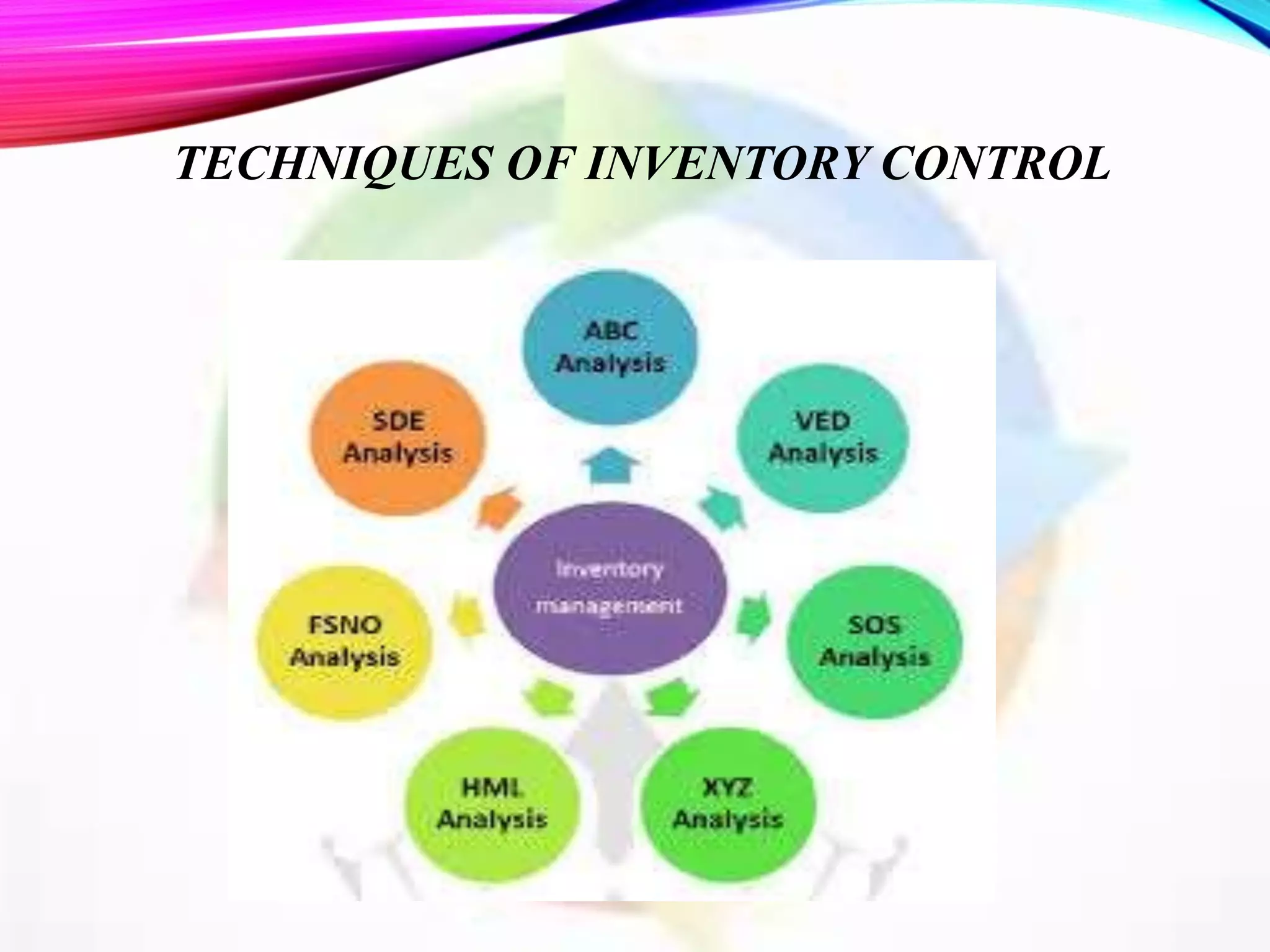
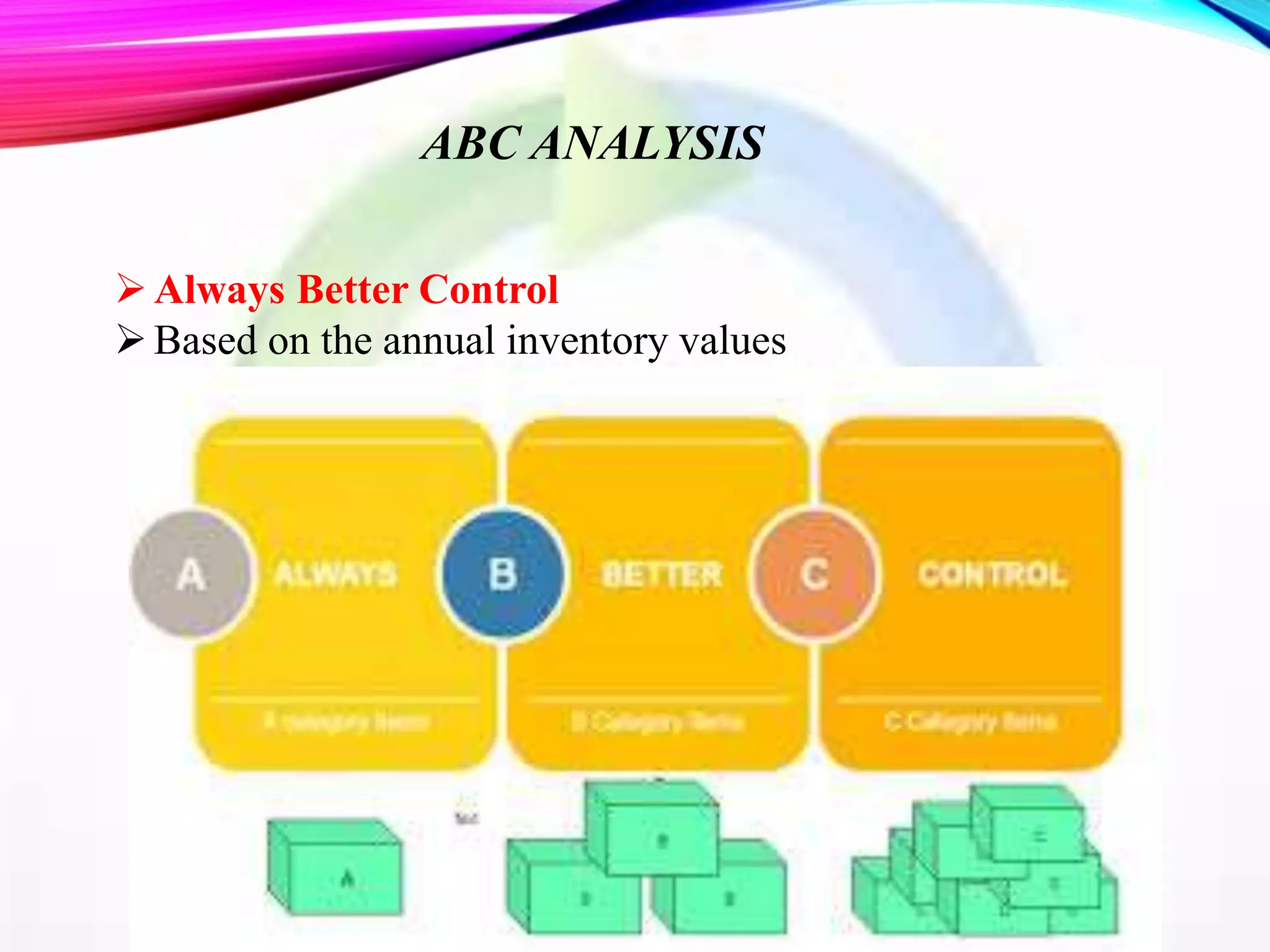
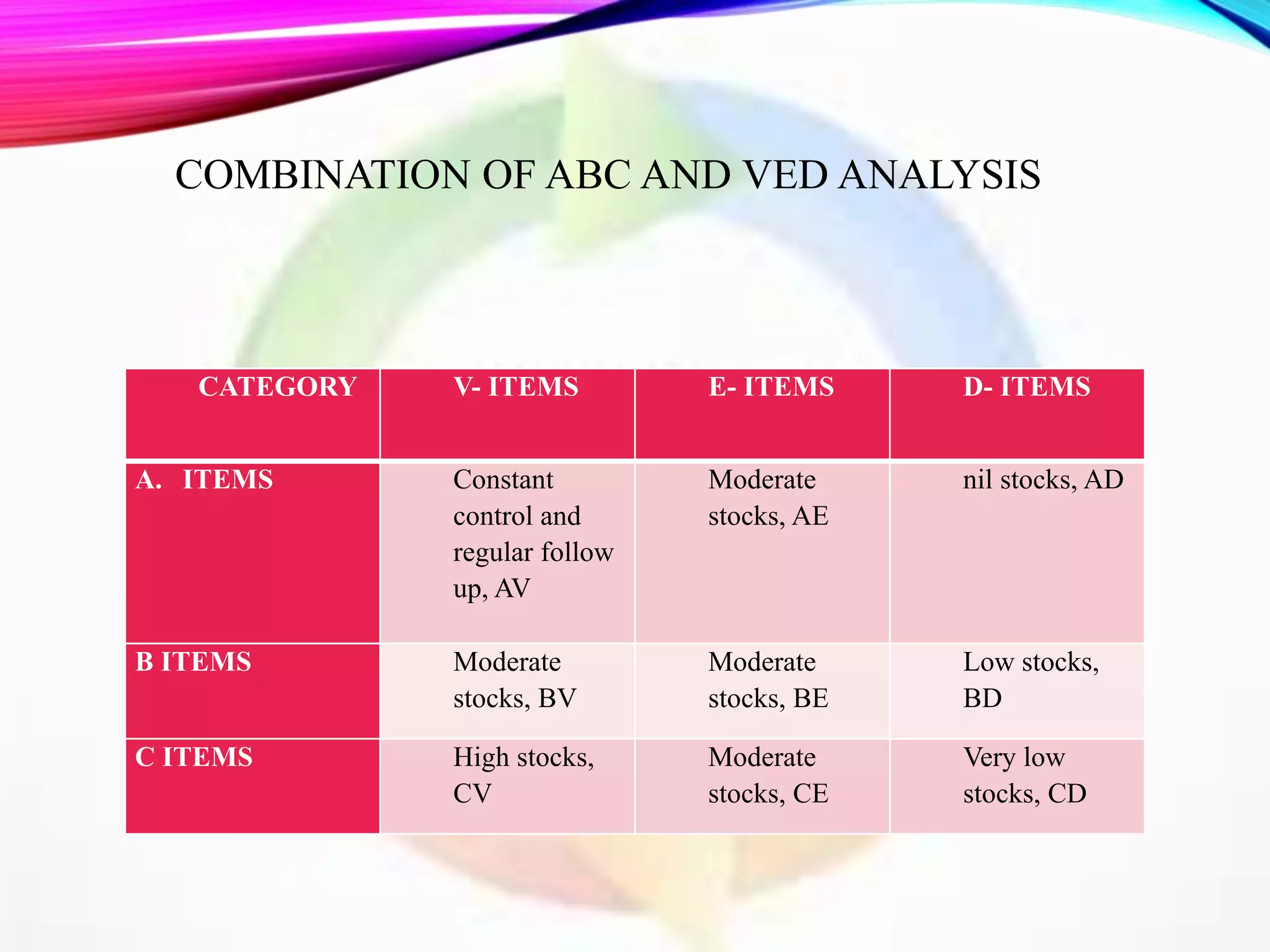
This document discusses objectives and policies of CGMP (current good manufacturing practices) and inventory management and control. It outlines the importance of CGMP in assuring quality standards and preventing issues. CGMP regulations provide systems to properly design, monitor, and control manufacturing processes. The document also describes objectives of inventory control such as minimizing costs and ensuring adequate stock levels. It provides details on inventory management policies, documentation requirements, and quality control standards under CGMP.