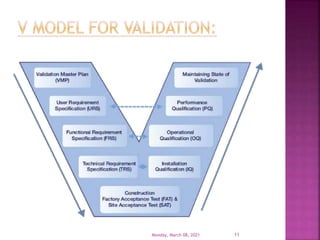
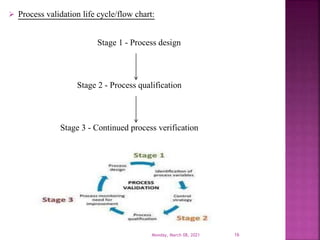
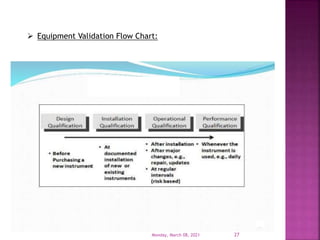
This document summarizes a presentation on validation given by Prajakta Sawant, a first year M.Pharm student at Alard College of Pharmacy in Pune, India. The presentation covered the need for validation, types of validation including process, cleaning, equipment and analytical method validation. It discussed validation concepts such as the validation master plan, documentation, and ICH and WHO guidelines. The goal of validation is to ensure consistent production of pharmaceuticals meeting quality standards.