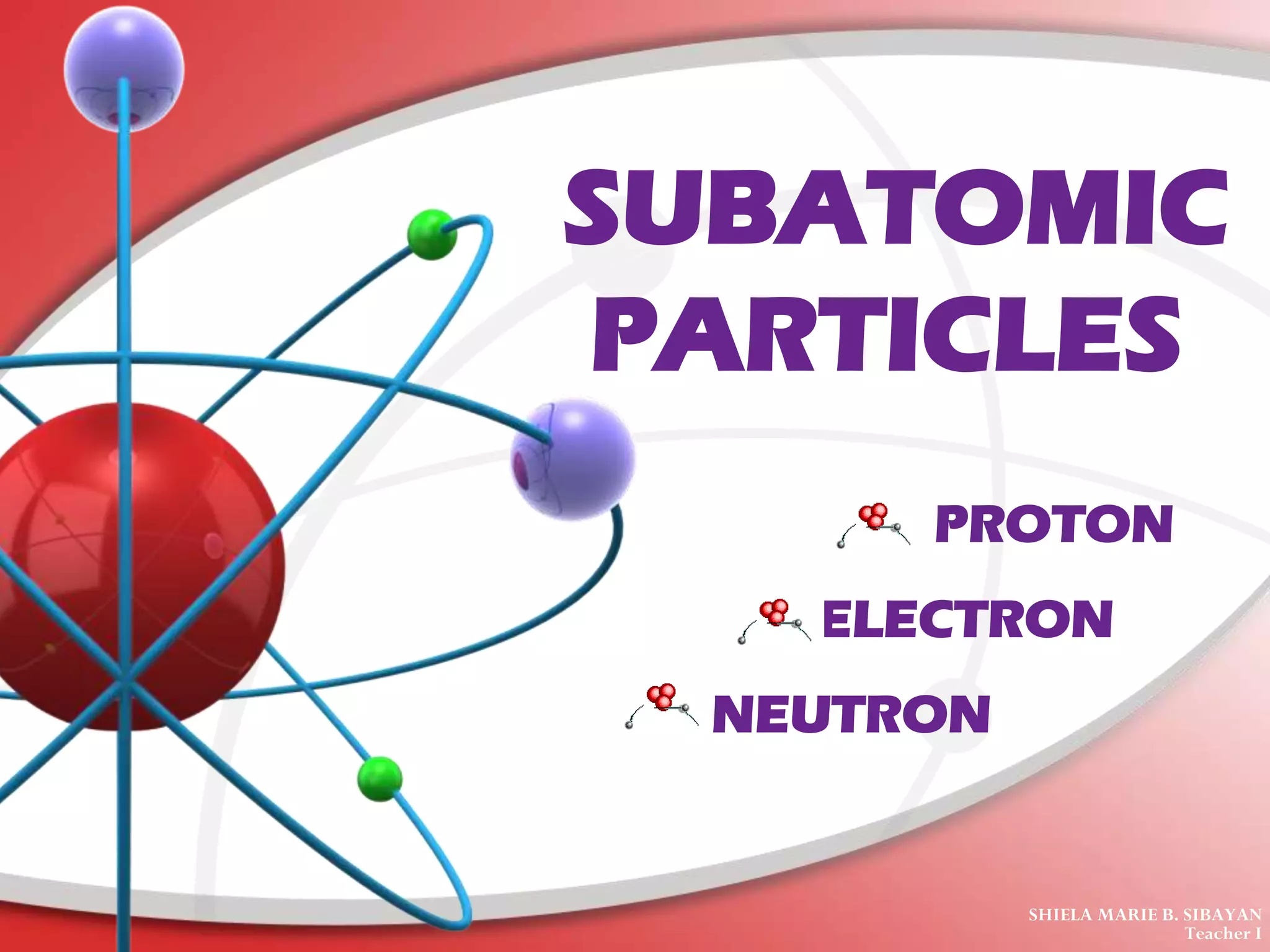
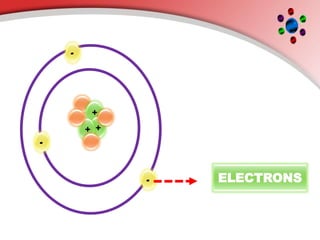
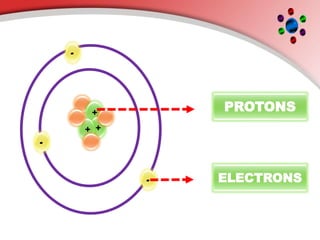
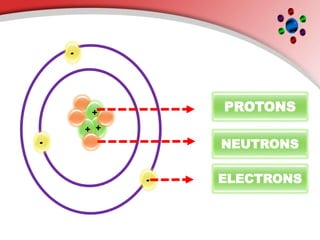
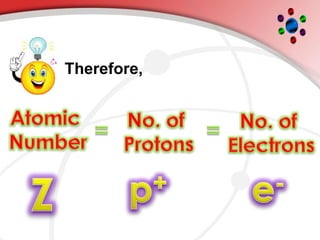
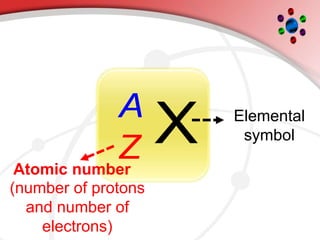
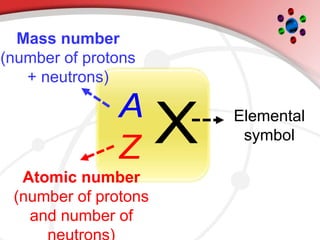
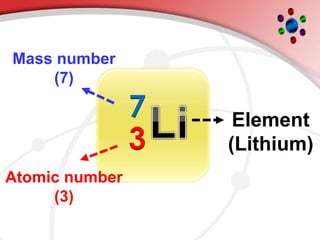
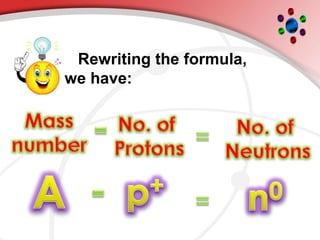
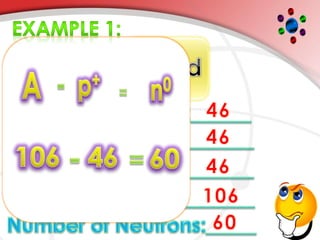
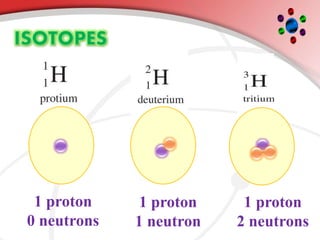
The document describes the three main subatomic particles that make up atoms: protons, neutrons, and electrons. Protons are positively charged and located in the atom's nucleus. Neutrons have no charge and are also in the nucleus. Electrons are negatively charged and orbit the nucleus. The atomic number of an element is the number of its protons, which also determines the number of electrons. The mass number is the total number of protons and neutrons. Isotopes are atoms of the same element that have different numbers of neutrons.