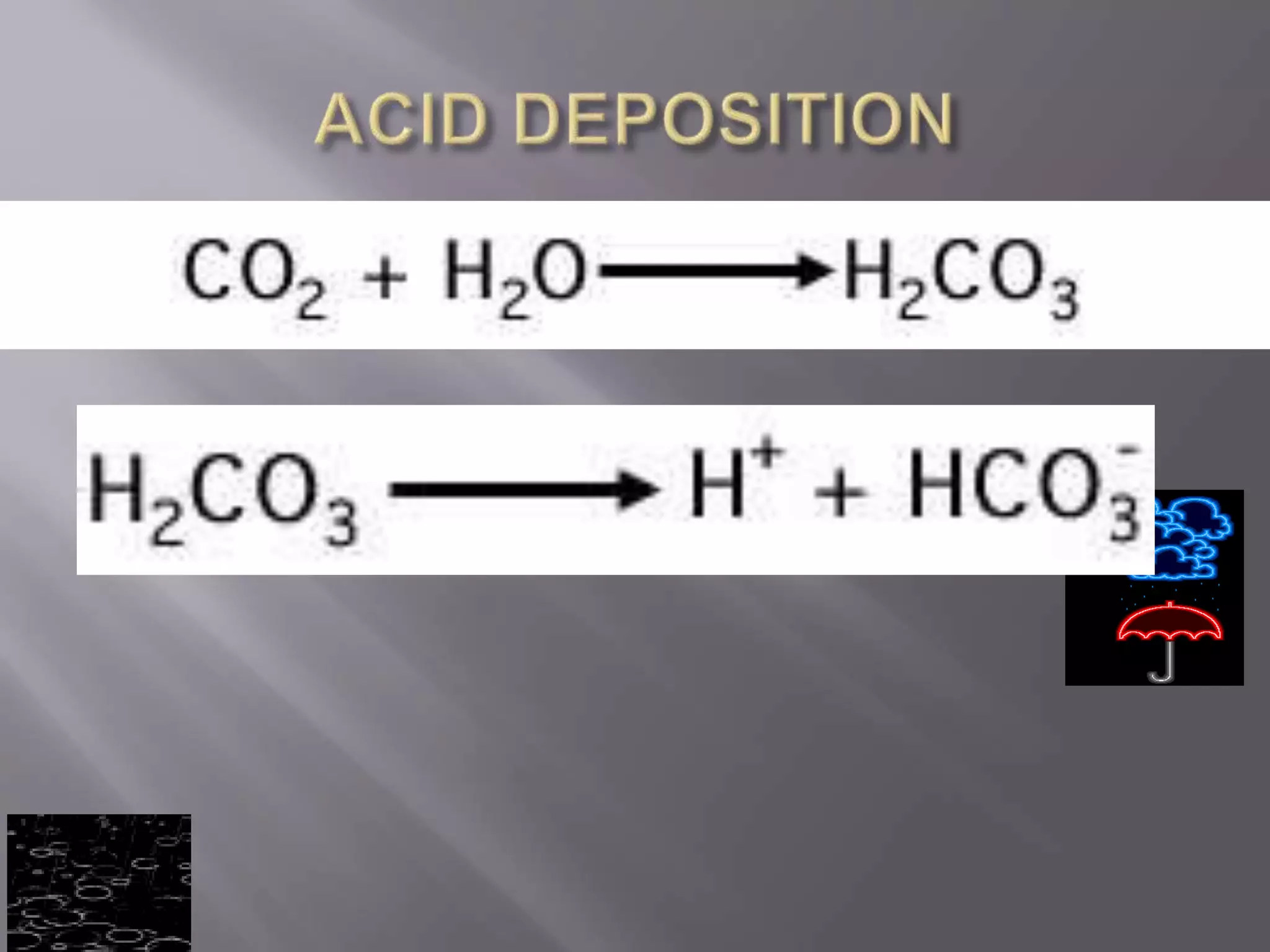
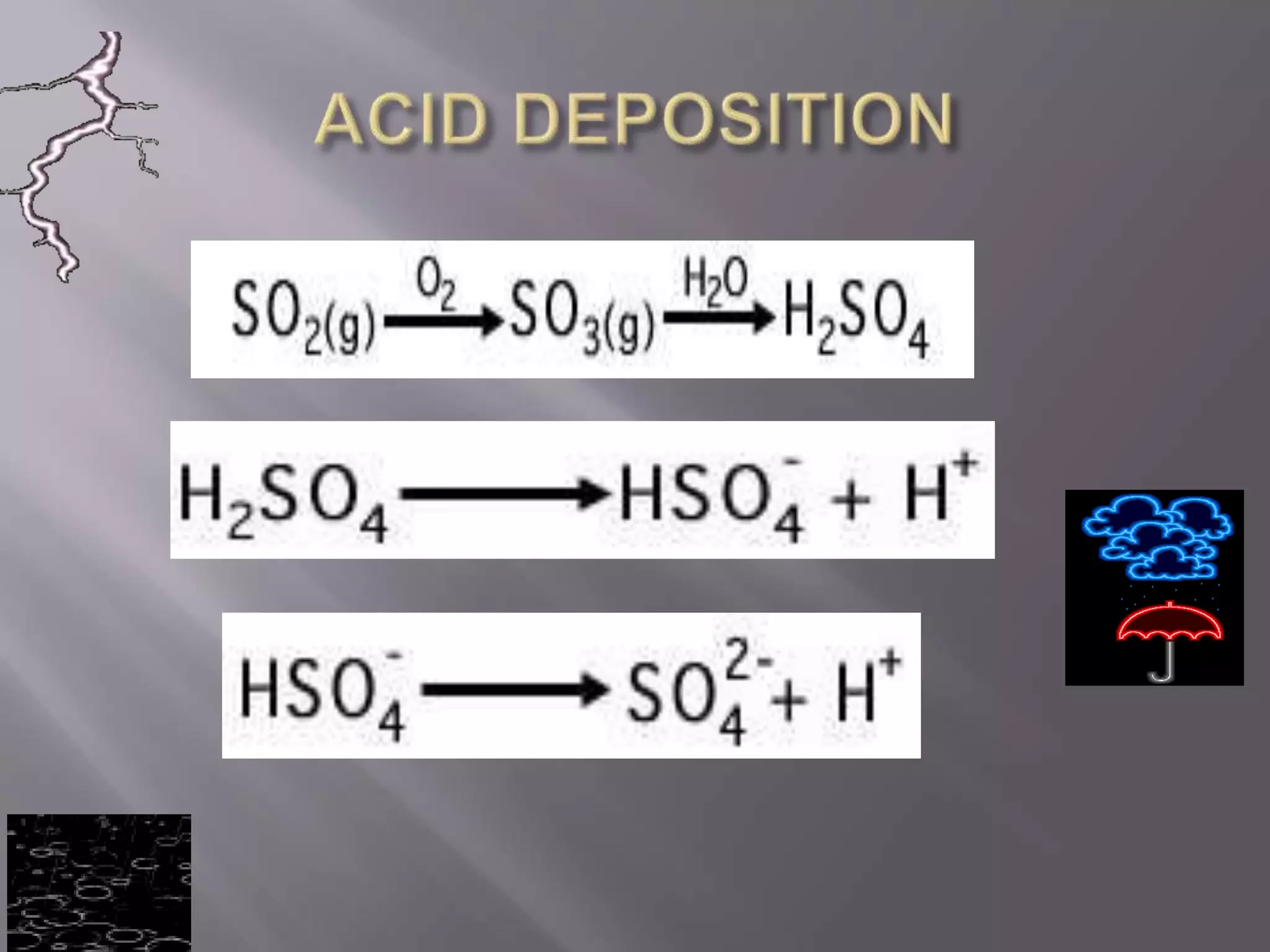
Acid deposition occurs when acids or acidic compounds accumulate on surfaces from atmospheric separation. It is caused by emissions like carbon dioxide, nitric oxide, and sulfur dioxide which react to form acids. The main sources are natural emissions and anthropogenic emissions from vehicles and coal burning.
While acid deposition can supply nutrients to soil, it often harms forests by leaching nutrients from soil and releasing aluminum, slowing or killing tree growth. It also harms aquatic organisms and biodiversity by creating conditions that individual fish cannot survive in or are killed by. Additionally, it damages materials like buildings, statues, and bridges made of marble, limestone, and sandstone through erosion. Affected areas include eastern US, eastern Europe, southeast China,