Shape of noble gas compounds XeFx
•Download as PPTX, PDF•
0 likes•447 views
The document discusses the shapes of XeF2, XeF4, and XeF6 based on the hybridization of xenon and fluorine. XeF2 has a linear shape due to sp3d hybridization of xenon and XeF4 adopts an octahedral geometry from sp3d2 hybridization. XeF6 forms a distorted octahedron structure through sp3d3 hybridization of xenon.
Report
Share
Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Semi Micro Qualitative Organic chemistry lab manual

Semi Micro Qualitative Organic chemistry lab manual
photo chemistry of ligand in coordination compound

photo chemistry of ligand in coordination compound
Molecular orbitals diagrams of hexacyanoferrate(III) and hexafluoroferrate(III)

Molecular orbitals diagrams of hexacyanoferrate(III) and hexafluoroferrate(III)
Labile & inert and substitution reactions in octahedral complexes

Labile & inert and substitution reactions in octahedral complexes
Part 2, Substitution reactions in square planar complexes, Factors.pptx

Part 2, Substitution reactions in square planar complexes, Factors.pptx
More from Mithil Fal Desai
More from Mithil Fal Desai (20)
How long could one survive in a perfectly airtight room.docx

How long could one survive in a perfectly airtight room.docx
Writing Third Year Project Report: Some Handy Guidelines

Writing Third Year Project Report: Some Handy Guidelines
Determine the strength in grams per litre of a given AgNO3 solution being pro...

Determine the strength in grams per litre of a given AgNO3 solution being pro...
Estimation of water of crystallization in mohr’s salt by titrating with stand...

Estimation of water of crystallization in mohr’s salt by titrating with stand...
Molecular orbitals diagrams without pi interactions

Molecular orbitals diagrams without pi interactions
Recently uploaded
Recently uploaded (20)
Including Mental Health Support in Project Delivery, 14 May.pdf

Including Mental Health Support in Project Delivery, 14 May.pdf
Personalisation of Education by AI and Big Data - Lourdes Guàrdia

Personalisation of Education by AI and Big Data - Lourdes Guàrdia
dusjagr & nano talk on open tools for agriculture research and learning

dusjagr & nano talk on open tools for agriculture research and learning
Diuretic, Hypoglycemic and Limit test of Heavy metals and Arsenic.-1.pdf

Diuretic, Hypoglycemic and Limit test of Heavy metals and Arsenic.-1.pdf
Michaelis Menten Equation and Estimation Of Vmax and Tmax.pptx

Michaelis Menten Equation and Estimation Of Vmax and Tmax.pptx
21st_Century_Skills_Framework_Final_Presentation_2.pptx

21st_Century_Skills_Framework_Final_Presentation_2.pptx
Shape of noble gas compounds XeFx
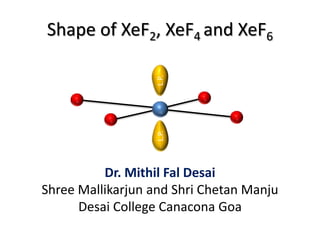
- 1. Shape of XeF2, XeF4 and XeF6 Dr. Mithil Fal Desai Shree Mallikarjun and Shri Chetan Manju Desai College Canacona Goa L.P.L.P.
- 2. Hybridisation in [XeF2] Xe (Z=54) [Kr] 4d10, 5s2,5p6 Xe in excited state 5s 5p 5d Xe in hybrid state Xe in ground state sp3d F (Z=9) [He] 2s2, 2p5 pentagonal bipyramid?
- 3. L.P. L.P. L.P. Linear Shape of XeF2 = fluorine = xenon
- 4. Hybridisation in XeF4 Xe (Z=54) [Kr] 4d10, 5s2,5p6 Xe in excited state 5s 5p 5d Xe in hybrid state Xe in ground state sp3d2 F (Z=9) [He] 2s2, 2p5 Octahedral
- 5. L.P.L.P. Square planar Shape of XeF4
- 6. Hybridisation in XeF6 Xe (Z=54) [Kr] 4d10, 5s2,5p6 Xe in excited state 5s 5p 5d Xe in hybrid state Xe in ground state sp3d3 F (Z=9) [He] 2s2, 2p5 Shape?
- 7. Shape of XeF6 Distorted octahedron