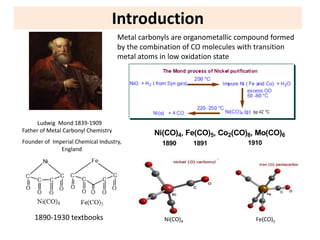
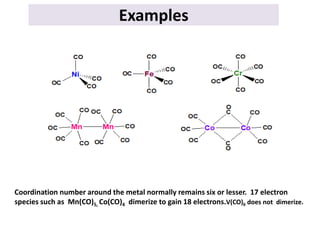
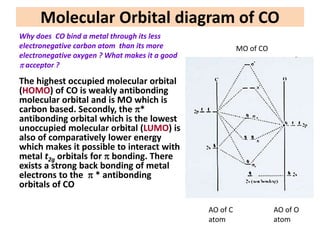
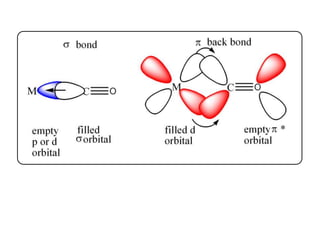
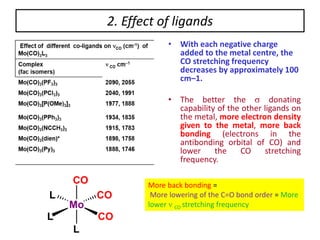
This document provides an overview of metal carbonyls. It discusses how metal carbonyls are formed from transition metals and carbon monoxide, and examples like nickel tetracarbonyl and iron pentacarbonyl. The molecular orbital diagram of carbon monoxide is shown, explaining why it can participate in pi-backbonding. Infrared spectroscopy is described as a useful technique for analyzing metal carbonyls, as it can distinguish terminal from bridging carbonyl ligands based on the infrared absorption frequency. Factors like metal charge and other ligands that affect the carbonyl stretching frequency are also outlined. Finally, some applications of infrared spectra of metal carbonyls are mentioned.