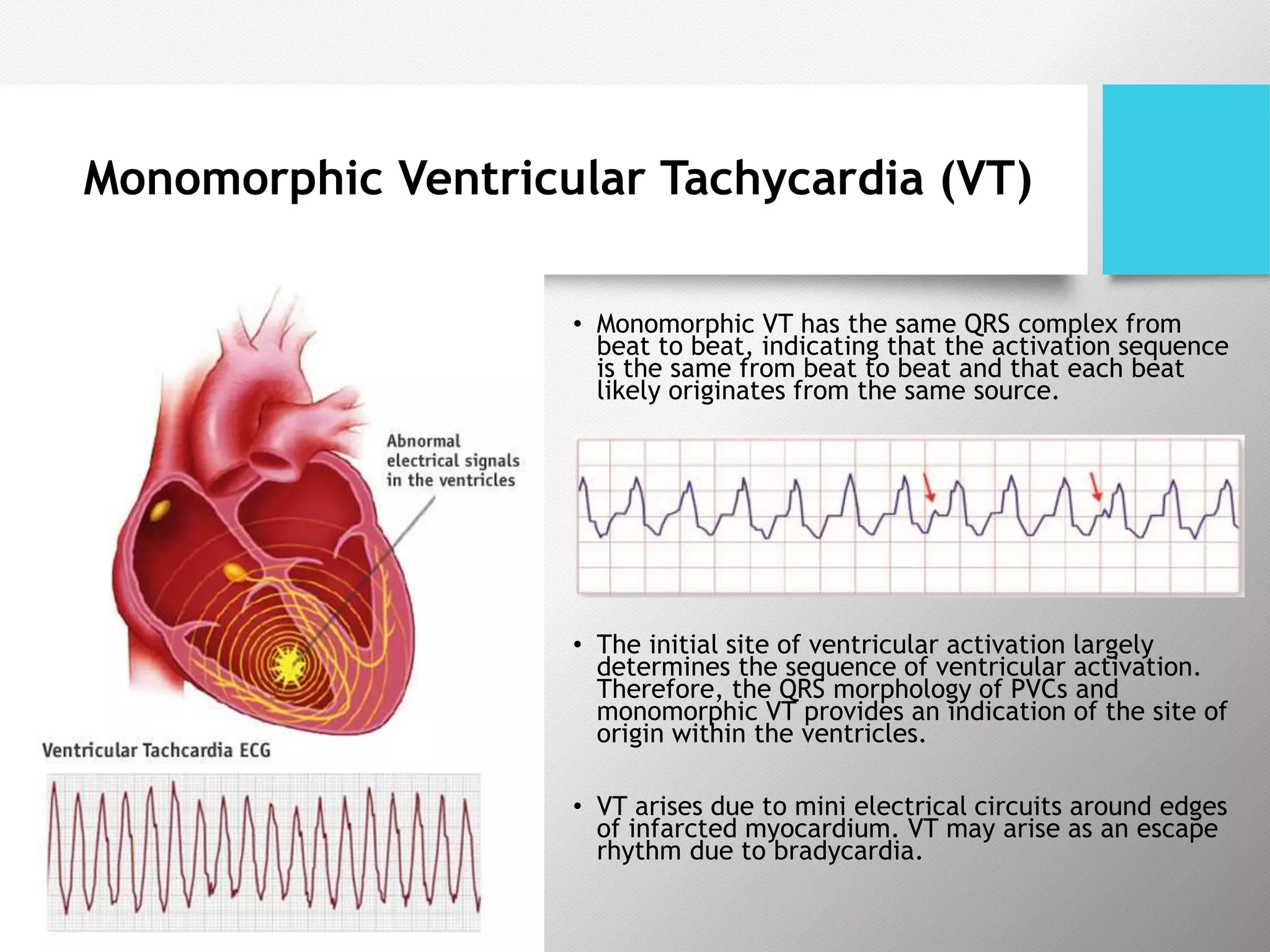
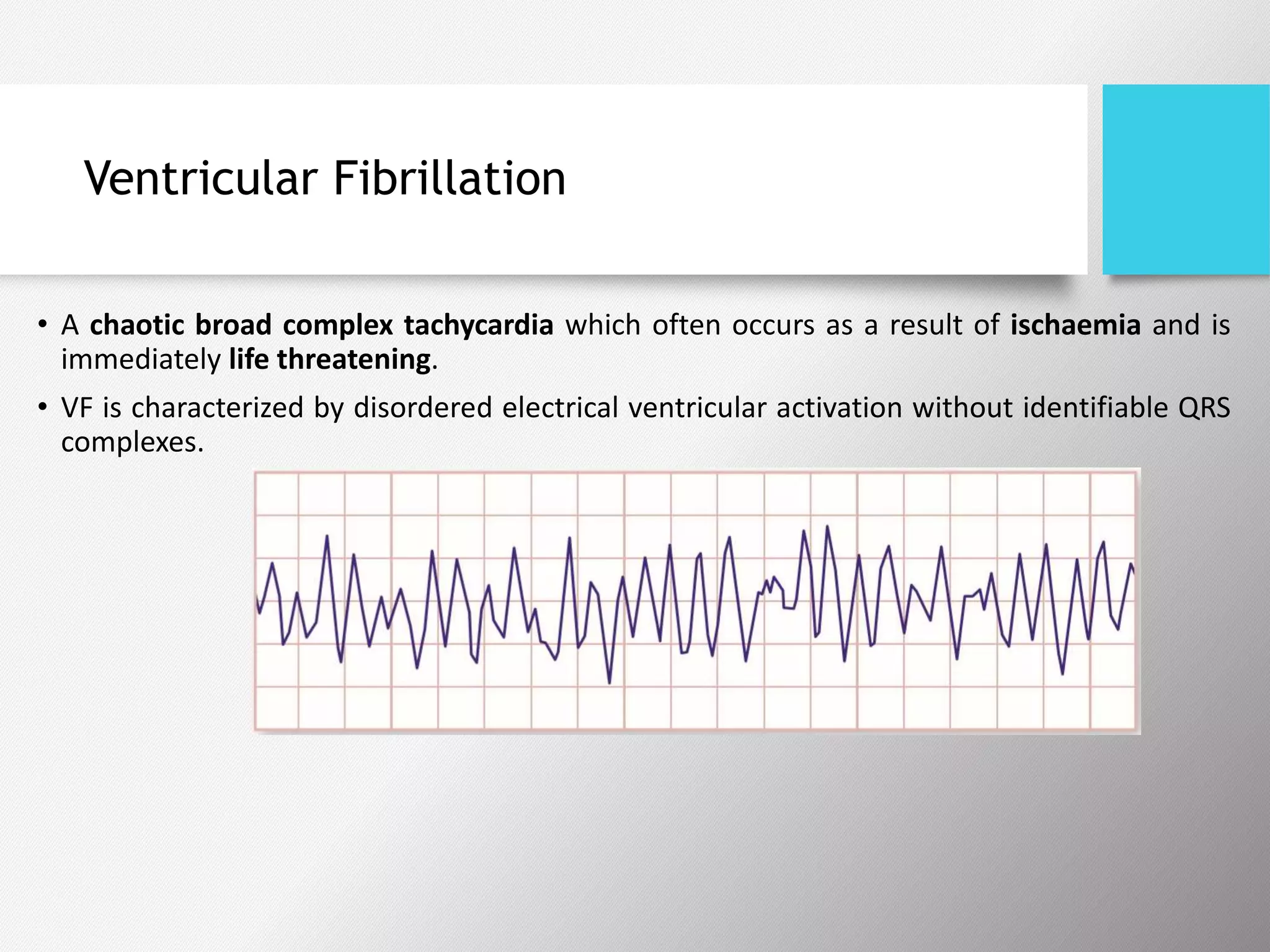
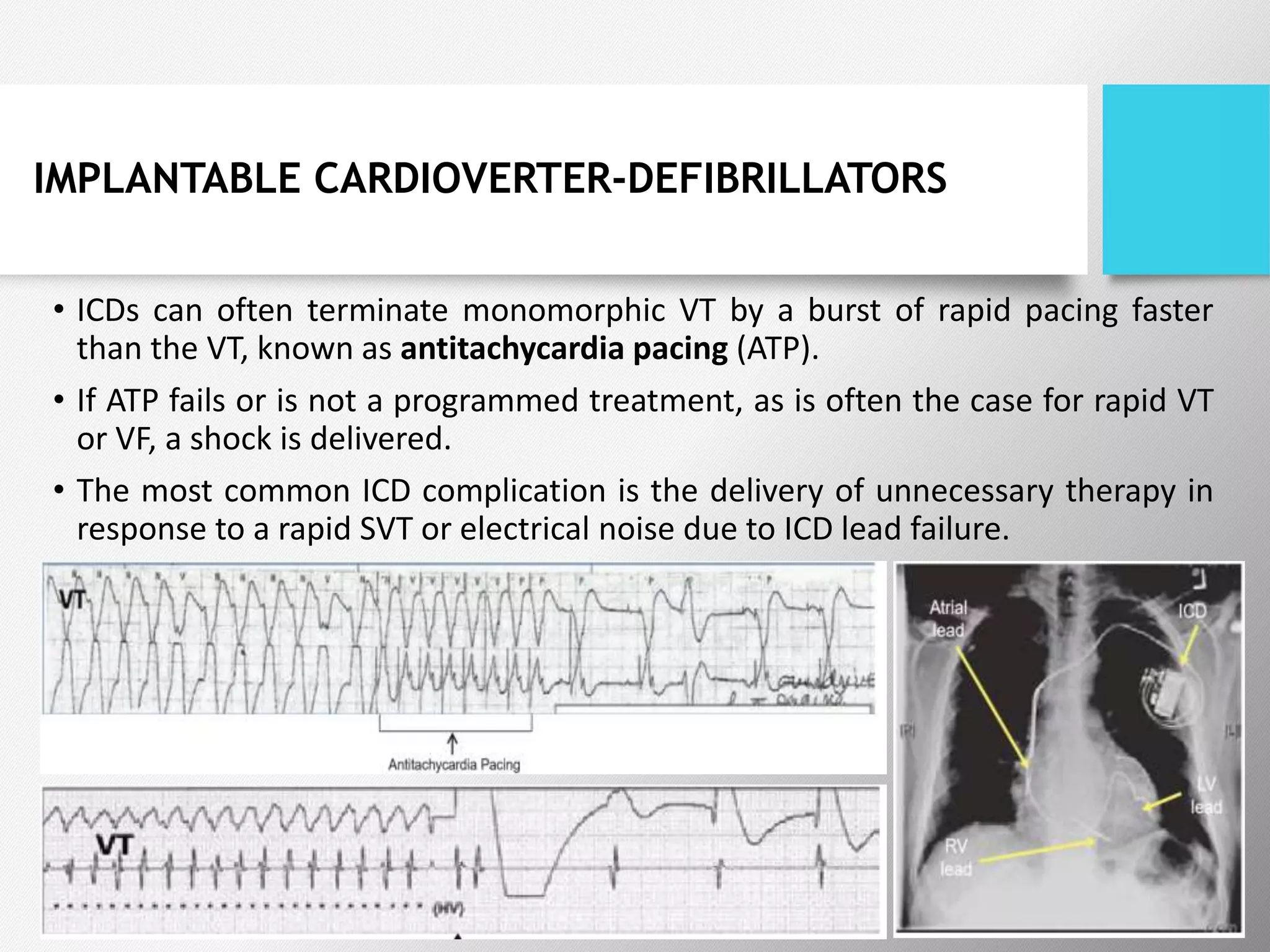
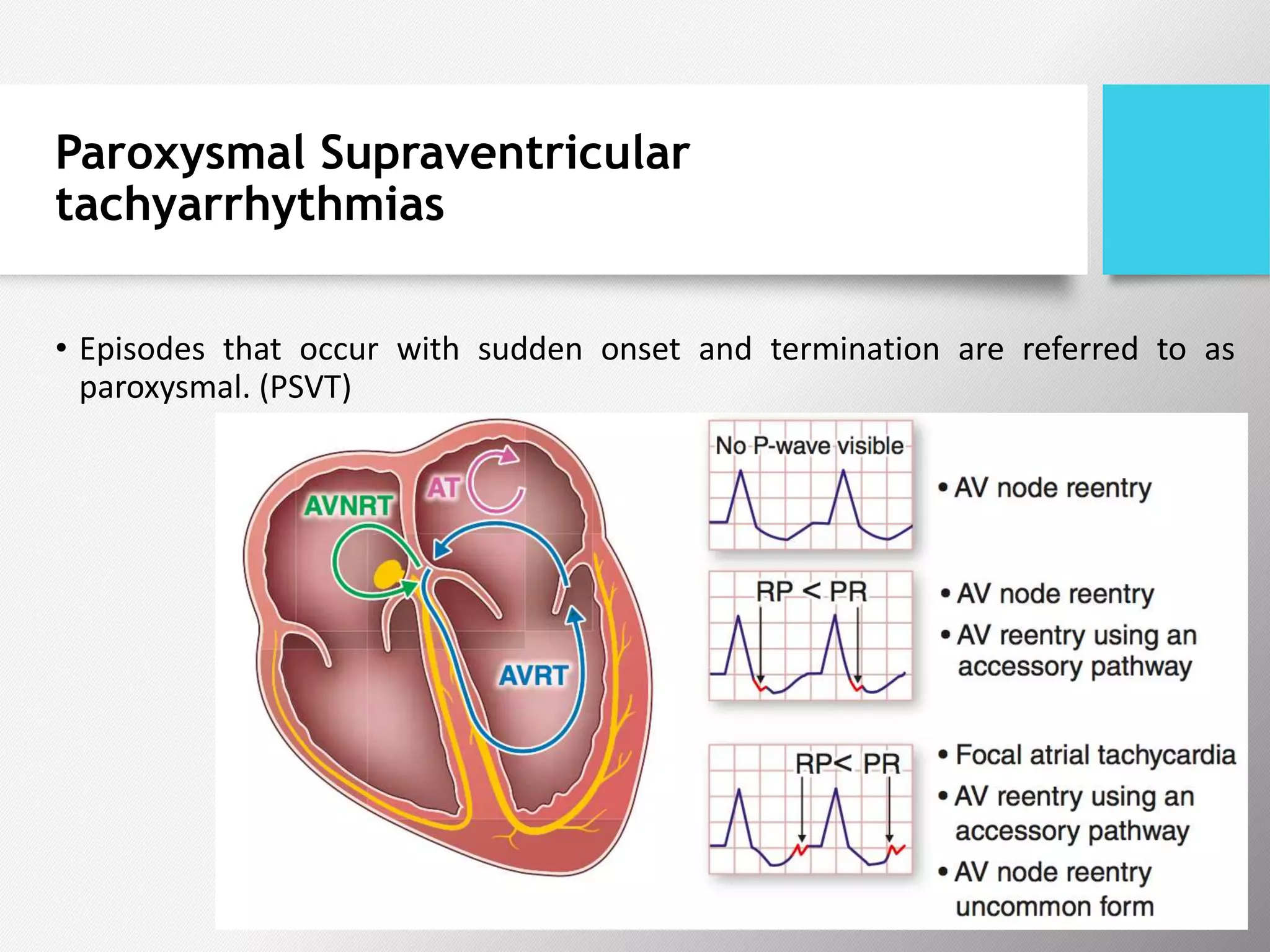
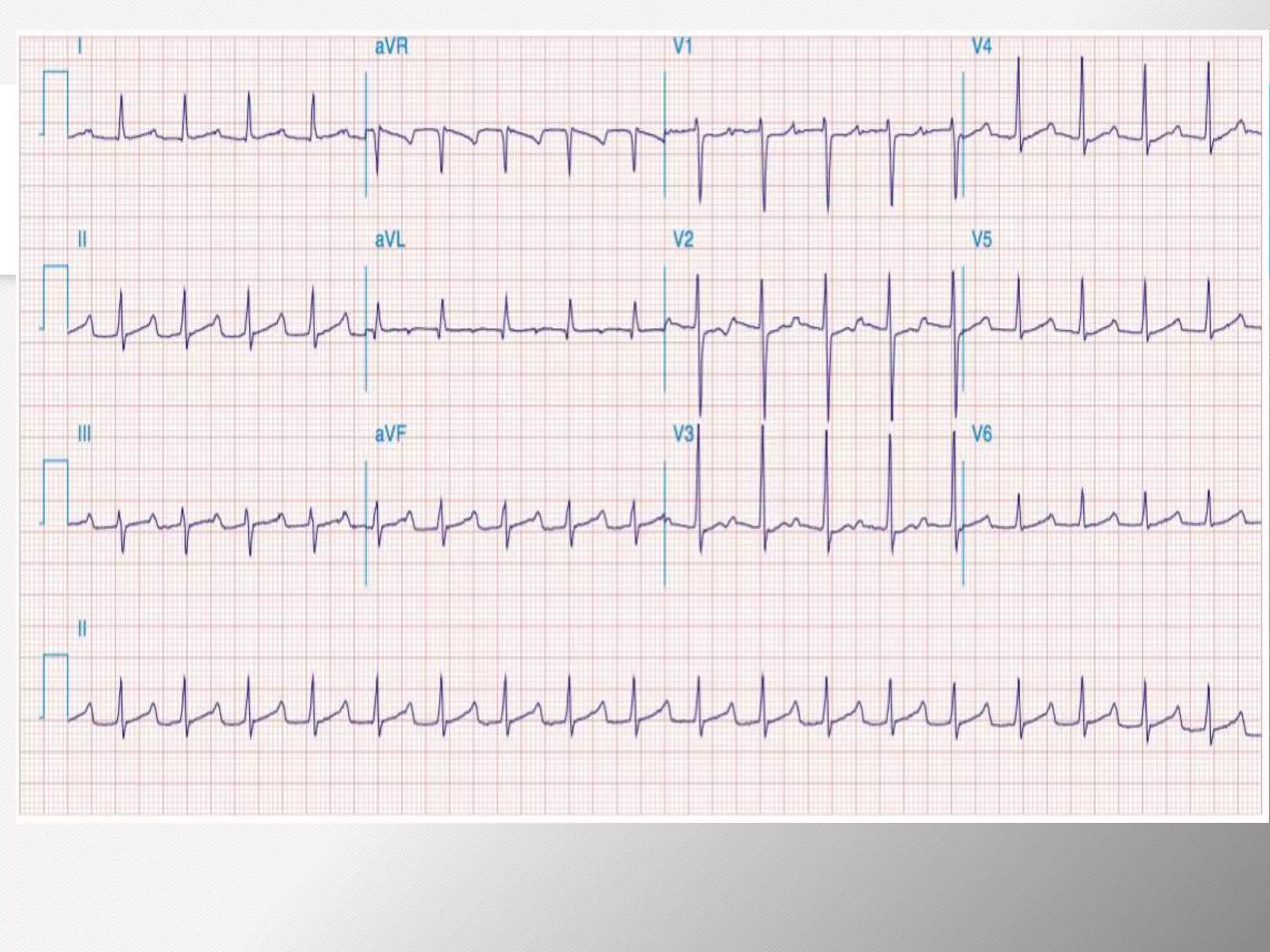
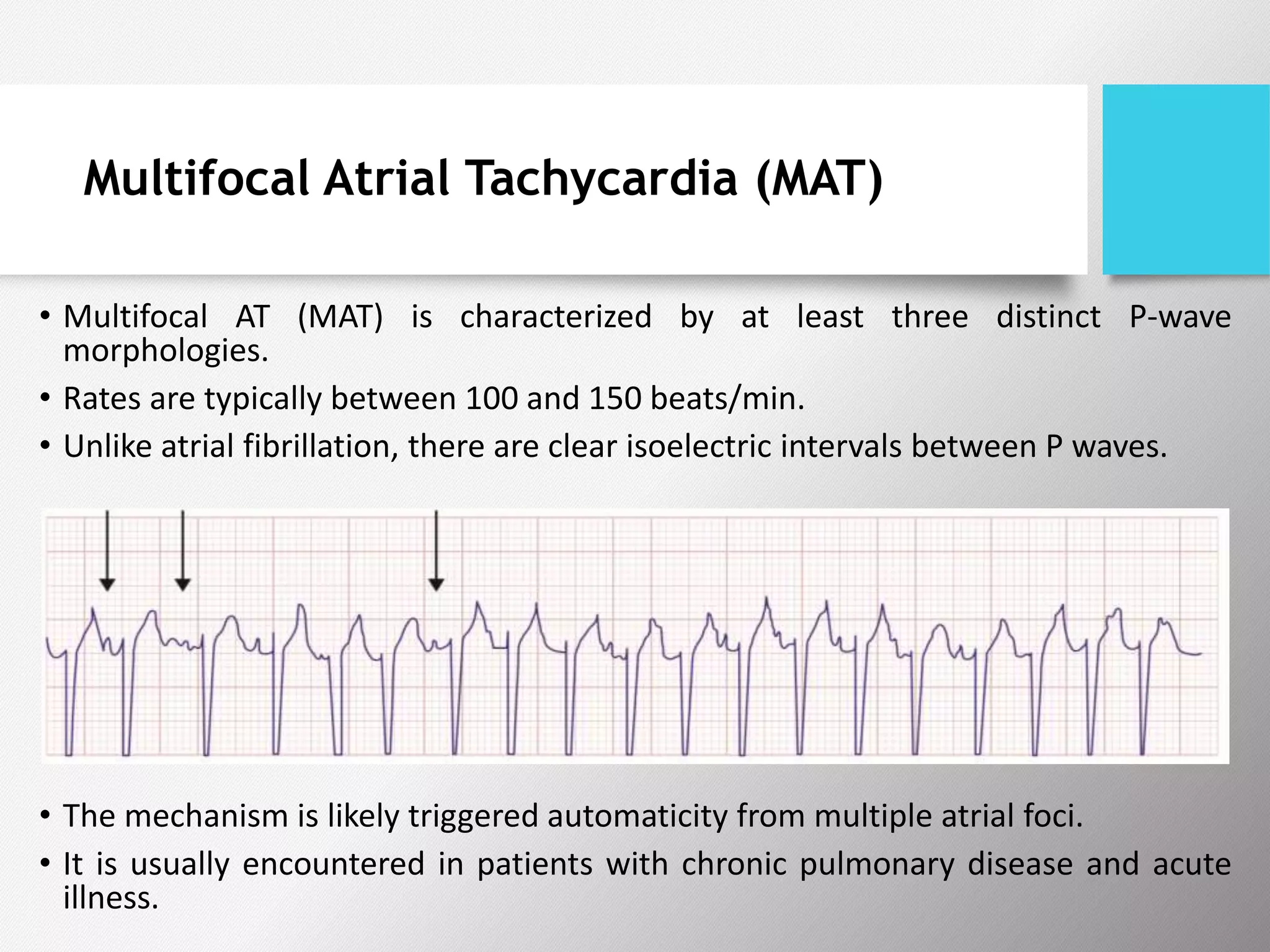
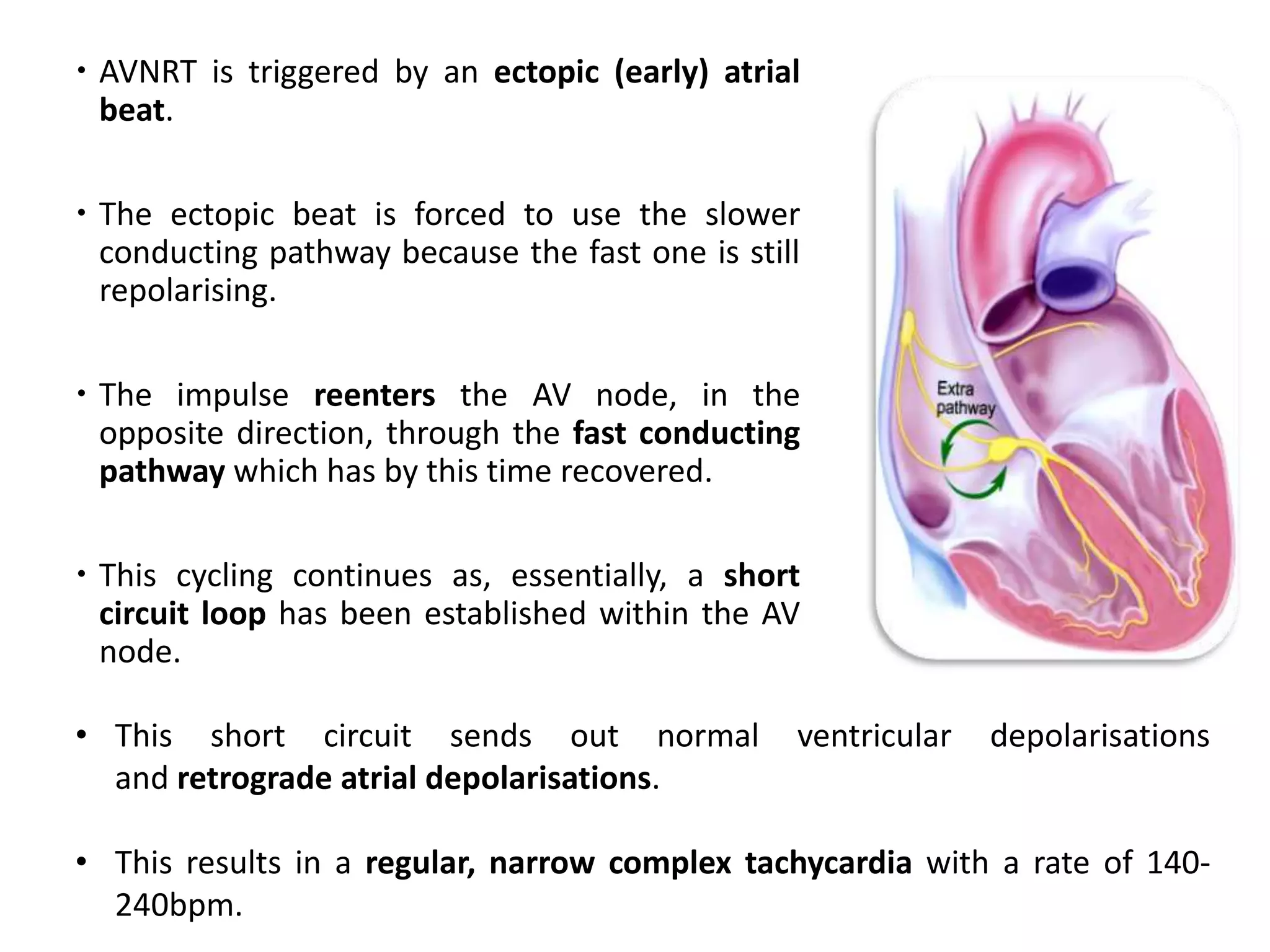
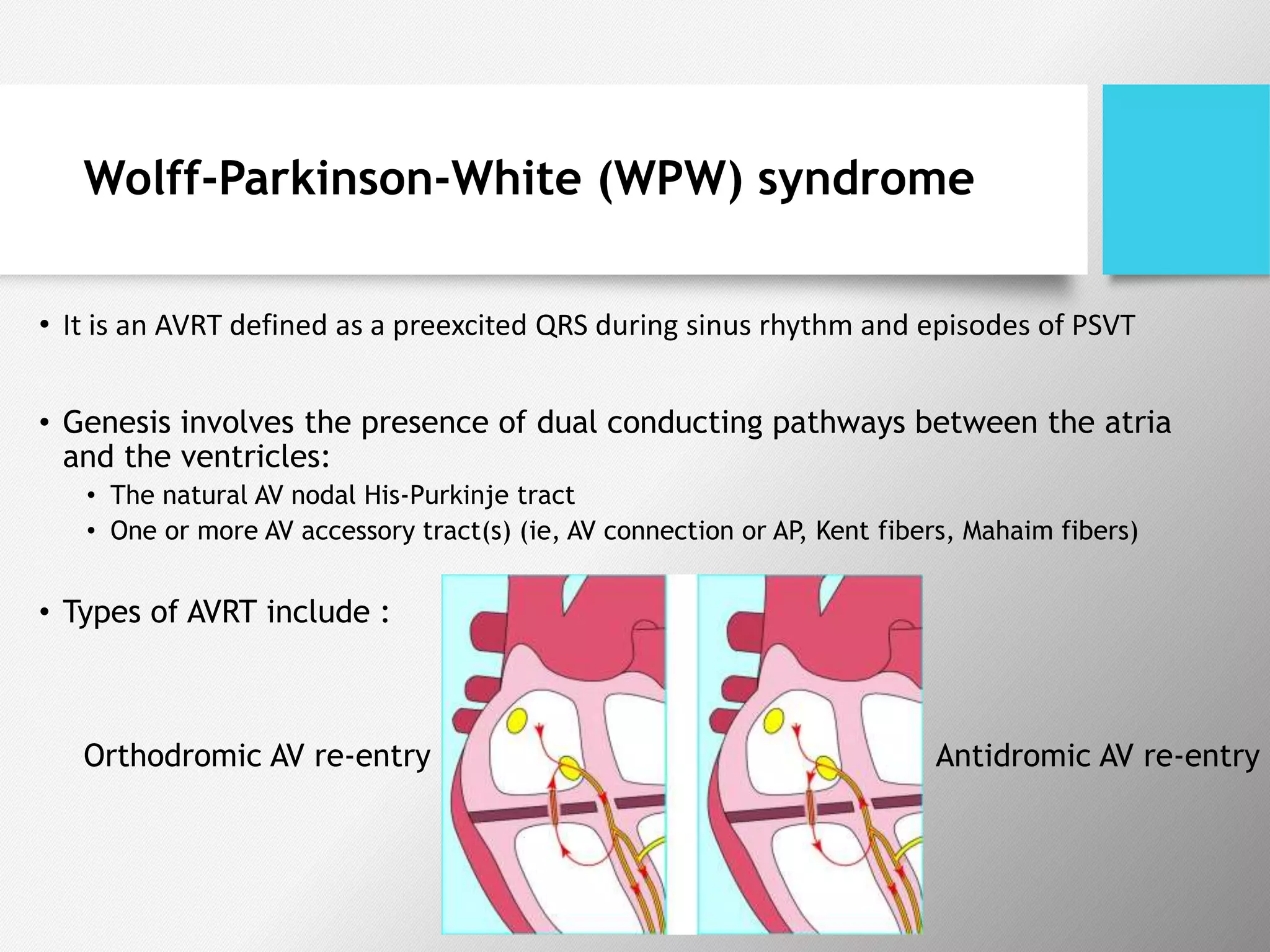
Tachyarrhythmias are classified based on the mechanisms of electrical impulse generation and conduction abnormalities. They can exhibit either broad or narrow QRS complexes on ECG and are categorized further into various types such as atrial flutter, atrial fibrillation, and different forms of supraventricular tachycardia. Treatment may include techniques like vagal maneuvers, medications, and radiofrequency ablation, depending on the specific type of tachyarrhythmia present.





















































![Premature ventricular beats
• also referred to as premature ventricular contractions [PVCs]are single ventricular beats
that fall earlier than the next anticipated supraventricular beat.
• PVCs that originate from the same focus will have the same QRS morphology and are
referred to as unifocal.
• PVCs that originate from different ventricular sites have different QRS morphologies and
are referred to as multifocal.
• Two consecutive ventricular beats are ventricular couplets.](https://image.slidesharecdn.com/tachyarrhythmias-seminar-arun-28march2016autosaved-171016040821/75/Tachy-Arrhythmias-Approach-to-Management-61-2048.jpg)