ψ And ψ2 significance
•Download as PPTX, PDF•
0 likes•3,344 views
Ψ represents the wave function in the Schrodinger wave equation and describes the state of an electron. Its square, Ψ2, gives the probability of finding the electron in a particular region of space. Acceptable solutions to the Schrodinger equation must have properties such as continuity, finiteness, and being single valued. The wave function and its square are represented graphically. Several wave functions can satisfy the Schrodinger equation, each with a corresponding energy level. These wave functions are called orbitals.
Report
Share
Report
Share
Recommended
Electrochemistry

Types of electrodes, Applications of EMF, Determination of pH, Faradays law of electrolysis, Electrolysis of sodium chloride, Ionic strength
Coordination chemistry-2

Theories of coordination compounds, CFSE, Bonding in octahedral and tetrahedral complex, color of transition metal complex, magnetic properties, selection rules, Nephelxeuatic effect, angular overlap model
Recommended
Electrochemistry

Types of electrodes, Applications of EMF, Determination of pH, Faradays law of electrolysis, Electrolysis of sodium chloride, Ionic strength
Coordination chemistry-2

Theories of coordination compounds, CFSE, Bonding in octahedral and tetrahedral complex, color of transition metal complex, magnetic properties, selection rules, Nephelxeuatic effect, angular overlap model
Determination of equivalence conductance, degree of dissociation and dissocia...

liberated iodine? is it soluble in water?
Factors affecting the magnitude of 10 Dq

The presentation discusses briefly the factors affecting the magnitude of 10 Dq and also the term Dq for undergraduates.
https://youtu.be/2lFoaP9N_5Y
Postulates of quantum mechanics

Postulates of quantum mechanics, useful for BSc and MSc chemistry students.
Physical chemistry presentation

This presentation consists of three topics that are:
1. conductance of electrolytic solution
2. Specific Conductance, Molar Conductance & Equivalent Conductance
3. Kohlrausch's Law
Electrochemistry

CONDUCTIVITY-TYPES-VARIATION WITH DILUTION-KOHLRAUSCH LAW - TRANSFERENCE NUMBER -DETERMINATION - IONIC MOBILITY - APPLICATION OF CONDUCTANCE MEASUREMENTS - CONDUCTOMENTRIC TITRATION
Postulates of quantum mechanics 

this slide is introduce the postulates of quantum mechanics in which has all important definable objects is defined. so that presentation is helpful for the undergraduate students
Lect. 23 rotational vibrational raman spectroscopy

rotational Raman spectrum vibrational Raman spectrum,
Schrodinger equation and its applications: Chapter 2

Wave function and its physical significance, Schrodinger time dependent equation, Separation in time dependent and time independent parts, Operators in quantum Mechanics, Eigen functions and Eigen values, Particle in one dimensional and three dimensional box (Energy eigen values). Qualitative analysis of potential barrier Tunneling effect). Simple Harmonic Oscillator (Qualitative analysis of Zero point energy)
Mot

It is about molecular orbital theory specially mo diagram of diatomic atoms,their bond orders,bond lengths and stability and experimental evidences of ionisation energy from PES.
Stochastic Schrödinger equations

We use stochastic methods to present mathematically correct representation of the wave function. Informal construction was developed by R. Feynman. This approach were introduced first by H. Doss Sur une Resolution Stochastique de l'Equation de Schrödinger à Coefficients Analytiques. Communications in Mathematical Physics
October 1980, Volume 73, Issue 3, pp 247–264.
Primary intention is to discuss formal stochastic representation of the Schrodinger equation solution with its applications to the theory of demolition quantum measurements.
I will appreciate your comments.
More Related Content
What's hot
Determination of equivalence conductance, degree of dissociation and dissocia...

liberated iodine? is it soluble in water?
Factors affecting the magnitude of 10 Dq

The presentation discusses briefly the factors affecting the magnitude of 10 Dq and also the term Dq for undergraduates.
https://youtu.be/2lFoaP9N_5Y
Postulates of quantum mechanics

Postulates of quantum mechanics, useful for BSc and MSc chemistry students.
Physical chemistry presentation

This presentation consists of three topics that are:
1. conductance of electrolytic solution
2. Specific Conductance, Molar Conductance & Equivalent Conductance
3. Kohlrausch's Law
Electrochemistry

CONDUCTIVITY-TYPES-VARIATION WITH DILUTION-KOHLRAUSCH LAW - TRANSFERENCE NUMBER -DETERMINATION - IONIC MOBILITY - APPLICATION OF CONDUCTANCE MEASUREMENTS - CONDUCTOMENTRIC TITRATION
Postulates of quantum mechanics 

this slide is introduce the postulates of quantum mechanics in which has all important definable objects is defined. so that presentation is helpful for the undergraduate students
Lect. 23 rotational vibrational raman spectroscopy

rotational Raman spectrum vibrational Raman spectrum,
Schrodinger equation and its applications: Chapter 2

Wave function and its physical significance, Schrodinger time dependent equation, Separation in time dependent and time independent parts, Operators in quantum Mechanics, Eigen functions and Eigen values, Particle in one dimensional and three dimensional box (Energy eigen values). Qualitative analysis of potential barrier Tunneling effect). Simple Harmonic Oscillator (Qualitative analysis of Zero point energy)
Mot

It is about molecular orbital theory specially mo diagram of diatomic atoms,their bond orders,bond lengths and stability and experimental evidences of ionisation energy from PES.
What's hot (20)
Determination of equivalence conductance, degree of dissociation and dissocia...

Determination of equivalence conductance, degree of dissociation and dissocia...
Lect. 23 rotational vibrational raman spectroscopy

Lect. 23 rotational vibrational raman spectroscopy
Schrodinger equation and its applications: Chapter 2

Schrodinger equation and its applications: Chapter 2
Similar to ψ And ψ2 significance
Stochastic Schrödinger equations

We use stochastic methods to present mathematically correct representation of the wave function. Informal construction was developed by R. Feynman. This approach were introduced first by H. Doss Sur une Resolution Stochastique de l'Equation de Schrödinger à Coefficients Analytiques. Communications in Mathematical Physics
October 1980, Volume 73, Issue 3, pp 247–264.
Primary intention is to discuss formal stochastic representation of the Schrodinger equation solution with its applications to the theory of demolition quantum measurements.
I will appreciate your comments.
Series solutions at ordinary point and regular singular point

ppt of Series solutions at ordinary point and regular singular point
Fuzzy random variables and Kolomogrov’s important results

:In this paper an attempt is made to transform Kolomogrov Maximal inequality, Koronecker Lemma, Loeve’s Lemma and Kolomogrov’s strong law of large numbers for independent, identically distributive fuzzy Random variables. The applications of this results is extensive and could produce intensive insights on Fuzzy Random variables
Similar to ψ And ψ2 significance (20)
Solution to schrodinger equation with dirac comb potential 

Solution to schrodinger equation with dirac comb potential
Series solutions at ordinary point and regular singular point

Series solutions at ordinary point and regular singular point
Solution of a singular class of boundary value problems by variation iteratio...

Solution of a singular class of boundary value problems by variation iteratio...
11.solution of a singular class of boundary value problems by variation itera...

11.solution of a singular class of boundary value problems by variation itera...
Digital Text Book :POTENTIAL THEORY AND ELLIPTIC PARTIAL DIFFERENTIAL EQUATIONS 

Digital Text Book :POTENTIAL THEORY AND ELLIPTIC PARTIAL DIFFERENTIAL EQUATIONS
PaperNo8-HabibiSafari-IJAM-CHAOTICITY OF A PAIR OF OPERATORS

PaperNo8-HabibiSafari-IJAM-CHAOTICITY OF A PAIR OF OPERATORS
Fuzzy random variables and Kolomogrov’s important results

Fuzzy random variables and Kolomogrov’s important results
More from Mithil Fal Desai
Goan Shimgo sahitya (some of the folk songs)

This is a small attempt to preserve heritage and folk culture of Goa
How long could one survive in a perfectly airtight room.docx

How long could one survive in a perfectly airtight room?
Given
Inhaled air contains 21% oxygen while exhaled breath contains approximately 16% oxygen and 5% carbon dioxide
The minimum oxygen level required for survival is 19 %.
Dimensions of room in meters: 5 X 5 X 3
Writing Third Year Project Report: Some Handy Guidelines

This is a brief presentation to elaborate the best practices in Academic Undergraduate Project writing
Determination of acidity of a water sample..pdf

The acidity of water is a measure of the quantitative capacity to react with a mineral base. The measured value usually varies with the pH indicator used in the experiment. The solution of phenolphthalein is colourless in acidic or mild basic medium (pH<8.0), while in basic medium (pH<9.6) solution is pink. The solution of methyl orange is generally red at pH lower than 3.1 and above pH 4.4 the solutions are yellow.
The dissolved strong mineral acids, weak organic acids, dissolved carbon dioxide and hydrolyzed mineral salts may contribute to the measured acidity of natural water. The acidity of water is a significant parameter of water as the presence of acids in natural water contributes to corrosiveness and influences chemical reaction rates. The biological processes are dependent on the acidity of water. The quantitative acidity measurement also reflects the quality of the source of water. The acidity of water is usually expressed in terms of CaCO3.
Determine the strength in grams per litre of a given AgNO3 solution being pro...

Mohr’s method is used to determine the chloride ion concentration by titrating with silver nitrate. Using pure sodium chloride the strength of silver ions can be also determined as the Ag(I) ions react with chloride ions, quantitatively, to form a precipitate of silver chloride.
अडण्णे शिगमो लोकसाहित्य

This is a small attempt to preserve heritage and folk culture of Goan village Adnem
Credits to Ghanashyam K. Devidas, Lakshimikant Bhavu, Vithobha Bhavu
Estimation of water of crystallization in mohr’s salt by titrating with stand...

water of crystallization
More from Mithil Fal Desai (20)
How long could one survive in a perfectly airtight room.docx

How long could one survive in a perfectly airtight room.docx
Writing Third Year Project Report: Some Handy Guidelines

Writing Third Year Project Report: Some Handy Guidelines
Determine the strength in grams per litre of a given AgNO3 solution being pro...

Determine the strength in grams per litre of a given AgNO3 solution being pro...
Estimation of water of crystallization in mohr’s salt by titrating with stand...

Estimation of water of crystallization in mohr’s salt by titrating with stand...
Recently uploaded
Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46

Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46
Event Link:-
https://meetups.mulesoft.com/events/details/mulesoft-mysore-presents-exploring-gemini-ai-and-integration-with-mulesoft/
Agenda
● Java 17 Upgrade Overview
● Why and by when do customers need to upgrade to Java 17?
● Is there any immediate impact to upgrading to Mule Runtime 4.6 and beyond?
● Which MuleSoft products are in scope?
For Upcoming Meetups Join Mysore Meetup Group - https://meetups.mulesoft.com/mysore/
YouTube:- youtube.com/@mulesoftmysore
Mysore WhatsApp group:- https://chat.whatsapp.com/EhqtHtCC75vCAX7gaO842N
Speaker:-
Shubham Chaurasia - https://www.linkedin.com/in/shubhamchaurasia1/
Priya Shaw - https://www.linkedin.com/in/priya-shaw
Organizers:-
Shubham Chaurasia - https://www.linkedin.com/in/shubhamchaurasia1/
Giridhar Meka - https://www.linkedin.com/in/giridharmeka
Priya Shaw - https://www.linkedin.com/in/priya-shaw
Shyam Raj Prasad-
https://www.linkedin.com/in/shyam-raj-prasad/
Polish students' mobility in the Czech Republic

Polish students mobility to the Czech Republic within eTwinning project "Medieval adventures with Marco Polo"
Operation Blue Star - Saka Neela Tara

Operation “Blue Star” is the only event in the history of Independent India where the state went into war with its own people. Even after about 40 years it is not clear if it was culmination of states anger over people of the region, a political game of power or start of dictatorial chapter in the democratic setup.
The people of Punjab felt alienated from main stream due to denial of their just demands during a long democratic struggle since independence. As it happen all over the word, it led to militant struggle with great loss of lives of military, police and civilian personnel. Killing of Indira Gandhi and massacre of innocent Sikhs in Delhi and other India cities was also associated with this movement.
Sectors of the Indian Economy - Class 10 Study Notes pdf

The Indian economy is classified into different sectors to simplify the analysis and understanding of economic activities. For Class 10, it's essential to grasp the sectors of the Indian economy, understand their characteristics, and recognize their importance. This guide will provide detailed notes on the Sectors of the Indian Economy Class 10, using specific long-tail keywords to enhance comprehension.
For more information, visit-www.vavaclasses.com
The geography of Taylor Swift - some ideas

Geographical themes connected with Taylor Swift's ERAS tour - coming to the UK in June 2024
ESC Beyond Borders _From EU to You_ InfoPack general.pdf

ESC Beyond Borders _From EU to You_ InfoPack general.pdfFundacja Rozwoju Społeczeństwa Przedsiębiorczego
Wolontariat grupowyHome assignment II on Spectroscopy 2024 Answers.pdf

Answers to Home assignment on UV-Visible spectroscopy: Calculation of wavelength of UV-Visible absorption
Chapter 3 - Islamic Banking Products and Services.pptx

Chapter 3 - Islamic Banking Products and Services.pptxMohd Adib Abd Muin, Senior Lecturer at Universiti Utara Malaysia
This slide is prepared for master's students (MIFB & MIBS) UUM. May it be useful to all.The French Revolution Class 9 Study Material pdf free download

The French Revolution, which began in 1789, was a period of radical social and political upheaval in France. It marked the decline of absolute monarchies, the rise of secular and democratic republics, and the eventual rise of Napoleon Bonaparte. This revolutionary period is crucial in understanding the transition from feudalism to modernity in Europe.
For more information, visit-www.vavaclasses.com
Palestine last event orientationfvgnh .pptx

An EFL lesson about the current events in Palestine. It is intended to be for intermediate students who wish to increase their listening skills through a short lesson in power point.
TESDA TM1 REVIEWER FOR NATIONAL ASSESSMENT WRITTEN AND ORAL QUESTIONS WITH A...

TESDA TM1 REVIEWER FOR NATIONAL ASSESSMENT WRITTEN AND ORAL QUESTIONS WITH ANSWERS.
Template Jadual Bertugas Kelas (Boleh Edit)

Jadual Bertugas kelas dalam bentuk softcopy PowerPoint. Cikgu boleh download >> edit >> print >> laminate. Semoga bermanfaat.
MARUTI SUZUKI- A Successful Joint Venture in India.pptx

Let us know about Maruti Suzuki, a successful Joint venture in India.
Cambridge International AS A Level Biology Coursebook - EBook (MaryFosbery J...

for studentd in cabridge board
Thesis Statement for students diagnonsed withADHD.ppt

Presentation required for the master in Education.
Recently uploaded (20)
Basic phrases for greeting and assisting costumers

Basic phrases for greeting and assisting costumers
Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46

Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46
Sectors of the Indian Economy - Class 10 Study Notes pdf

Sectors of the Indian Economy - Class 10 Study Notes pdf
ESC Beyond Borders _From EU to You_ InfoPack general.pdf

ESC Beyond Borders _From EU to You_ InfoPack general.pdf
Home assignment II on Spectroscopy 2024 Answers.pdf

Home assignment II on Spectroscopy 2024 Answers.pdf
Chapter 3 - Islamic Banking Products and Services.pptx

Chapter 3 - Islamic Banking Products and Services.pptx
The French Revolution Class 9 Study Material pdf free download

The French Revolution Class 9 Study Material pdf free download
TESDA TM1 REVIEWER FOR NATIONAL ASSESSMENT WRITTEN AND ORAL QUESTIONS WITH A...

TESDA TM1 REVIEWER FOR NATIONAL ASSESSMENT WRITTEN AND ORAL QUESTIONS WITH A...
MARUTI SUZUKI- A Successful Joint Venture in India.pptx

MARUTI SUZUKI- A Successful Joint Venture in India.pptx
Cambridge International AS A Level Biology Coursebook - EBook (MaryFosbery J...

Cambridge International AS A Level Biology Coursebook - EBook (MaryFosbery J...
Thesis Statement for students diagnonsed withADHD.ppt

Thesis Statement for students diagnonsed withADHD.ppt
ψ And ψ2 significance
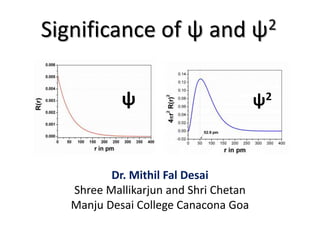
- 1. Significance of ψ and ψ2 Dr. Mithil Fal Desai Shree Mallikarjun and Shri Chetan Manju Desai College Canacona Goa ψ ψ2
- 2. Ψ and Ψ2 Ψ (Psi) is a wave function represented in the Schrodinger wave equation, which represents the state of the electron. Its corresponding probability density (Ψ2) gives the probability of finding the electron in a particular region in space.
- 4. Acceptable solutions to the Schrodinger wave equation must have certain properties Ψ must be continuous Ψ must be finite Ψ must be single valued 0 ∞ Ψ𝟐 = 1
- 5. Continuous and noncontinuous function -1 -0.5 0 0.5 1 0 100 200 300 400 sin(θ) -6 -4 -2 0 2 4 6 0 100 200 300 400 tan(θ) Continuous function noncontinuous function θ θ
- 6. Finite and infinite function A = {0, 1, 2, 3,4, 5} finite set A = {0, 1, 2, 3,4, 5,……} infinite set 𝑓(𝑥) = 𝑥 + −1𝐴 = 𝜋𝑟2 𝑓(𝑥) = 𝑥 0 𝑓(𝑥) = 𝑥2 ×(∞)
- 7. -10 -5 0 5 10 0 20 40 60 80 100 More than one value for the function y x At same value of x, we observe two different value of Y 𝑦2 = 4𝑝𝑥 27
- 8. Integrating function from a to b Integrating any function gives area under the curve formed by that function 𝑎 𝑏 𝑓𝑥 𝑑𝑥 = 𝑎𝑟𝑒𝑎 3 4 π 1 2 π 1 4 π π 0π 0.314 0.314 0.314 0.314 0.314 0.314 0.314 0.314 0.314 0.314 0 0.4 0.8 0 π 𝑠𝑖𝑛𝑥 𝑑𝑥 = 2 𝑓(𝑥) = 𝑠𝑖𝑛𝑥
- 9. Function and function square -1 -0.75 -0.5 -0.25 0 0.25 0.5 0.75 1 0 100 200 300 sin(θ) or [sin(θ)] 2 θ
- 10. End note Several wave function satisfy these properties and each of these have corresponding energies. These wave functions are called orbitals. In hydrogen atom the wave function which has lowest energy level(E1) is 𝝍1. There are number of acceptable solutions of the wave function and each orbital is described by set of three quantum numbers n, l and m.