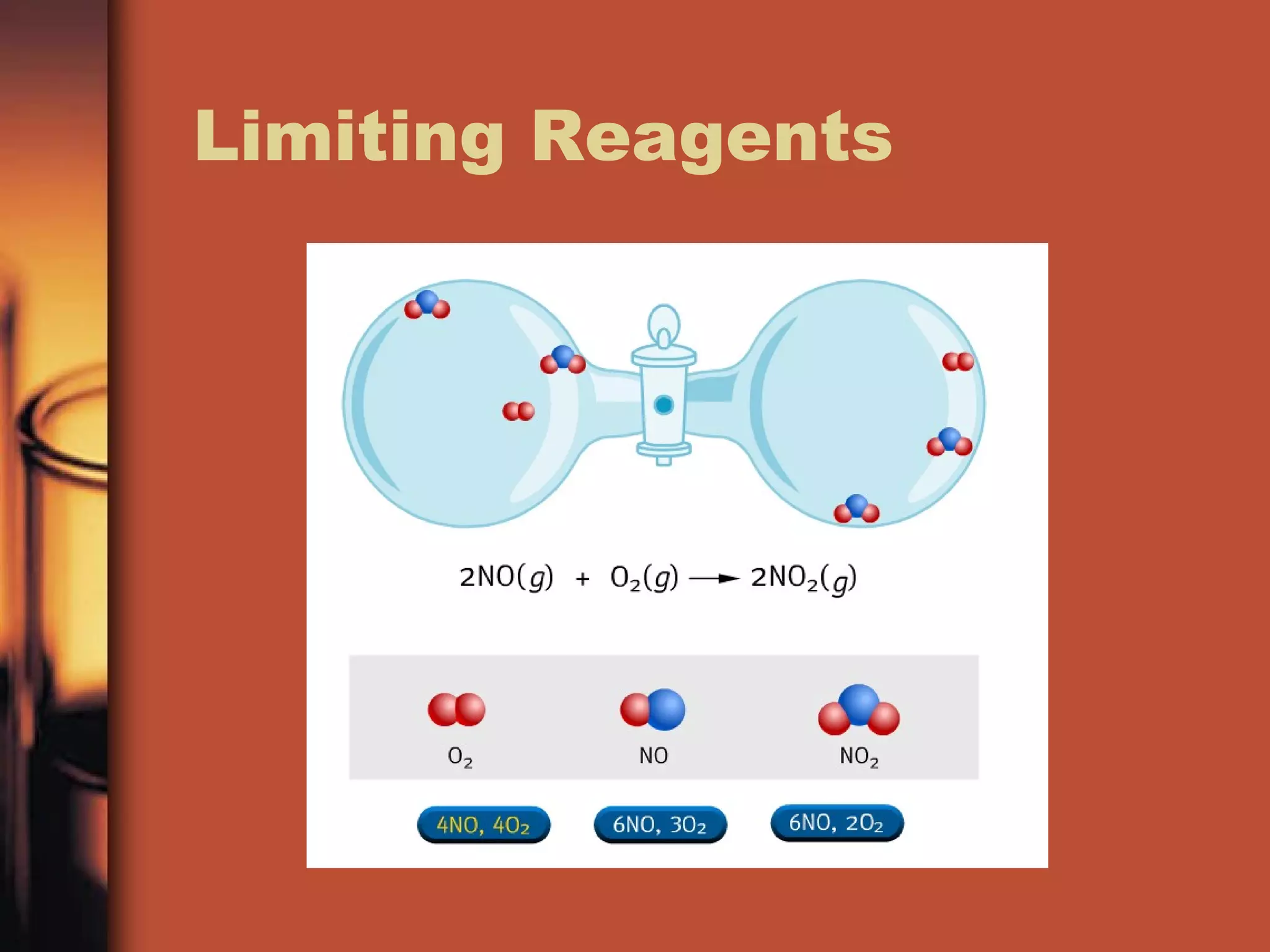
The document discusses chemical reactions and stoichiometry. It defines stoichiometry as using ratios to determine quantities of reactants and products in a chemical reaction. It explains that coefficients in a balanced chemical equation represent molar ratios and can be used to determine moles, mass, and volume of substances in a reaction. It provides examples of solving stoichiometry problems, including determining limiting reactants.