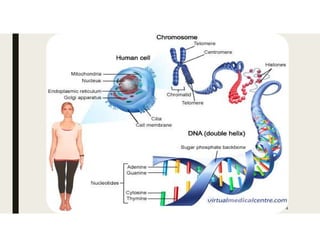
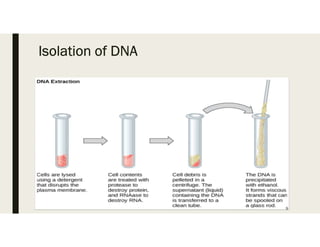
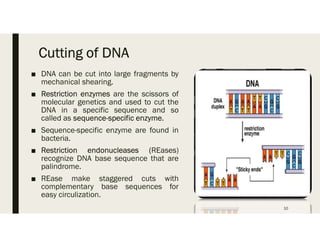
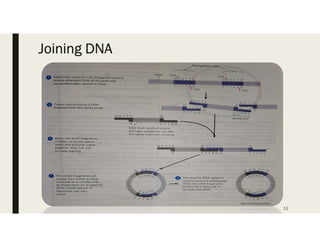
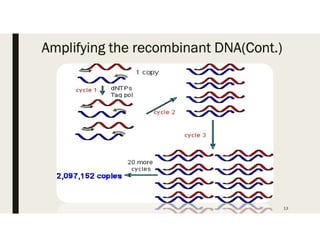
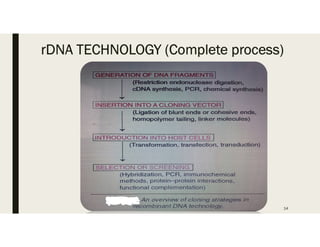
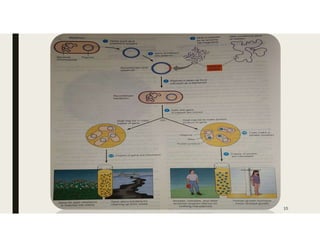
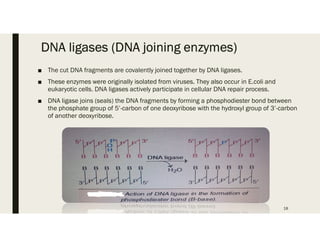
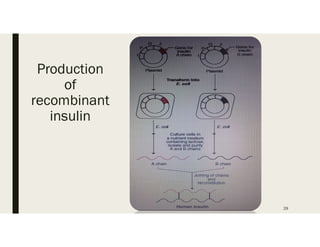
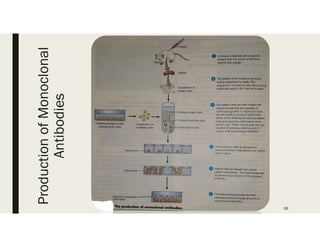
This document provides an extensive overview of recombinant DNA technology and hybridoma technology, explaining their definitions, principles, processes, tools, and applications. It details the significance of biotechnology in areas such as agriculture, pharmacology, and medicine, highlighting various recombinant products like insulin and monoclonal antibodies. Additionally, it discusses the advantages and limitations of these technologies, setting the stage for future advancements in biotechnology.