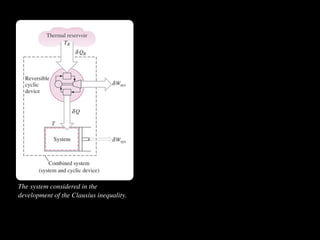
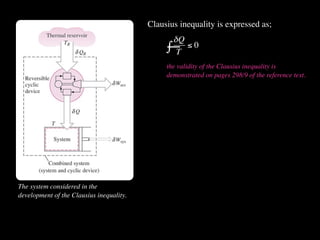
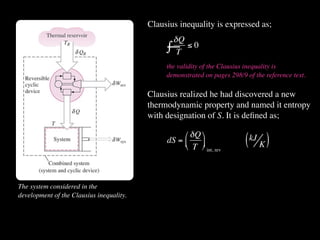
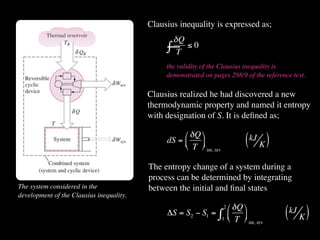
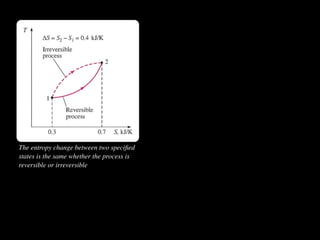
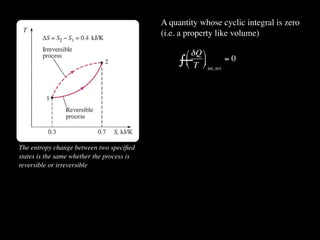
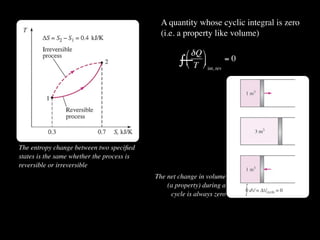
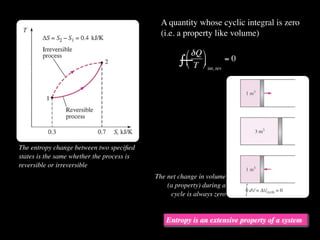
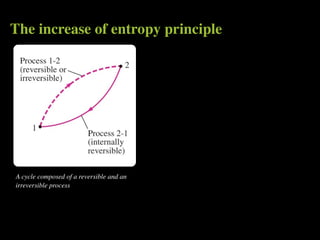
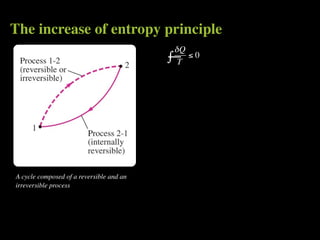
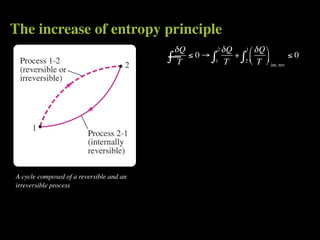
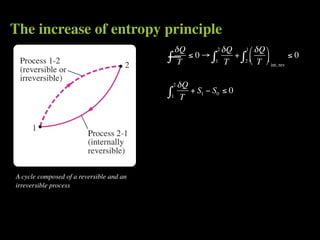
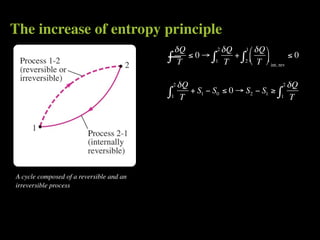
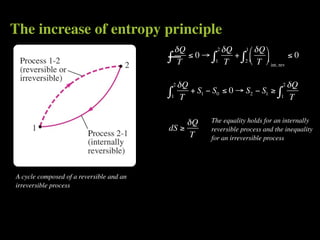
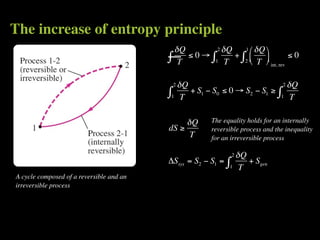
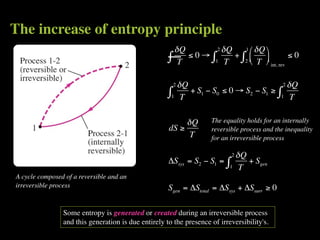
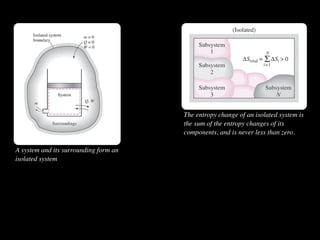
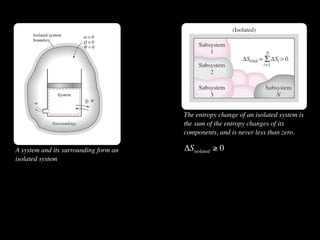
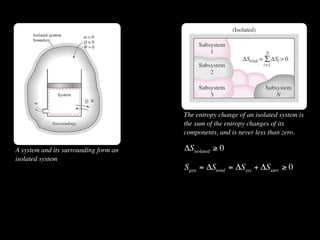
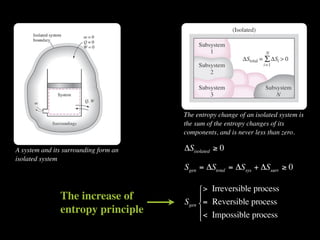
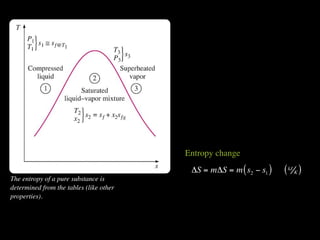
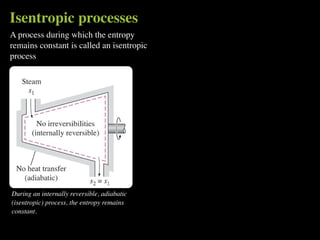
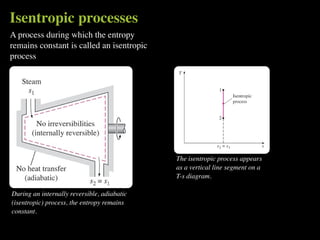
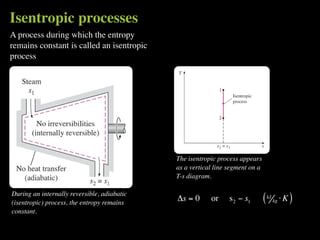
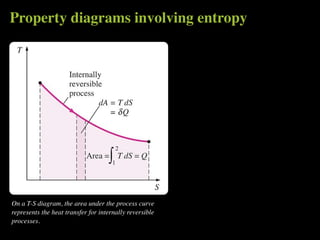
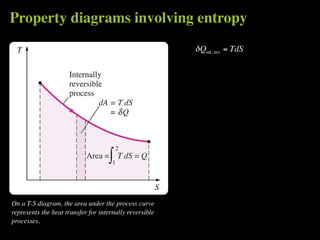
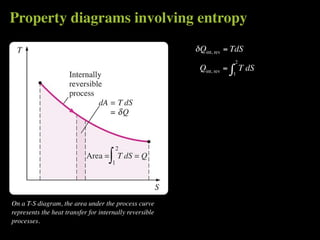
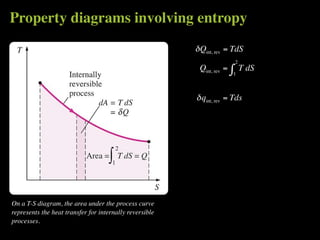
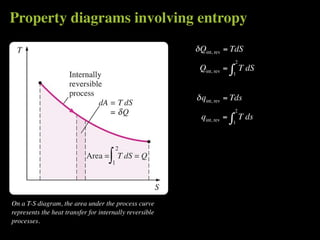
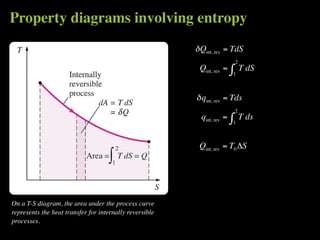
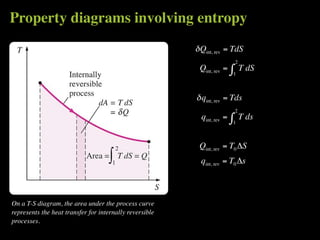
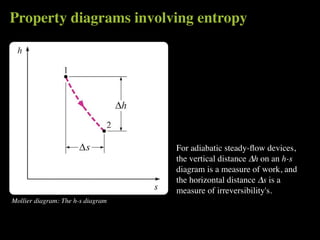
The document discusses entropy and the second law of thermodynamics. It defines the objectives as applying the second law to processes, defining entropy to quantify second law effects, and establishing the increase of entropy principle. It then defines entropy as an abstract property, discusses Clausius inequality and how Clausius realized he discovered entropy. The key points are that entropy is a state function, its change depends on initial and final states only, and the change between two states is the same for reversible and irreversible processes. It also provides an equation for determining entropy change during isothermal heat transfer.