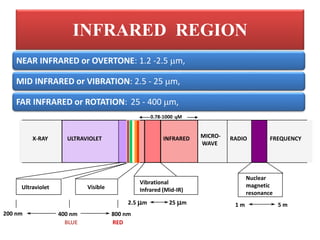
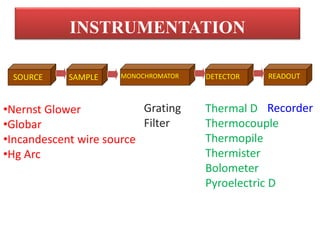
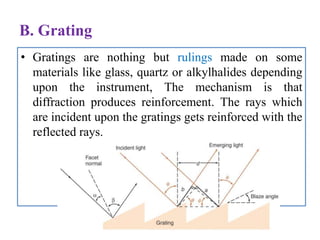
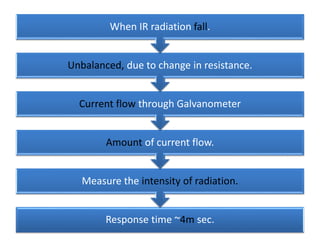
The document discusses infrared (IR) spectroscopy, which analyzes the interaction of infrared radiation with matter. IR spectroscopy can provide information about a compound's chemical structure and molecular structure by measuring its absorption of IR radiation. It is widely used to analyze organic materials and some inorganic molecules. The document then describes various components of IR instrumentation, including IR radiation sources like the Nernst glower and globar, monochromators that separate wavelengths, sample cells and techniques, and detectors like thermocouples, bolometers, and thermistors that measure the radiation absorbed.