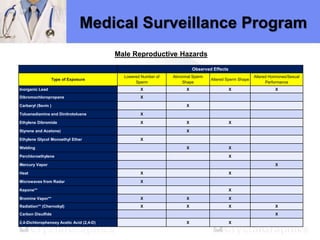
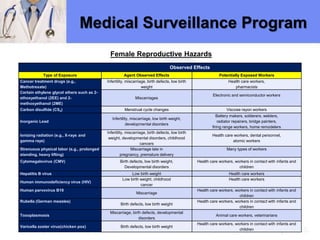
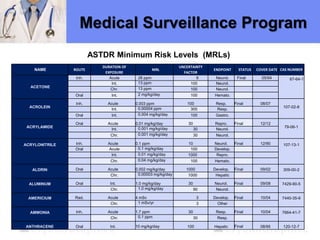
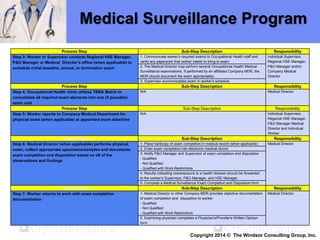
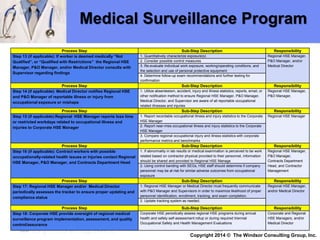
The document outlines a medical surveillance program for workers in the Caribbean upstream oil and gas industry, detailing various examinations and assessments needed to ensure occupational health and safety. It includes pre-placement and periodic evaluations to monitor exposure to hazardous substances, fitness for duty assessments, and emergency treatment provisions. Additionally, it emphasizes the importance of identifying individual risk factors and implementing appropriate protective measures to maintain worker health.










![Name, address, and phone number (including fax) of physician/ health center performing examination:
New Applicants ONLY:
Your Current Occupation:____
Your Current Employer:
Time in Current Position (in years/months):
Name of Department:
Examinee’s Name: Position/Job Title: SS#
Address: Work Location: Region:
Home Phone: Work Phone:
Date of Scheduled Exam: Date of Birth: Gender: Male Female
REPSOL PROGRAM MANAGER
EXAMINING PHYSICIAN (Please Note - Core Exam Must Always be Completed, Plus All Function-Specific Services
TYPE OF EXAMINATION
o Pre-placement/Baseline/Exit o Periodic o Exit
SPECIFY FUNCTION AND/OR CLEARANCES REQUESTED
(Check ALL That Apply)
Respirator User [complete Request for Respirator Clearance
form]
Health, Safety, and Environmental
Subsurface Reservoir Development
Commercial Drivers License
Facilities and Construction
Diver and Diving Team
Well Maintenance, Deck, Inspection, and Construction
Crane/Derrick/Aerial Lift Operator/Rigger
Commercial Vehicle Operator
Security
E&P Operated Asset – TSP
Laboratory/Chemical Worker
Well Drill and Oil/Gas Production
PRE-PLACEMENT/BASELINE CORE EXAM
Required Services: (Check those services completed)
Authorization for Disclosure Form
General Medical History
General Physical Examination
Chemistry Panel (including Glucose, Bilirubin (total),
Cholesterol, HDL-C, LDL-C, Triglycerides, GGTP, LDH,
SGOT, SGPT), Complete Blood Count, and Urinalysis
Urinalysis
Audiometry (including noise exposure history)
Electrocardiogram
Spirometry
Vision Screening (Corrected and Uncorrected Near and Far;
Color; Peripheral; Depth Perception)
Drug/Alcohol screen (cocaine/marijuana)
Plus other Function or Clearance-required services (see the
following page)
ANNUAL/PERIODIC/EXIT CORE EXAM
Required Services: (Check those services completed)
Authorization for Disclosure Form
General Medical History
General Physical Examination
Chemistry Panel (including Glucose, Bilirubin (total),
Cholesterol, HDL-C, LDL-C, Triglycerides, GGTP, LDH,
SGOT, SGPT), Complete Blood Count, and Urinalysis
Plus other Function or Clearance-required services (see
the following page)
Note: For Respirator User exams, the General Physical
Examination may be a brief, limited exam or a more extensive
exam, depending on the health of the examinee and the
judgment of the examiner. Also, laboratory tests (e.g.,
chemistry panel, blood count, and urinalysis) and procedures
(e.g., electrocardiograms) are intended to be at the discretion
of the examiner, rather than required services. Refer to the
Occupational Medicine Surveillance Program for further
guidance. For all Respirator User exams, completion of the
Request for Respirator Clearance form must precede this exam
and be attached to this exam form when completed.
Medical Surveillance Program
Standard Medical History and Examination Form
The individual to be examined is to complete the shaded medical history portions of this form prior to his/her appointment.](https://image.slidesharecdn.com/themodelformedicalsurveillanceprogramforcaribbean-140609140234-phpapp02/85/The-Model-for-Medical-Surveillance-Program-for-Upstream-Oil-Gas-Producer-11-320.jpg)