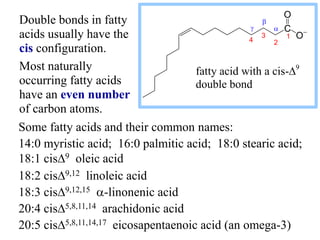
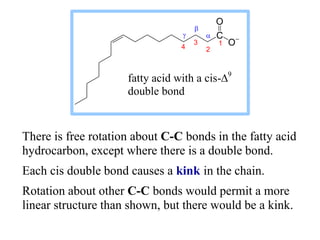
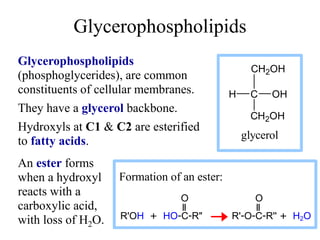
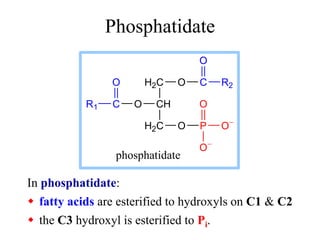
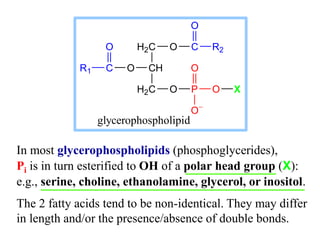
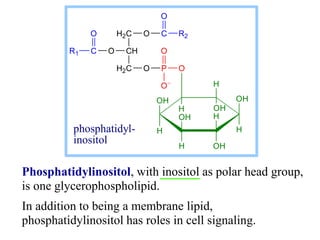
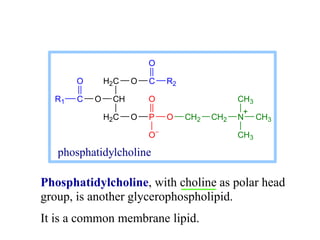
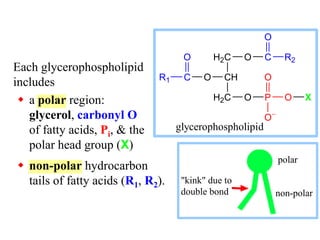
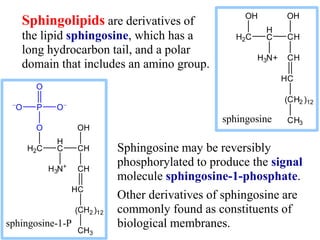
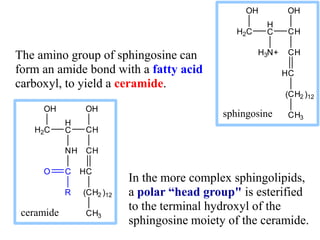
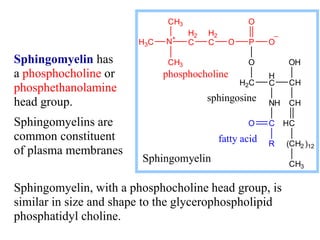
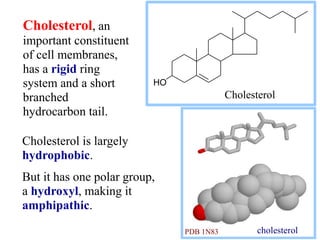
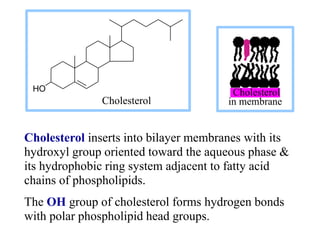
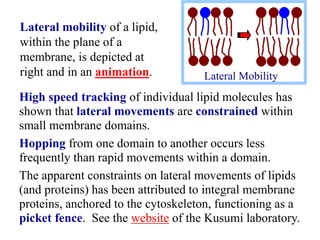
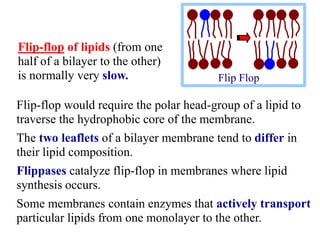
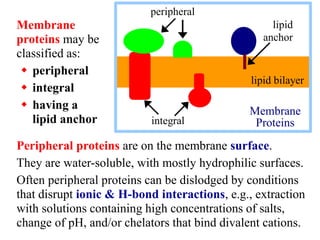
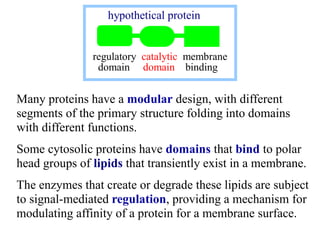
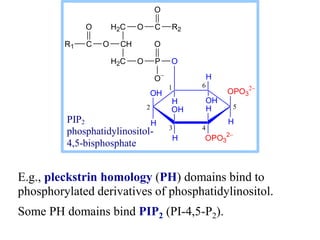
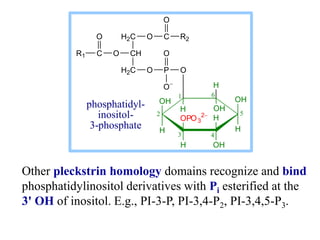
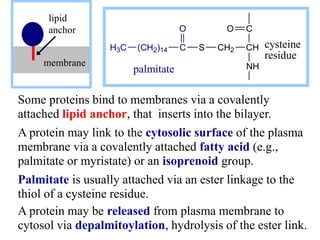
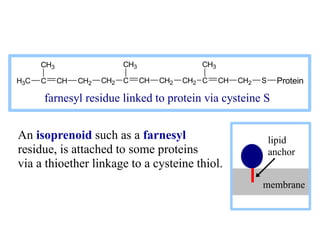
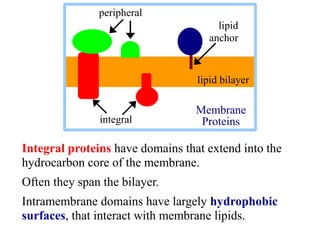
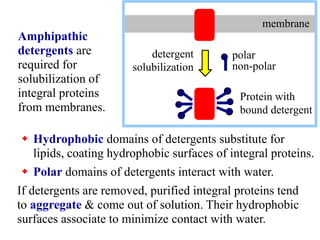
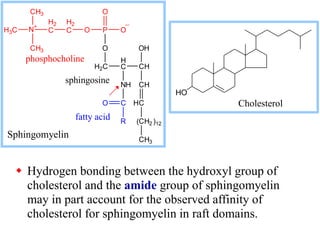
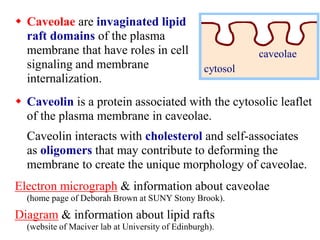
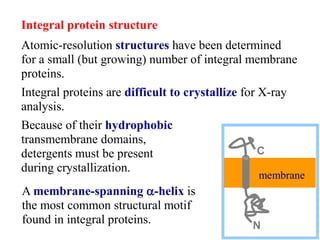
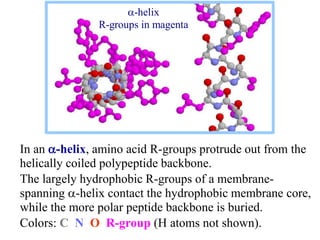
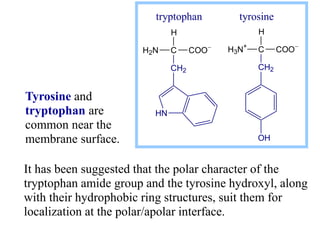
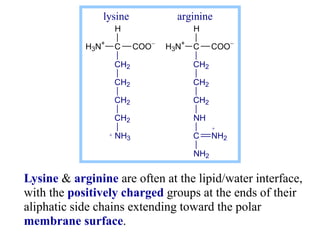
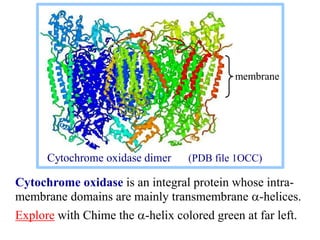
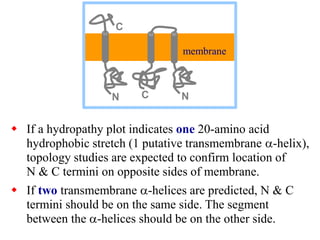
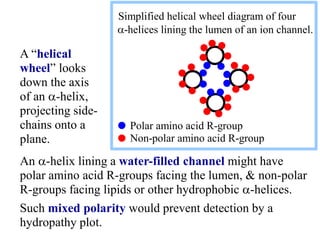
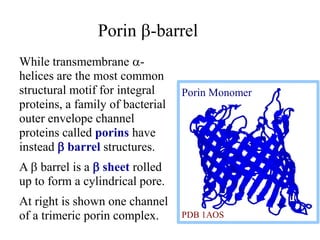
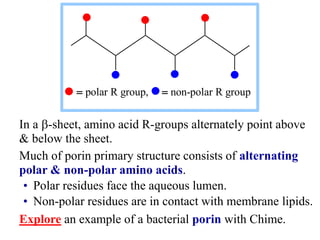
This document discusses lipids and membranes. It describes the basic structures of lipids like fatty acids, glycerophospholipids, sphingolipids, and cholesterol. These lipids can assemble into structures like micelles and bilayers in aqueous environments due to their amphipathic nature. Bilayers allow for the formation of cell membranes. Membranes contain proteins that can be integral, peripheral, or lipid-anchored. Lipid composition and proteins influence membrane properties like fluidity.