ψ(Wave function) and ψ2
•
0 likes•728 views
Ψ (Psi) represents the state of an electron in the Schrodinger wave equation. Its probability density, Ψ2, gives the probability of finding the electron in a particular region of space. Several wave functions satisfy the Schrodinger equation and properties of the wave function, with each having a corresponding energy level. The wave function with the lowest energy level for a hydrogen atom is ψ1.
Report
Share
Report
Share
Download to read offline
Recommended
Postulates of quantum mechanics 

this slide is introduce the postulates of quantum mechanics in which has all important definable objects is defined. so that presentation is helpful for the undergraduate students
Postulates of quantum mechanics

The document discusses the postulates of quantum mechanics. It describes 6 postulates: 1) the wave function describes the physical state of a system, 2) the wave function and its derivatives must satisfy certain conditions, 3) observable quantities are represented by Hermitian operators, 4) the allowed values of observables are the eigenvalues of the operator equations, 5) the average value of an observable is obtained from an integral relation, and 6) quantum operators are constructed from classical expressions by converting variables to operators. It also provides examples of wave functions and calculating eigenvalues.
Postulates of quantum mechanics & operators

1) Quantum mechanics describes the state of a micro system using a wavefunction that contains all information about the system.
2) Observables like position, momentum, and energy are represented by quantum mechanical operators.
3) The possible values of physical quantities are the eigenvalues obtained by operating the corresponding operator on the wavefunction. The average value of a physical quantity is the expectation value calculated using the wavefunction.
Classical Statistics and Quantum Statistics

This document discusses classical and quantum statistics. It explains that classical statistics, developed by Maxwell, Boltzmann, and Gibbs, were able to explain many macroscopic phenomena but failed to explain others observed at low temperatures. This led to the development of quantum statistics by Bose, Einstein, Fermi and Dirac. Bose-Einstein statistics applies to indistinguishable particles with integer spin like photons that can occupy the same state. Fermi-Dirac statistics applies to particles with half-integer spin like electrons that cannot occupy the same state due to the Pauli exclusion principle. Quantum statistics accounts for the discrete, probabilistic nature of energy at the quantum scale.
Fermi dirac distribution

This document introduces the Fermi-Dirac distribution function. It begins by discussing basic concepts like the Fermi level and Fermi energy. It then covers Fermi and Bose statistics, and the postulates of Fermi particles. The derivation of the Fermi-Dirac distribution function is shown, which gives the probability of a quantum state being occupied at a given energy and temperature. Graphs are presented showing how the distribution varies with different temperatures. The classical limit of the distribution is discussed. References are provided at the end.
One dimensional box

This document discusses the Schrodinger wave equation and its application to modeling a particle in a one-dimensional box. It introduces the history and derivation of the Schrodinger equation, then applies it to solve for the energy and wave functions of a particle confined to a box. The energy is found to be quantized and depend on an integer quantum number. Graphs of the wave functions and probability densities are presented.
Rigit rotar

The rigid rotor model treats a diatomic molecule as two masses connected by a fixed distance that rotate around the center of mass like a spinning dumbbell. This model can be simplified using the concept of reduced mass to treat it as a single rotating body. The rotational energy of the system depends on the moment of inertia, which for a diatomic rigid rotor is equal to the reduced mass times the square of the bond length.
Eigenvalues

Eigenvalues and eigenfunctions are key concepts in linear algebra. An eigenfunction is a function that when operated on by a linear operator produces a constant multiplied version of itself. The constant is the corresponding eigenvalue. Eigenvalues are the solutions to the characteristic polynomial of the linear operator. Eigenfunctions are not unique as any constant multiple of an eigenfunction is also an eigenfunction with the same eigenvalue. The spectrum of an operator is the set of all its eigenvalues.
Recommended
Postulates of quantum mechanics 

this slide is introduce the postulates of quantum mechanics in which has all important definable objects is defined. so that presentation is helpful for the undergraduate students
Postulates of quantum mechanics

The document discusses the postulates of quantum mechanics. It describes 6 postulates: 1) the wave function describes the physical state of a system, 2) the wave function and its derivatives must satisfy certain conditions, 3) observable quantities are represented by Hermitian operators, 4) the allowed values of observables are the eigenvalues of the operator equations, 5) the average value of an observable is obtained from an integral relation, and 6) quantum operators are constructed from classical expressions by converting variables to operators. It also provides examples of wave functions and calculating eigenvalues.
Postulates of quantum mechanics & operators

1) Quantum mechanics describes the state of a micro system using a wavefunction that contains all information about the system.
2) Observables like position, momentum, and energy are represented by quantum mechanical operators.
3) The possible values of physical quantities are the eigenvalues obtained by operating the corresponding operator on the wavefunction. The average value of a physical quantity is the expectation value calculated using the wavefunction.
Classical Statistics and Quantum Statistics

This document discusses classical and quantum statistics. It explains that classical statistics, developed by Maxwell, Boltzmann, and Gibbs, were able to explain many macroscopic phenomena but failed to explain others observed at low temperatures. This led to the development of quantum statistics by Bose, Einstein, Fermi and Dirac. Bose-Einstein statistics applies to indistinguishable particles with integer spin like photons that can occupy the same state. Fermi-Dirac statistics applies to particles with half-integer spin like electrons that cannot occupy the same state due to the Pauli exclusion principle. Quantum statistics accounts for the discrete, probabilistic nature of energy at the quantum scale.
Fermi dirac distribution

This document introduces the Fermi-Dirac distribution function. It begins by discussing basic concepts like the Fermi level and Fermi energy. It then covers Fermi and Bose statistics, and the postulates of Fermi particles. The derivation of the Fermi-Dirac distribution function is shown, which gives the probability of a quantum state being occupied at a given energy and temperature. Graphs are presented showing how the distribution varies with different temperatures. The classical limit of the distribution is discussed. References are provided at the end.
One dimensional box

This document discusses the Schrodinger wave equation and its application to modeling a particle in a one-dimensional box. It introduces the history and derivation of the Schrodinger equation, then applies it to solve for the energy and wave functions of a particle confined to a box. The energy is found to be quantized and depend on an integer quantum number. Graphs of the wave functions and probability densities are presented.
Rigit rotar

The rigid rotor model treats a diatomic molecule as two masses connected by a fixed distance that rotate around the center of mass like a spinning dumbbell. This model can be simplified using the concept of reduced mass to treat it as a single rotating body. The rotational energy of the system depends on the moment of inertia, which for a diatomic rigid rotor is equal to the reduced mass times the square of the bond length.
Eigenvalues

Eigenvalues and eigenfunctions are key concepts in linear algebra. An eigenfunction is a function that when operated on by a linear operator produces a constant multiplied version of itself. The constant is the corresponding eigenvalue. Eigenvalues are the solutions to the characteristic polynomial of the linear operator. Eigenfunctions are not unique as any constant multiple of an eigenfunction is also an eigenfunction with the same eigenvalue. The spectrum of an operator is the set of all its eigenvalues.
Statics presentation ppt(1)

This document provides an introduction to statistical mechanics and different types of statistics. It discusses classical statistics, which includes Maxwell-Boltzmann statistics, and quantum statistics, which includes Bose-Einstein (B-E) and Fermi-Dirac (F-D) statistics. Maxwell-Boltzmann statistics treats particles as distinguishable and applies to ideal gases, while B-E and F-D statistics treat particles as indistinguishable and apply to photons/bosons and electrons/fermions, respectively. The key differences between the statistics are whether particles can occupy the same state (B-E allows multiple occupancy, F-D allows only single occupancy) and the formulas that describe the most probable distribution of particles
statistic mechanics

This document provides an overview of statistical mechanics. It defines microstates and macrostates, and explains that statistical mechanics studies systems with many microstates corresponding to a given macrostate. The Boltzmann distribution is derived, which gives the probability of finding a system in a particular microstate as being proportional to the exponential of the negative of the energy of that microstate divided by the temperature. Maxwell-Boltzmann statistics are described as applying to classical distinguishable particles, yielding the Maxwell-Boltzmann distribution. References for further reading are also included.
Ph 101-9 QUANTUM MACHANICS

The document provides an outline for a course on quantum mechanics. It discusses key topics like the time-dependent Schrodinger equation, eigenvalues and eigenfunctions, boundary conditions for wave functions, and applications like the particle in a box model. Specific solutions to the Schrodinger equation are explored for stationary states with definite energy, including the wave function for a free particle and the quantization of energy for a particle confined to a one-dimensional box.
Basic and fundamental of quantum mechanics (Theory)

Quantum mechanics arose in the early 20th century to explain experimental phenomena that classical mechanics could not, such as black body radiation and the photoelectric effect. The document discusses the origins and fundamental concepts of quantum mechanics, including the dual wave-particle nature of matter and light, the uncertainty principle, and Schrodinger's formulation of quantum mechanics using wave functions and his time-independent equation. It explains that wave functions provide probabilistic information about finding particles in particular regions rather than definite trajectories, replacing Bohr's orbital model.
Particle in a Box problem Quantum Chemistry

1. The particle is confined to a one-dimensional box of length L, with potential energy V=0 inside the box and V=infinity outside.
2. The wave functions and energy levels of the particle are quantized. The wave functions are sinusoidal with n nodes, and the energy is proportional to n^2.
3. The energy levels are spaced further apart at higher n values, with the spacing between levels increasing as the box size decreases.
ψ And ψ2 significance

Ψ represents the wave function in the Schrodinger wave equation and describes the state of an electron. Its square, Ψ2, gives the probability of finding the electron in a particular region of space. Acceptable solutions to the Schrodinger equation must have properties such as continuity, finiteness, and being single valued. The wave function and its square are represented graphically. Several wave functions can satisfy the Schrodinger equation, each with a corresponding energy level. These wave functions are called orbitals.
Quantum mechanics I

CLASSICAL MECHANICS - FAILURES - PHOTOELECTRIC EFFECT- COMPTON EFFECT - DAVISON AND GERMER EXPERIMENT - HEISENBERG UNCERTAINTY PRINCIPLE - QUANTUM MECHANICS POSTULATES - OPERATORS - COOMMUTATORS
Statistical mechanics 

This document discusses statistical mechanics and the distribution of energy among particles in a system. It provides 3 main types of statistical distributions based on the properties of identical particles: Maxwell-Boltzmann, Bose-Einstein, and Fermi-Dirac statistics. Maxwell-Boltzmann statistics applies to distinguishable particles, while Bose-Einstein and Fermi-Dirac apply to indistinguishable particles (bosons and fermions respectively), with the key difference being that fermions obey the Pauli exclusion principle. The document also discusses applications of these distributions, including the Maxwell-Boltzmann distribution law for molecular energies in an ideal gas.
Particle in a box- Application of Schrodinger wave equation

The document summarizes key concepts from quantum chemistry, including:
1) It introduces the historical development of quantum mechanics from classical mechanics and discusses how quantum theory was needed to describe atomic and subatomic phenomena.
2) It then summarizes the particle-like and wave-like properties of light and matter and introduces the Schrodinger equation.
3) The document concludes by presenting the particle-in-a-box model and explaining how solving the Schrodinger equation for this system shows that a particle's energy is quantized into discrete energy levels when confined in a box.
Debye huckle theory

1) Three major factors that increase the conductance of strong electrolytes are the relaxation effect, electrophoretic effect, and frictional resistance of the medium.
2) The relaxation effect occurs when an ion moves and has to rebuild its ionic atmosphere around it, slowing its motion. The electrophoretic effect occurs when ions move against the stream of oppositely charged solvent molecules in their ionic atmosphere.
3) The Debye-Hückel-Onsager equation mathematically relates equivalent conductance to concentration, taking into account these factors. It indicates equivalent conductance decreases with increasing concentration.
Born-Oppenheimer approximation.pptx

1) The Born-Oppenheimer approximation separates the motions of electrons and nuclei in molecules by treating the nuclei as fixed due to their large mass compared to electrons.
2) The Hamiltonian operator for a molecule is divided into electronic and nuclear terms.
3) A Slater determinant describes the wavefunction of a multi-electron system by incorporating the Pauli exclusion principle through its sign change upon exchange of electrons.
Hinshel wood theory

Sir Cyril Hinshelwood and Nikolaevich received the 1956 Nobel Prize in Chemistry for their research on chemical reaction mechanisms. Hinshelwood modified Lindemann's explanation for unimolecular reactions by proposing that energized molecules (A*) may store energy in various molecular bonds and vibrational degrees of freedom, rather than immediately reacting. This statistical distribution of energy among s degrees of freedom leads to a modified rate constant expression containing an additional term of 1/(s-1) that can account for much higher observed reaction rates. However, Hinshelwood's theory does not fully explain some experimental observations such as the temperature dependence of rate constants and nonlinear plots of 1/k1 versus concentration.
SCHRODINGER EQUATION 

The Schrodinger equation plays the role of Newton's laws and conservation of energy in classical mechanics by predicting the future behavior of dynamic systems. It is a wave equation that uses the wavefunction to analytically and precisely predict the probability of events or outcomes, though not the strict determination of a detailed outcome. The kinetic and potential energies are transformed into the Hamiltonian which acts on the wavefunction to generate its evolution in time and space, giving the quantized energies of the system and the form of the wavefunction to calculate other properties.
The heat capacity of a solid 

This document discusses theories of heat capacity in solids. It begins by describing Dulong and Petit's 1819 observation that the heat capacity of solids is approximately 3R. Einstein's theory from 1907 treated solids as assemblies of independent oscillators, predicting the heat capacity approaches 3R only at very high temperatures. Debye's 1912 theory improved on this by treating solids as continuous elastic mediums with phonon vibrational waves, removing the restriction of single oscillator frequencies. The document provides equations for calculating heat capacity based on Einstein's and Debye's models, and notes Debye temperature is important in determining when the heat capacity approaches the classical Dulong-Petit limit.
Mo theory

This document provides an overview of molecular orbital theory. It explains that molecular orbital theory describes molecules in terms of orbitals and electron configurations similar to atomic orbital theory. The key points are:
- Molecular orbitals are formed from the overlapping of atomic orbitals on different atoms.
- Bonding orbitals are formed from constructive interference and lower the energy, while antibonding orbitals are formed from destructive interference and increase the energy.
- Homonuclear diatomic molecules like H2, O2, and N2 are discussed as examples, with their molecular orbitals, bond orders, and magnetic properties explained.
47rrkmtheory11 190428142321

This document summarizes Rice-Ramsperger-Kassel-Marcus (RRKM) theory, which describes the rates of unimolecular reactions. RRKM theory originated from Lindemann-Christiansen theory and considers how energy is distributed among vibrational modes. During the 1950s-1952, R.A. Marcus merged transition state theory with RRK theory. RRKM theory accounts for individual vibrational frequencies and classifies energy as either active or inactive. It can explain high pre-exponential factors and has been used to study reactions like isomerization, though limitations include uncertainty in activated complex vibrational frequencies.
Simple harmonic oscillator

The document discusses the quantum mechanical treatment of the simple harmonic oscillator. It shows that the Schrodinger equation for a simple harmonic oscillator can be solved to obtain the Hermite equation. The solutions to the Hermite equation are the Hermite polynomials, which give the normalized wave functions and allowed energy levels of the simple harmonic oscillator. The energy levels are found to be quantized with energies equal to integer multiples of one half the angular frequency of oscillation.
Rotational spectroscopy

Rotational Spectroscopy is also known as microwave Spectroscopy and is used for bond length calculations
Fundamentals of modern physics, the de-Broglie hypothesis

The presentation uploaded here educates about the failure of classical physics to explain Blackbody radiation and the success of quantum theory to explain the Blackbody radiation spectrum and other phenomena, the de-Broglie hypothesis and its significance, nature of de-broglie waves and the representation. Numerical problems are given at the end.
Goan Shimgo sahitya (some of the folk songs)

This is a small attempt to preserve heritage and folk culture of Goa
How long could one survive in a perfectly airtight room.docx

How long could one survive in a perfectly airtight room?
Given
Inhaled air contains 21% oxygen while exhaled breath contains approximately 16% oxygen and 5% carbon dioxide
The minimum oxygen level required for survival is 19 %.
Dimensions of room in meters: 5 X 5 X 3
More Related Content
What's hot
Statics presentation ppt(1)

This document provides an introduction to statistical mechanics and different types of statistics. It discusses classical statistics, which includes Maxwell-Boltzmann statistics, and quantum statistics, which includes Bose-Einstein (B-E) and Fermi-Dirac (F-D) statistics. Maxwell-Boltzmann statistics treats particles as distinguishable and applies to ideal gases, while B-E and F-D statistics treat particles as indistinguishable and apply to photons/bosons and electrons/fermions, respectively. The key differences between the statistics are whether particles can occupy the same state (B-E allows multiple occupancy, F-D allows only single occupancy) and the formulas that describe the most probable distribution of particles
statistic mechanics

This document provides an overview of statistical mechanics. It defines microstates and macrostates, and explains that statistical mechanics studies systems with many microstates corresponding to a given macrostate. The Boltzmann distribution is derived, which gives the probability of finding a system in a particular microstate as being proportional to the exponential of the negative of the energy of that microstate divided by the temperature. Maxwell-Boltzmann statistics are described as applying to classical distinguishable particles, yielding the Maxwell-Boltzmann distribution. References for further reading are also included.
Ph 101-9 QUANTUM MACHANICS

The document provides an outline for a course on quantum mechanics. It discusses key topics like the time-dependent Schrodinger equation, eigenvalues and eigenfunctions, boundary conditions for wave functions, and applications like the particle in a box model. Specific solutions to the Schrodinger equation are explored for stationary states with definite energy, including the wave function for a free particle and the quantization of energy for a particle confined to a one-dimensional box.
Basic and fundamental of quantum mechanics (Theory)

Quantum mechanics arose in the early 20th century to explain experimental phenomena that classical mechanics could not, such as black body radiation and the photoelectric effect. The document discusses the origins and fundamental concepts of quantum mechanics, including the dual wave-particle nature of matter and light, the uncertainty principle, and Schrodinger's formulation of quantum mechanics using wave functions and his time-independent equation. It explains that wave functions provide probabilistic information about finding particles in particular regions rather than definite trajectories, replacing Bohr's orbital model.
Particle in a Box problem Quantum Chemistry

1. The particle is confined to a one-dimensional box of length L, with potential energy V=0 inside the box and V=infinity outside.
2. The wave functions and energy levels of the particle are quantized. The wave functions are sinusoidal with n nodes, and the energy is proportional to n^2.
3. The energy levels are spaced further apart at higher n values, with the spacing between levels increasing as the box size decreases.
ψ And ψ2 significance

Ψ represents the wave function in the Schrodinger wave equation and describes the state of an electron. Its square, Ψ2, gives the probability of finding the electron in a particular region of space. Acceptable solutions to the Schrodinger equation must have properties such as continuity, finiteness, and being single valued. The wave function and its square are represented graphically. Several wave functions can satisfy the Schrodinger equation, each with a corresponding energy level. These wave functions are called orbitals.
Quantum mechanics I

CLASSICAL MECHANICS - FAILURES - PHOTOELECTRIC EFFECT- COMPTON EFFECT - DAVISON AND GERMER EXPERIMENT - HEISENBERG UNCERTAINTY PRINCIPLE - QUANTUM MECHANICS POSTULATES - OPERATORS - COOMMUTATORS
Statistical mechanics 

This document discusses statistical mechanics and the distribution of energy among particles in a system. It provides 3 main types of statistical distributions based on the properties of identical particles: Maxwell-Boltzmann, Bose-Einstein, and Fermi-Dirac statistics. Maxwell-Boltzmann statistics applies to distinguishable particles, while Bose-Einstein and Fermi-Dirac apply to indistinguishable particles (bosons and fermions respectively), with the key difference being that fermions obey the Pauli exclusion principle. The document also discusses applications of these distributions, including the Maxwell-Boltzmann distribution law for molecular energies in an ideal gas.
Particle in a box- Application of Schrodinger wave equation

The document summarizes key concepts from quantum chemistry, including:
1) It introduces the historical development of quantum mechanics from classical mechanics and discusses how quantum theory was needed to describe atomic and subatomic phenomena.
2) It then summarizes the particle-like and wave-like properties of light and matter and introduces the Schrodinger equation.
3) The document concludes by presenting the particle-in-a-box model and explaining how solving the Schrodinger equation for this system shows that a particle's energy is quantized into discrete energy levels when confined in a box.
Debye huckle theory

1) Three major factors that increase the conductance of strong electrolytes are the relaxation effect, electrophoretic effect, and frictional resistance of the medium.
2) The relaxation effect occurs when an ion moves and has to rebuild its ionic atmosphere around it, slowing its motion. The electrophoretic effect occurs when ions move against the stream of oppositely charged solvent molecules in their ionic atmosphere.
3) The Debye-Hückel-Onsager equation mathematically relates equivalent conductance to concentration, taking into account these factors. It indicates equivalent conductance decreases with increasing concentration.
Born-Oppenheimer approximation.pptx

1) The Born-Oppenheimer approximation separates the motions of electrons and nuclei in molecules by treating the nuclei as fixed due to their large mass compared to electrons.
2) The Hamiltonian operator for a molecule is divided into electronic and nuclear terms.
3) A Slater determinant describes the wavefunction of a multi-electron system by incorporating the Pauli exclusion principle through its sign change upon exchange of electrons.
Hinshel wood theory

Sir Cyril Hinshelwood and Nikolaevich received the 1956 Nobel Prize in Chemistry for their research on chemical reaction mechanisms. Hinshelwood modified Lindemann's explanation for unimolecular reactions by proposing that energized molecules (A*) may store energy in various molecular bonds and vibrational degrees of freedom, rather than immediately reacting. This statistical distribution of energy among s degrees of freedom leads to a modified rate constant expression containing an additional term of 1/(s-1) that can account for much higher observed reaction rates. However, Hinshelwood's theory does not fully explain some experimental observations such as the temperature dependence of rate constants and nonlinear plots of 1/k1 versus concentration.
SCHRODINGER EQUATION 

The Schrodinger equation plays the role of Newton's laws and conservation of energy in classical mechanics by predicting the future behavior of dynamic systems. It is a wave equation that uses the wavefunction to analytically and precisely predict the probability of events or outcomes, though not the strict determination of a detailed outcome. The kinetic and potential energies are transformed into the Hamiltonian which acts on the wavefunction to generate its evolution in time and space, giving the quantized energies of the system and the form of the wavefunction to calculate other properties.
The heat capacity of a solid 

This document discusses theories of heat capacity in solids. It begins by describing Dulong and Petit's 1819 observation that the heat capacity of solids is approximately 3R. Einstein's theory from 1907 treated solids as assemblies of independent oscillators, predicting the heat capacity approaches 3R only at very high temperatures. Debye's 1912 theory improved on this by treating solids as continuous elastic mediums with phonon vibrational waves, removing the restriction of single oscillator frequencies. The document provides equations for calculating heat capacity based on Einstein's and Debye's models, and notes Debye temperature is important in determining when the heat capacity approaches the classical Dulong-Petit limit.
Mo theory

This document provides an overview of molecular orbital theory. It explains that molecular orbital theory describes molecules in terms of orbitals and electron configurations similar to atomic orbital theory. The key points are:
- Molecular orbitals are formed from the overlapping of atomic orbitals on different atoms.
- Bonding orbitals are formed from constructive interference and lower the energy, while antibonding orbitals are formed from destructive interference and increase the energy.
- Homonuclear diatomic molecules like H2, O2, and N2 are discussed as examples, with their molecular orbitals, bond orders, and magnetic properties explained.
47rrkmtheory11 190428142321

This document summarizes Rice-Ramsperger-Kassel-Marcus (RRKM) theory, which describes the rates of unimolecular reactions. RRKM theory originated from Lindemann-Christiansen theory and considers how energy is distributed among vibrational modes. During the 1950s-1952, R.A. Marcus merged transition state theory with RRK theory. RRKM theory accounts for individual vibrational frequencies and classifies energy as either active or inactive. It can explain high pre-exponential factors and has been used to study reactions like isomerization, though limitations include uncertainty in activated complex vibrational frequencies.
Simple harmonic oscillator

The document discusses the quantum mechanical treatment of the simple harmonic oscillator. It shows that the Schrodinger equation for a simple harmonic oscillator can be solved to obtain the Hermite equation. The solutions to the Hermite equation are the Hermite polynomials, which give the normalized wave functions and allowed energy levels of the simple harmonic oscillator. The energy levels are found to be quantized with energies equal to integer multiples of one half the angular frequency of oscillation.
Rotational spectroscopy

Rotational Spectroscopy is also known as microwave Spectroscopy and is used for bond length calculations
Fundamentals of modern physics, the de-Broglie hypothesis

The presentation uploaded here educates about the failure of classical physics to explain Blackbody radiation and the success of quantum theory to explain the Blackbody radiation spectrum and other phenomena, the de-Broglie hypothesis and its significance, nature of de-broglie waves and the representation. Numerical problems are given at the end.
What's hot (20)
Basic and fundamental of quantum mechanics (Theory)

Basic and fundamental of quantum mechanics (Theory)
Particle in a box- Application of Schrodinger wave equation

Particle in a box- Application of Schrodinger wave equation
Fundamentals of modern physics, the de-Broglie hypothesis

Fundamentals of modern physics, the de-Broglie hypothesis
More from Mithil Fal Desai
Goan Shimgo sahitya (some of the folk songs)

This is a small attempt to preserve heritage and folk culture of Goa
How long could one survive in a perfectly airtight room.docx

How long could one survive in a perfectly airtight room?
Given
Inhaled air contains 21% oxygen while exhaled breath contains approximately 16% oxygen and 5% carbon dioxide
The minimum oxygen level required for survival is 19 %.
Dimensions of room in meters: 5 X 5 X 3
The role of Model systems.pdf

Model systems are smaller, simpler versions of complex biomolecules that can be used to study biological chemical processes. While not a perfect representation, model systems allow for controlled experiments that provide valuable information about real biological systems. Transition metal complexes with organic ligands are often used as model systems to mimic biological chemical reactions involving metal ions. Examples of model systems that have increased understanding of real systems include iron-porphyrin compounds for hemoglobin and copper complexes for copper-containing enzymes.
Writing Third Year Project Report: Some Handy Guidelines

The document provides guidelines for writing a third year project report. It discusses the different types of research projects including fundamental, descriptive, empirical, quantitative, applied, analytical and qualitative. It outlines the typical sections of a project report such as the title page, declaration, certificate, acknowledgment, index, abstract, introduction, experimental, results and discussion, conclusion and references. For each section, it provides details on the content and formatting. It emphasizes writing in a clear, concise and organized manner with proper citations and references. The document aims to help students structure their project reports and effectively communicate their research.
Chemical properties of Interhalogen compounds.pdf

Interhalogen compounds exhibit various chemical properties and reactivities. AX-type interhalogens like ICl hydrolyze to form hydrogen halides and hypohalous acids. ClF3 and BrF3 can fluorinate metal oxides to form metal fluorides and oxygen. Higher interhalogens like ClF5 and IF7 can ionize to form polyatomic ions. IF7 reacts with water to form IOF5 and HF, and with silicon dioxide to form IOF5 and silicon tetrafluoride.
Determination of conductivity of soil sample.pdf

This document provides instructions for determining the conductivity of a soil sample. It explains that soil conductivity is a measure of its ability to carry an electrical current and can indicate nutrient levels and contamination. The procedure calibrates a conductivity cell using potassium chloride solutions, measures the conductance of distilled water, mixes a soil sample with water, filters it, and measures the conductance to determine the soil sample's conductivity.
Determination of Bulk density of soil sample..pdf

This document provides instructions for determining the bulk density of a soil sample using the cylinder method. It defines bulk density as the dry weight of soil divided by its volume, which includes both soil particles and pore space. The procedure involves heating a soil sample to remove moisture, placing it in a measuring cylinder, and recording the weight and occupied volume without compaction. Bulk density can be expressed in units of g/mL or g/cm3 and provides information about soil properties like porosity, root growth, and nutrient availability.
Determination of acidity of a water sample..pdf

The acidity of water is a measure of the quantitative capacity to react with a mineral base. The measured value usually varies with the pH indicator used in the experiment. The solution of phenolphthalein is colourless in acidic or mild basic medium (pH<8.0), while in basic medium (pH<9.6) solution is pink. The solution of methyl orange is generally red at pH lower than 3.1 and above pH 4.4 the solutions are yellow.
The dissolved strong mineral acids, weak organic acids, dissolved carbon dioxide and hydrolyzed mineral salts may contribute to the measured acidity of natural water. The acidity of water is a significant parameter of water as the presence of acids in natural water contributes to corrosiveness and influences chemical reaction rates. The biological processes are dependent on the acidity of water. The quantitative acidity measurement also reflects the quality of the source of water. The acidity of water is usually expressed in terms of CaCO3.
Techniques of soil sampling.pdf

Soil sampling is important to determine the quality and composition of soil. A representative soil sample should include incremental samples taken from the surface and at depths of 0.5 and 1.0 meters. Individual samples should be labeled and stored properly. As soil can appear homogeneous but actually vary in composition, careful sampling procedures are necessary to obtain samples truly representative of the soil being analyzed. Where soil is heterogeneous over a large area, sampling must be done with great care.
Inorganic Section solved FYBSC 2022.pdf

The document is an answer key for an inorganic chemistry and organic chemistry exam containing multiple choice and descriptive questions.
Some of the questions addressed in the summary include drawing orbital diagrams, electronic configurations of atoms, types of chemical bonds, and properties of ionic compounds.
The descriptive questions require explanations about anomalous electronic configurations, molecular geometry, decomposition of carbonates, quantum numbers, and dual nature of matter.
IDP MFDdraft.pptx

The document provides guidelines for developing an Institutional Development Plan (IDP). It outlines 7 key sections:
1) General instructions for developing the IDP including defining objectives, goals, and activity plans.
2) The IDP will be prepared for 5 years and include sustainability measures. It will be a living document that evolves over time.
3) Key steps in the IDP development process including stakeholder consultations and drafting/finalizing the plan.
4) IDP implementation grants will be awarded competitively and approved plans will be published online.
5) Institutions will report progress against targets to the Department of Higher Education periodically.
Determine the strength in grams per litre of a given AgNO3 solution being pro...

1) The document describes the Mohr's method for determining the concentration of silver nitrate (AgNO3) solution by titrating it against a standard sodium chloride (NaCl) solution.
2) In the titration, silver ions from AgNO3 react quantitatively with chloride ions from NaCl to form a precipitate of silver chloride. The endpoint is reached when excess Ag+ ions react with chromate ions from a potassium chromate indicator to form a red-brown precipitate of silver chromate.
3) The procedure involves titrating a known concentration of NaCl solution against the AgNO3 solution to determine its molarity, using potassium chromate as the indicator.
अडण्णे शिगमो लोकसाहित्य

This is a small attempt to preserve heritage and folk culture of Goan village Adnem
Credits to Ghanashyam K. Devidas, Lakshimikant Bhavu, Vithobha Bhavu
Molecular orbital diagram of CO and NO

This document contains molecular orbital diagrams for carbon monoxide (CO) and nitric oxide (NO). The CO diagram shows the atomic orbitals of carbon and oxygen and the resulting molecular orbitals of 4σ, 2σ, 1σ, 3σ, and 2Π, 1Π. The NO diagram also shows the atomic orbitals of nitrogen and oxygen and their molecular orbitals of 2σ, 1σ, 3σ, 4σ, 2Π, and 1Π. Both diagrams provide the electron configurations of carbon, oxygen, nitrogen and illustrate the molecular orbital energy levels formed when they combine.
Estimation of water of crystallization in mohr’s salt by titrating with stand...

The document describes an experiment to determine the water of crystallization in Mohr's salt, (NH4)2Fe(SO4)2.XH2O, by titrating a solution of the salt with standardized potassium manganate(VII). The experiment involves standardizing the KMnO4 solution using sodium oxalate, then titrating samples of Mohr's salt solution and calculating the iron content. Based on comparing the experimental iron percentage to theoretical percentages for different values of X, the water of crystallization is determined to be 6 moles, giving the formula (NH4)2Fe(SO4)2.6H2O.
Estimation of oxalic acid experimental data

The document describes the titration of oxalic acid with potassium permanganate. It provides the theory of the redox reaction, where manganese is reduced and carbon is oxidized. It then gives the experimental procedure for standardizing the KMnO4 solution using sodium oxalate and then using the standardized solution to determine the concentration of an unknown sample of oxalic acid. The calculations section shows how to determine the equivalent weight of oxalic acid and use the titration data to find the exact normality of KMnO4 and the normality and mass of oxalic acid in the unknown sample.
Polarisation and polarisability

Polarisation occurs when the electron cloud of an atom or ion is distorted by the influence of a nearby positive atomic nucleus or electric field. Polarisability is the ability of an atom or ion to become polarized. Polarization can range from a slight distortion to a sufficient distortion that forms a covalent bond between ions. According to Fajans' Rules, ions with higher charges and smaller sizes polarize anions more, while ions with higher charges and larger sizes polarize cations more. Ions with partially filled d orbitals also polarize more than ions with noble gas configurations.
Molecular orbitals diagrams of [Co(NH3)6]3+ ![Molecular orbitals diagrams of [Co(NH3)6]3+](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Molecular orbitals diagrams of [Co(NH3)6]3+](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
The document presents an M.O. diagram for the [Co(NH3)6]3+ complex. It shows the metal (Co3+) orbitals interacting with the ligand (NH3) orbitals to form molecular orbitals. The Co3+ has a 3d6, 4s0 electronic configuration. The six ammonia ligands each contribute 2 sigma electrons, for a total of 12 ligand electrons. These interact with the metal orbitals to form the molecular orbital diagram shown, with no pi interactions included.
Evidences for covalent bonding in complexes

This document discusses evidence for covalent bonding in metal complexes from three perspectives:
1. The nephelauxetic effect shows that electron-electron repulsion is less in complexes than free metal ions due to delocalization of electrons over ligand orbitals.
2. Nephelauxetic parameters (β) quantify this effect, with softer ligands having smaller β values.
3. Electron paramagnetic resonance (EPR) spectroscopy reveals hyperfine splitting in complex spectra, showing interaction between ligand nuclear spins and metal electron spins, further indicating covalent bonding.
Molecular orbitals diagrams of [Ti(H2O)6]3+ ![Molecular orbitals diagrams of [Ti(H2O)6]3+](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Molecular orbitals diagrams of [Ti(H2O)6]3+](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
The document presents an M.O. diagram for the [Ti(H2O)6]3+ complex. It shows the metal (Ti3+) orbitals and ligand (H2O) orbitals that form the molecular orbitals of the complex. The diagram displays the orbital interactions between the titanium 3d1, 4s0 orbitals and the 12 sigma electrons from the six water ligands to produce the molecular orbital configuration of the [Ti(H2O)6]3+ complex.
More from Mithil Fal Desai (20)
How long could one survive in a perfectly airtight room.docx

How long could one survive in a perfectly airtight room.docx
Writing Third Year Project Report: Some Handy Guidelines

Writing Third Year Project Report: Some Handy Guidelines
Determine the strength in grams per litre of a given AgNO3 solution being pro...

Determine the strength in grams per litre of a given AgNO3 solution being pro...
Estimation of water of crystallization in mohr’s salt by titrating with stand...

Estimation of water of crystallization in mohr’s salt by titrating with stand...
Recently uploaded
A Visual Guide to 1 Samuel | A Tale of Two Hearts

These slides walk through the story of 1 Samuel. Samuel is the last judge of Israel. The people reject God and want a king. Saul is anointed as the first king, but he is not a good king. David, the shepherd boy is anointed and Saul is envious of him. David shows honor while Saul continues to self destruct.
Level 3 NCEA - NZ: A Nation In the Making 1872 - 1900 SML.ppt

The History of NZ 1870-1900.
Making of a Nation.
From the NZ Wars to Liberals,
Richard Seddon, George Grey,
Social Laboratory, New Zealand,
Confiscations, Kotahitanga, Kingitanga, Parliament, Suffrage, Repudiation, Economic Change, Agriculture, Gold Mining, Timber, Flax, Sheep, Dairying,
HYPERTENSION - SLIDE SHARE PRESENTATION.

IT WILL BE HELPFULL FOR THE NUSING STUDENTS
IT FOCUSED ON MEDICAL MANAGEMENT AND NURSING MANAGEMENT.
HIGHLIGHTS ON HEALTH EDUCATION.
How to Setup Default Value for a Field in Odoo 17

In Odoo, we can set a default value for a field during the creation of a record for a model. We have many methods in odoo for setting a default value to the field.
skeleton System.pdf (skeleton system wow)

🔥🔥🔥🔥🔥🔥🔥🔥🔥
إضغ بين إيديكم من أقوى الملازم التي صممتها
ملزمة تشريح الجهاز الهيكلي (نظري 3)
💀💀💀💀💀💀💀💀💀💀
تتميز هذهِ الملزمة بعِدة مُميزات :
1- مُترجمة ترجمة تُناسب جميع المستويات
2- تحتوي على 78 رسم توضيحي لكل كلمة موجودة بالملزمة (لكل كلمة !!!!)
#فهم_ماكو_درخ
3- دقة الكتابة والصور عالية جداً جداً جداً
4- هُنالك بعض المعلومات تم توضيحها بشكل تفصيلي جداً (تُعتبر لدى الطالب أو الطالبة بإنها معلومات مُبهمة ومع ذلك تم توضيح هذهِ المعلومات المُبهمة بشكل تفصيلي جداً
5- الملزمة تشرح نفسها ب نفسها بس تكلك تعال اقراني
6- تحتوي الملزمة في اول سلايد على خارطة تتضمن جميع تفرُعات معلومات الجهاز الهيكلي المذكورة في هذهِ الملزمة
واخيراً هذهِ الملزمة حلالٌ عليكم وإتمنى منكم إن تدعولي بالخير والصحة والعافية فقط
كل التوفيق زملائي وزميلاتي ، زميلكم محمد الذهبي 💊💊
🔥🔥🔥🔥🔥🔥🔥🔥🔥
Educational Technology in the Health Sciences

Plenary presentation at the NTTC Inter-university Workshop, 18 June 2024, Manila Prince Hotel.
THE SACRIFICE HOW PRO-PALESTINE PROTESTS STUDENTS ARE SACRIFICING TO CHANGE T...

The recent surge in pro-Palestine student activism has prompted significant responses from universities, ranging from negotiations and divestment commitments to increased transparency about investments in companies supporting the war on Gaza. This activism has led to the cessation of student encampments but also highlighted the substantial sacrifices made by students, including academic disruptions and personal risks. The primary drivers of these protests are poor university administration, lack of transparency, and inadequate communication between officials and students. This study examines the profound emotional, psychological, and professional impacts on students engaged in pro-Palestine protests, focusing on Generation Z's (Gen-Z) activism dynamics. This paper explores the significant sacrifices made by these students and even the professors supporting the pro-Palestine movement, with a focus on recent global movements. Through an in-depth analysis of printed and electronic media, the study examines the impacts of these sacrifices on the academic and personal lives of those involved. The paper highlights examples from various universities, demonstrating student activism's long-term and short-term effects, including disciplinary actions, social backlash, and career implications. The researchers also explore the broader implications of student sacrifices. The findings reveal that these sacrifices are driven by a profound commitment to justice and human rights, and are influenced by the increasing availability of information, peer interactions, and personal convictions. The study also discusses the broader implications of this activism, comparing it to historical precedents and assessing its potential to influence policy and public opinion. The emotional and psychological toll on student activists is significant, but their sense of purpose and community support mitigates some of these challenges. However, the researchers call for acknowledging the broader Impact of these sacrifices on the future global movement of FreePalestine.
Andreas Schleicher presents PISA 2022 Volume III - Creative Thinking - 18 Jun...

Andreas Schleicher, Director of Education and Skills at the OECD presents at the launch of PISA 2022 Volume III - Creative Minds, Creative Schools on 18 June 2024.
CapTechTalks Webinar Slides June 2024 Donovan Wright.pptx

Slides from a Capitol Technology University webinar held June 20, 2024. The webinar featured Dr. Donovan Wright, presenting on the Department of Defense Digital Transformation.
Elevate Your Nonprofit's Online Presence_ A Guide to Effective SEO Strategies...

Whether you're new to SEO or looking to refine your existing strategies, this webinar will provide you with actionable insights and practical tips to elevate your nonprofit's online presence.
Information and Communication Technology in Education

(𝐓𝐋𝐄 𝟏𝟎𝟎) (𝐋𝐞𝐬𝐬𝐨𝐧 2)-𝐏𝐫𝐞𝐥𝐢𝐦𝐬
𝐄𝐱𝐩𝐥𝐚𝐢𝐧 𝐭𝐡𝐞 𝐈𝐂𝐓 𝐢𝐧 𝐞𝐝𝐮𝐜𝐚𝐭𝐢𝐨𝐧:
Students will be able to explain the role and impact of Information and Communication Technology (ICT) in education. They will understand how ICT tools, such as computers, the internet, and educational software, enhance learning and teaching processes. By exploring various ICT applications, students will recognize how these technologies facilitate access to information, improve communication, support collaboration, and enable personalized learning experiences.
𝐃𝐢𝐬𝐜𝐮𝐬𝐬 𝐭𝐡𝐞 𝐫𝐞𝐥𝐢𝐚𝐛𝐥𝐞 𝐬𝐨𝐮𝐫𝐜𝐞𝐬 𝐨𝐧 𝐭𝐡𝐞 𝐢𝐧𝐭𝐞𝐫𝐧𝐞𝐭:
-Students will be able to discuss what constitutes reliable sources on the internet. They will learn to identify key characteristics of trustworthy information, such as credibility, accuracy, and authority. By examining different types of online sources, students will develop skills to evaluate the reliability of websites and content, ensuring they can distinguish between reputable information and misinformation.
BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 8 - CẢ NĂM - FRIENDS PLUS - NĂM HỌC 2023-2024 (B...

BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 8 - CẢ NĂM - FRIENDS PLUS - NĂM HỌC 2023-2024 (B...Nguyen Thanh Tu Collection
https://app.box.com/s/nrwz52lilmrw6m5kqeqn83q6vbdp8yzpINTRODUCTION TO HOSPITALS & AND ITS ORGANIZATION 

The document discuss about the hospitals and it's organization .
Recently uploaded (20)
Level 3 NCEA - NZ: A Nation In the Making 1872 - 1900 SML.ppt

Level 3 NCEA - NZ: A Nation In the Making 1872 - 1900 SML.ppt
THE SACRIFICE HOW PRO-PALESTINE PROTESTS STUDENTS ARE SACRIFICING TO CHANGE T...

THE SACRIFICE HOW PRO-PALESTINE PROTESTS STUDENTS ARE SACRIFICING TO CHANGE T...
Andreas Schleicher presents PISA 2022 Volume III - Creative Thinking - 18 Jun...

Andreas Schleicher presents PISA 2022 Volume III - Creative Thinking - 18 Jun...
CapTechTalks Webinar Slides June 2024 Donovan Wright.pptx

CapTechTalks Webinar Slides June 2024 Donovan Wright.pptx
Elevate Your Nonprofit's Online Presence_ A Guide to Effective SEO Strategies...

Elevate Your Nonprofit's Online Presence_ A Guide to Effective SEO Strategies...
Information and Communication Technology in Education

Information and Communication Technology in Education
BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 8 - CẢ NĂM - FRIENDS PLUS - NĂM HỌC 2023-2024 (B...

BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 8 - CẢ NĂM - FRIENDS PLUS - NĂM HỌC 2023-2024 (B...
ψ(Wave function) and ψ2
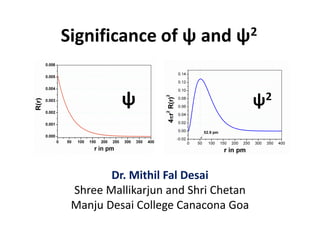
- 1. Significance of ψ and ψ2 Dr. Mithil Fal Desai Shree Mallikarjun and Shri Chetan Manju Desai College Canacona Goa ψ ψ2
- 2. Ψ and Ψ2 Ψ (Psi) is a wave function represented in the Schrodinger wave equation, which represents the state of the electron. Its corresponding probability density (Ψ2) gives the probability of finding the electron in a particular region in space.
- 4. Properties of wave function (ψ)
- 5. End note Several wave function satisfy these properties and each of these have corresponding energies. These wave functions are called orbitals. In hydrogen atom the wave function which has lowest energy level(E1) is 𝝍1. There are number of acceptable solutions of the wave function and each orbital is described by set of three quantum numbers n, l and m.