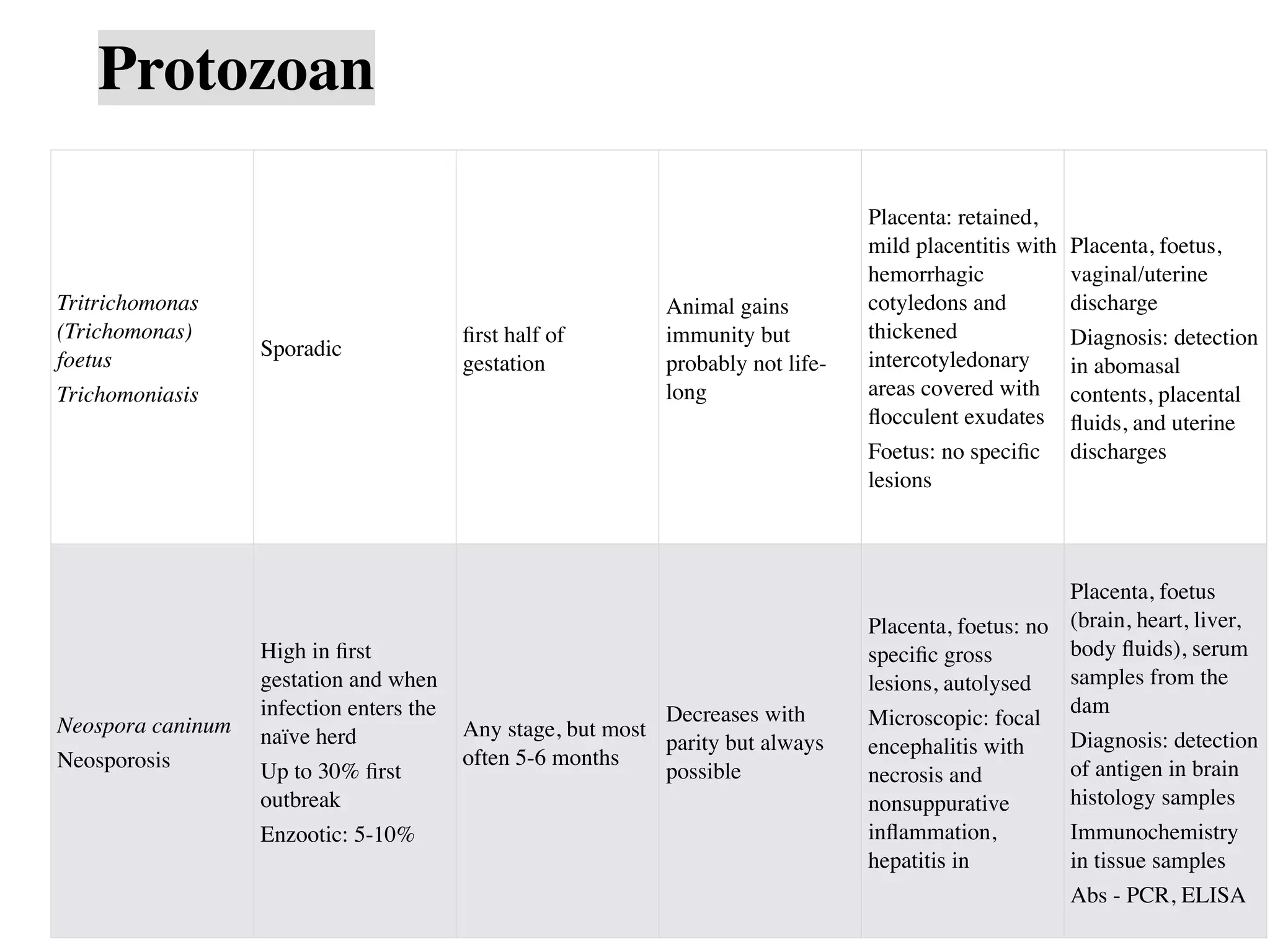
This document discusses causes of abortion in large animals. It describes infectious causes such as bacterial (e.g. Brucella abortus, Campylobacter fetus, Listeria monocytogenes), fungal (e.g. Aspergillus sp.), and protozoal (e.g. Neospora caninum, Tritrichomonas foetus) agents. It also discusses non-infectious causes including genetic factors, nutritional deficiencies, heat stress, and various toxins. The document provides details on abortion rates, timing, lesion presentation, and samples needed for diagnosis for many of the common infectious causes. It emphasizes the importance of rapid and proper sample collection and handling for determining the etiology of abortion