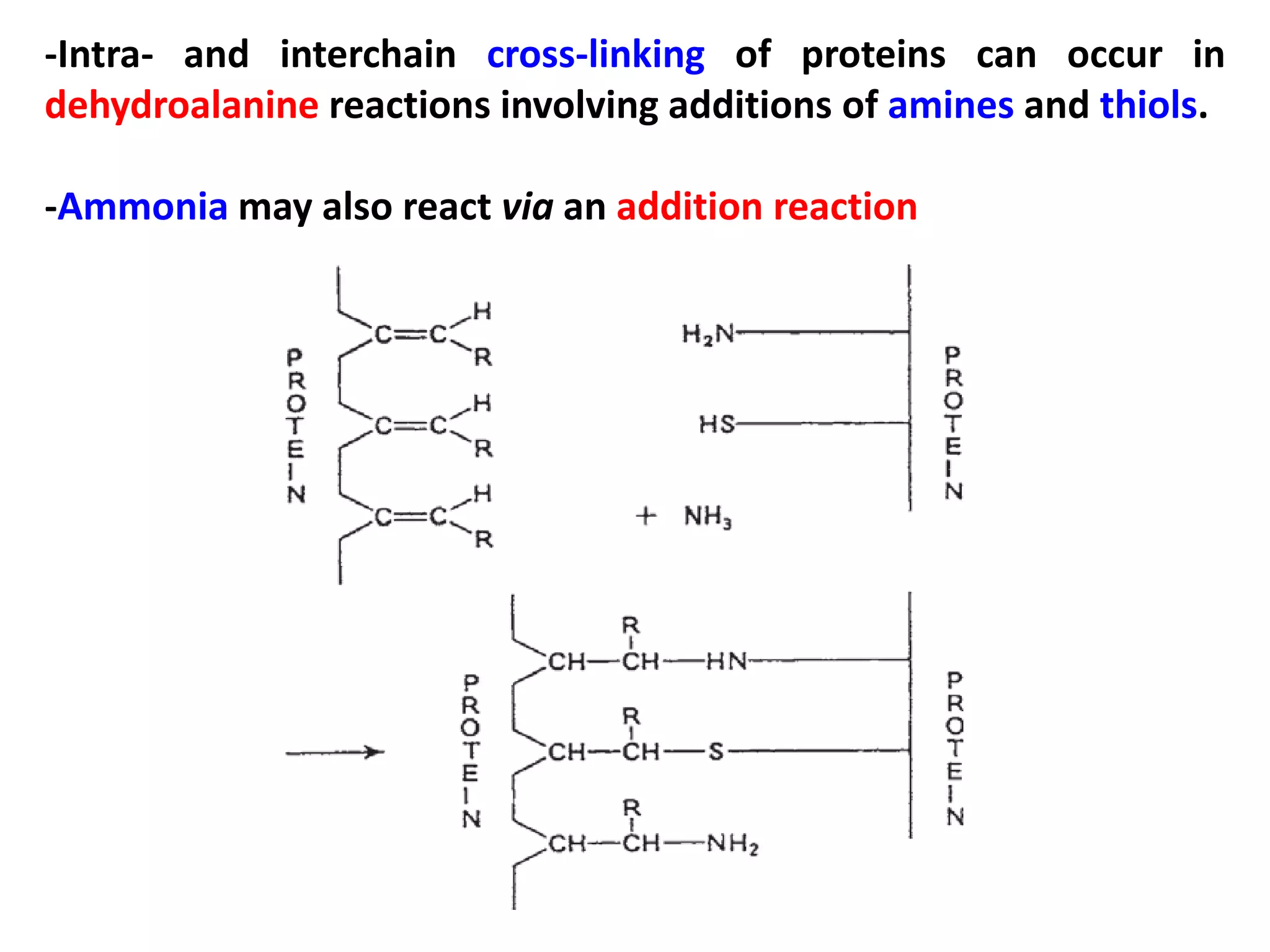
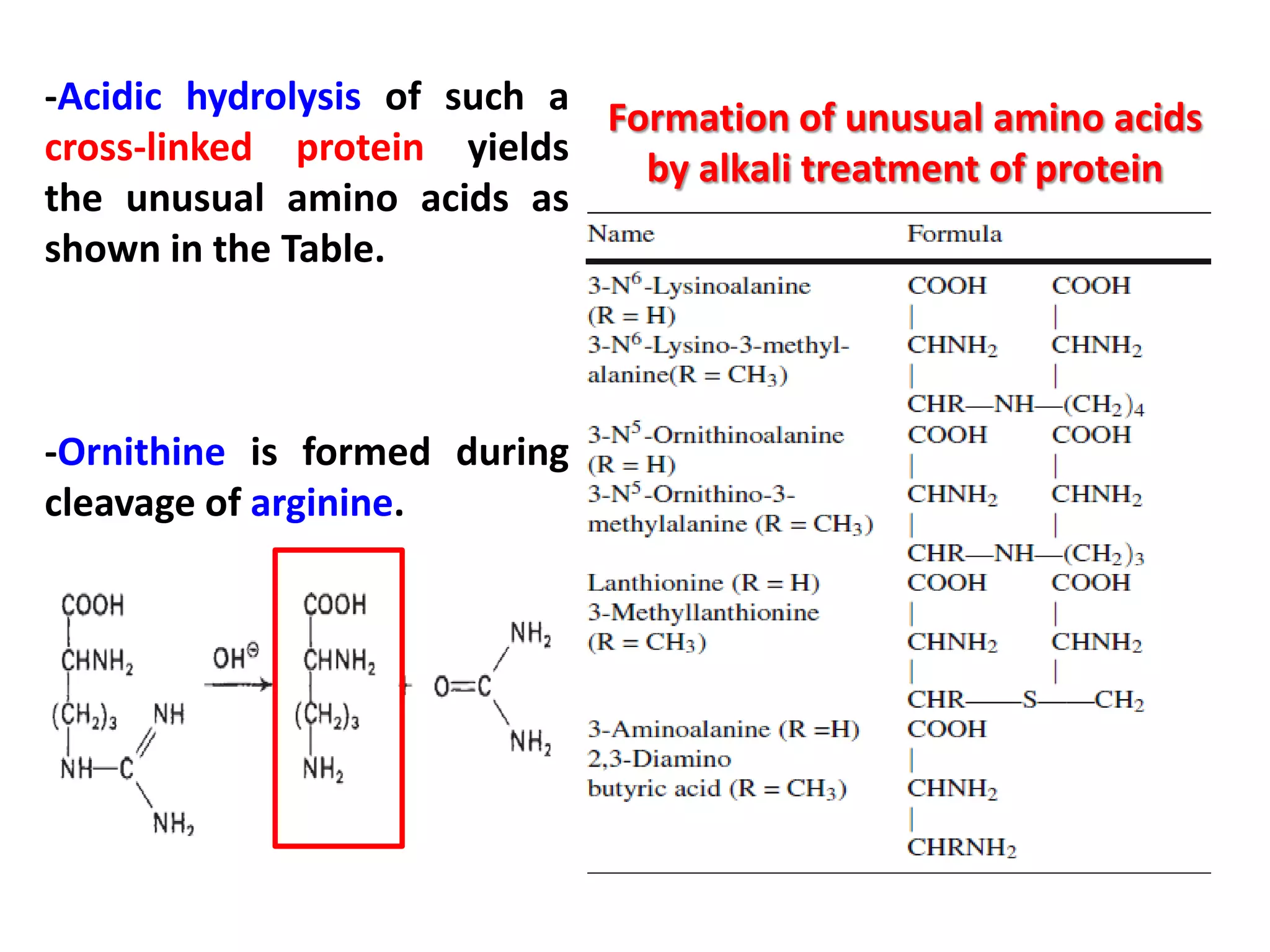
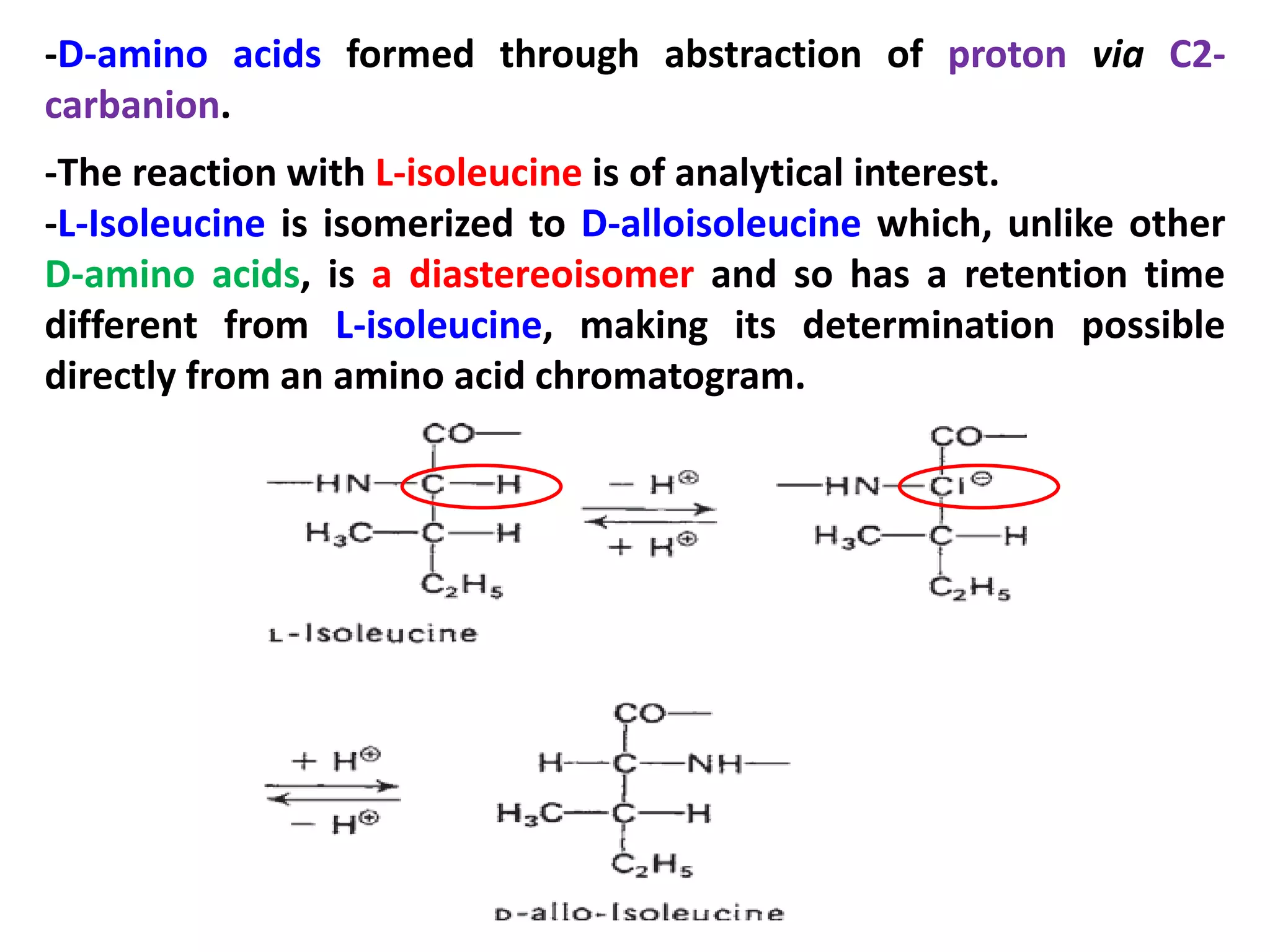
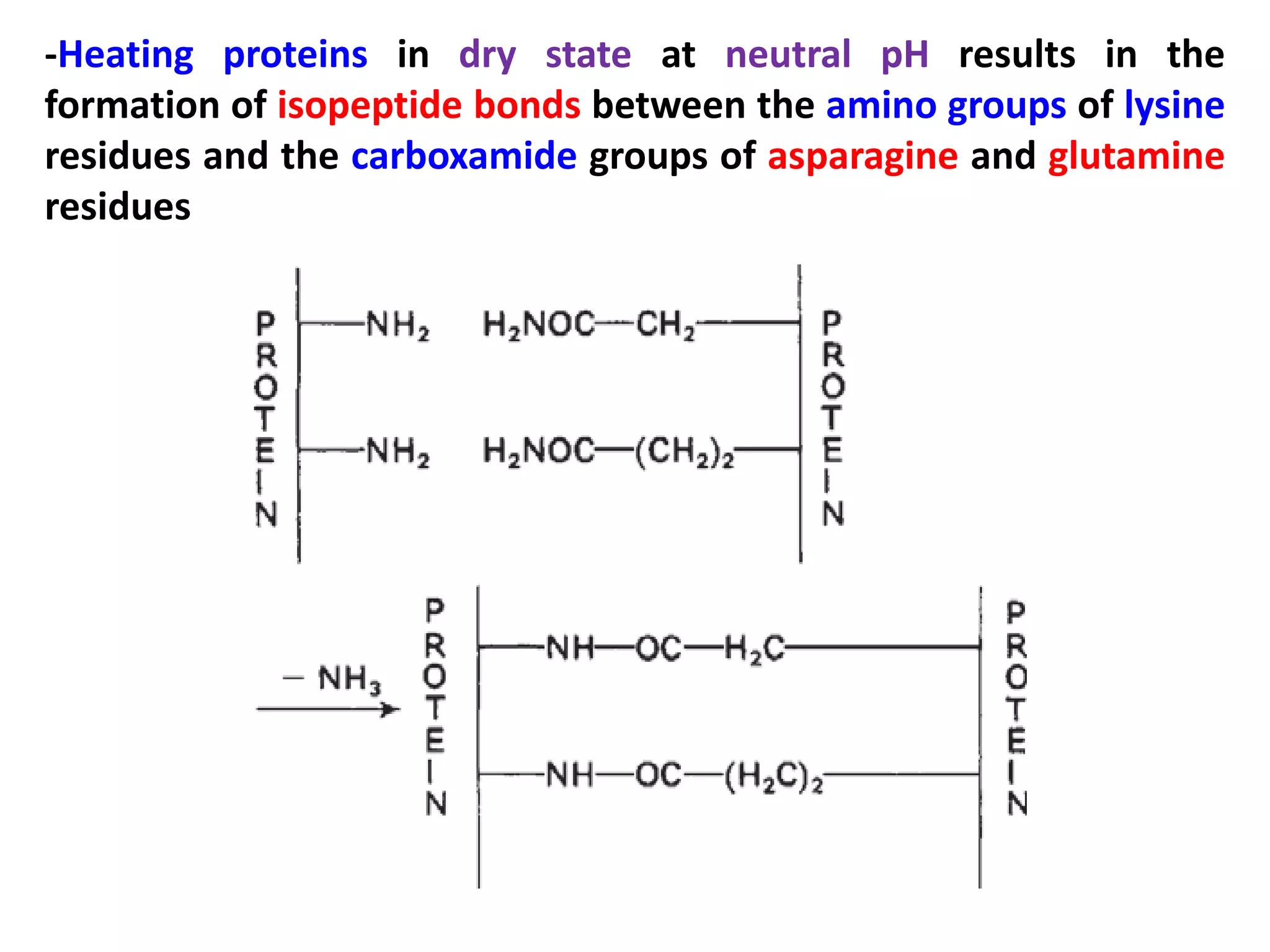
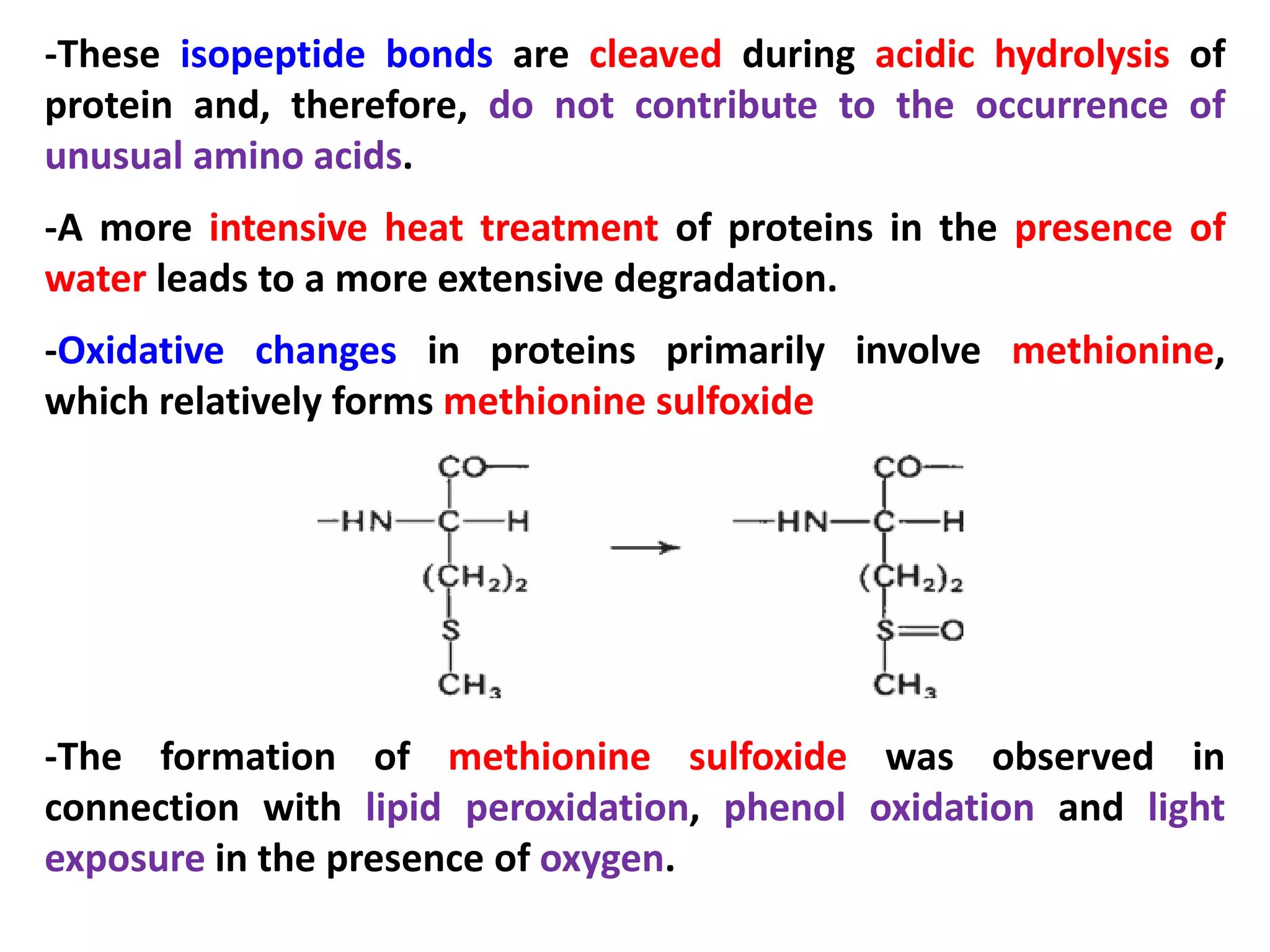
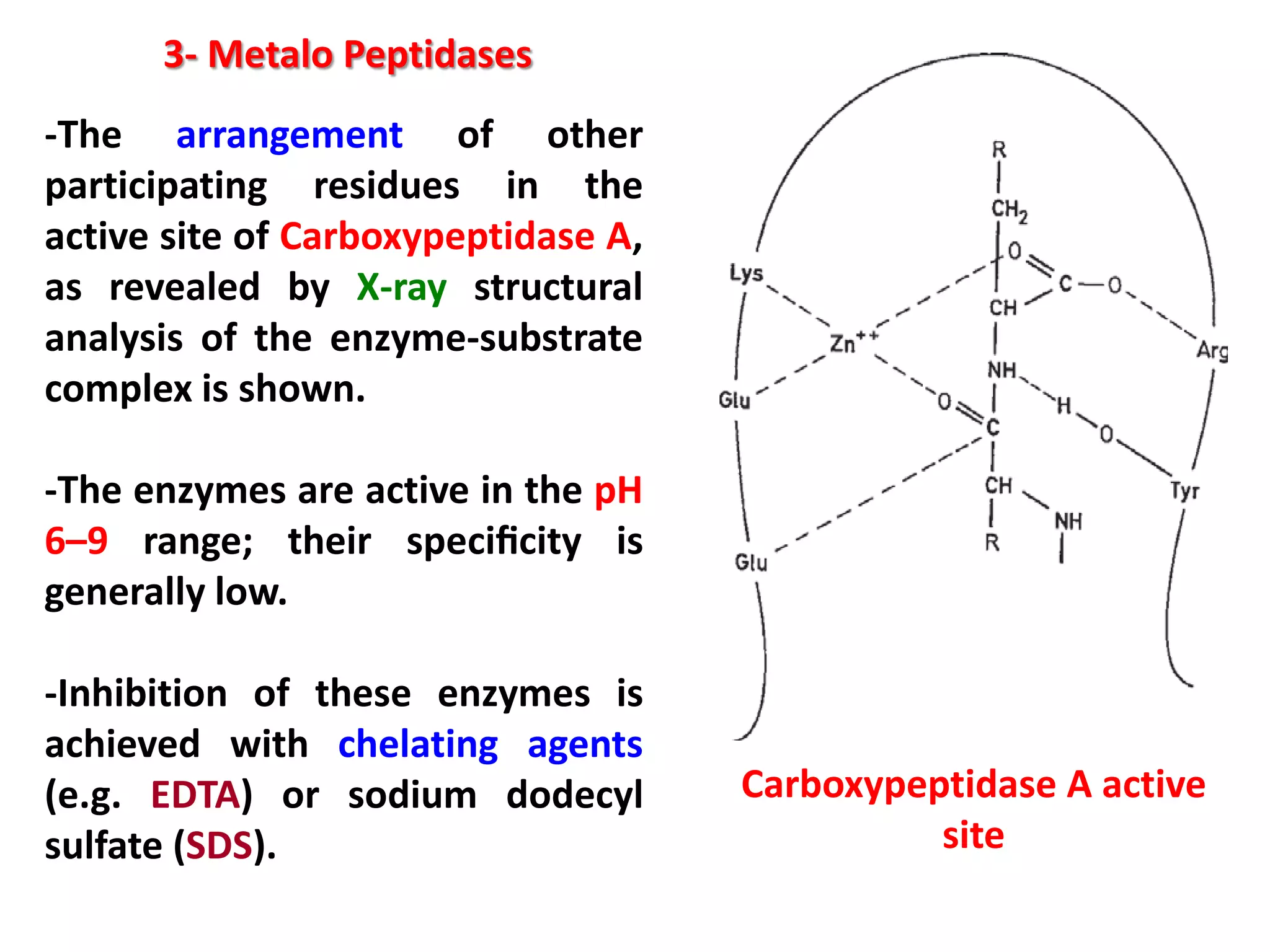
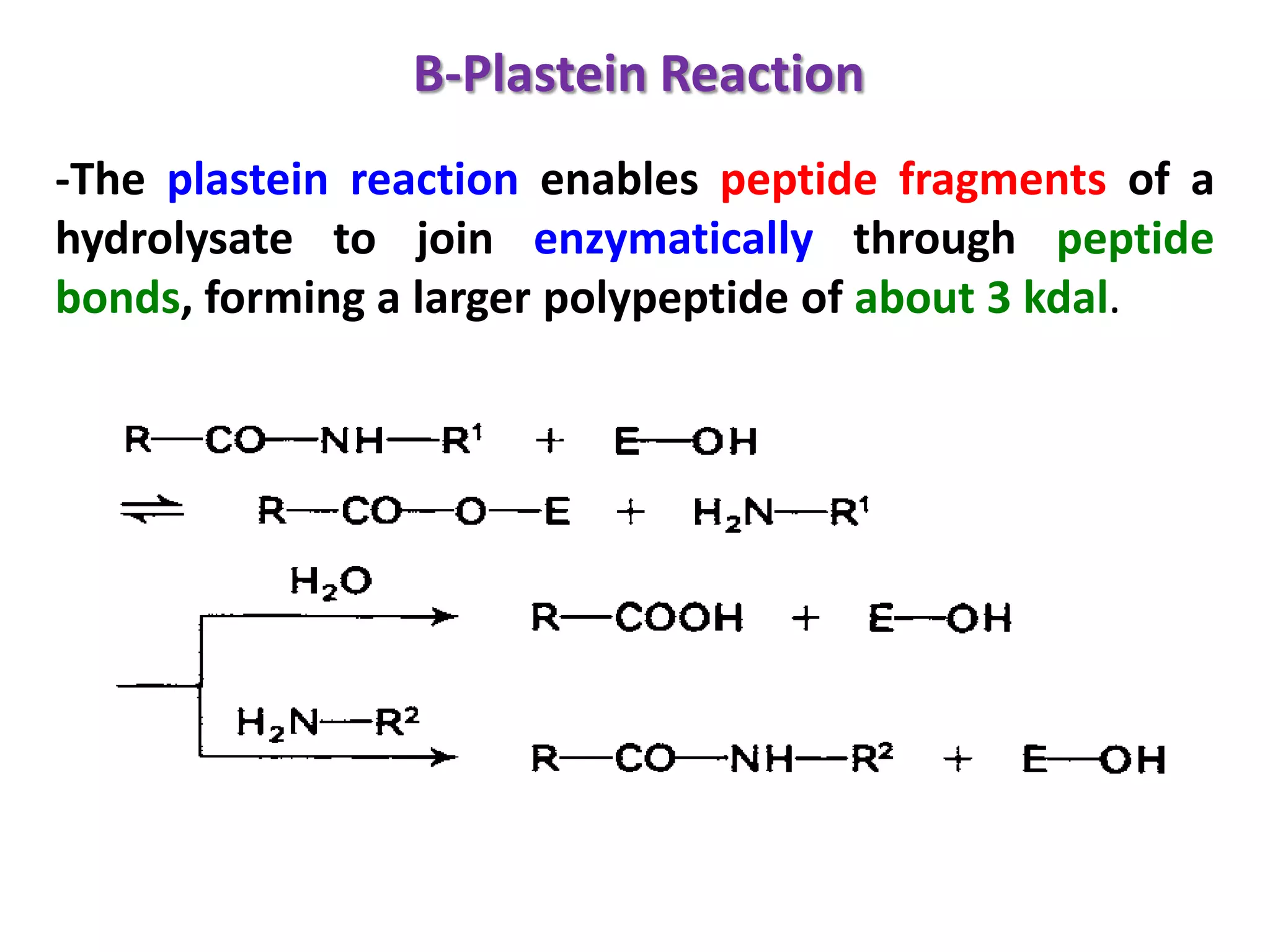
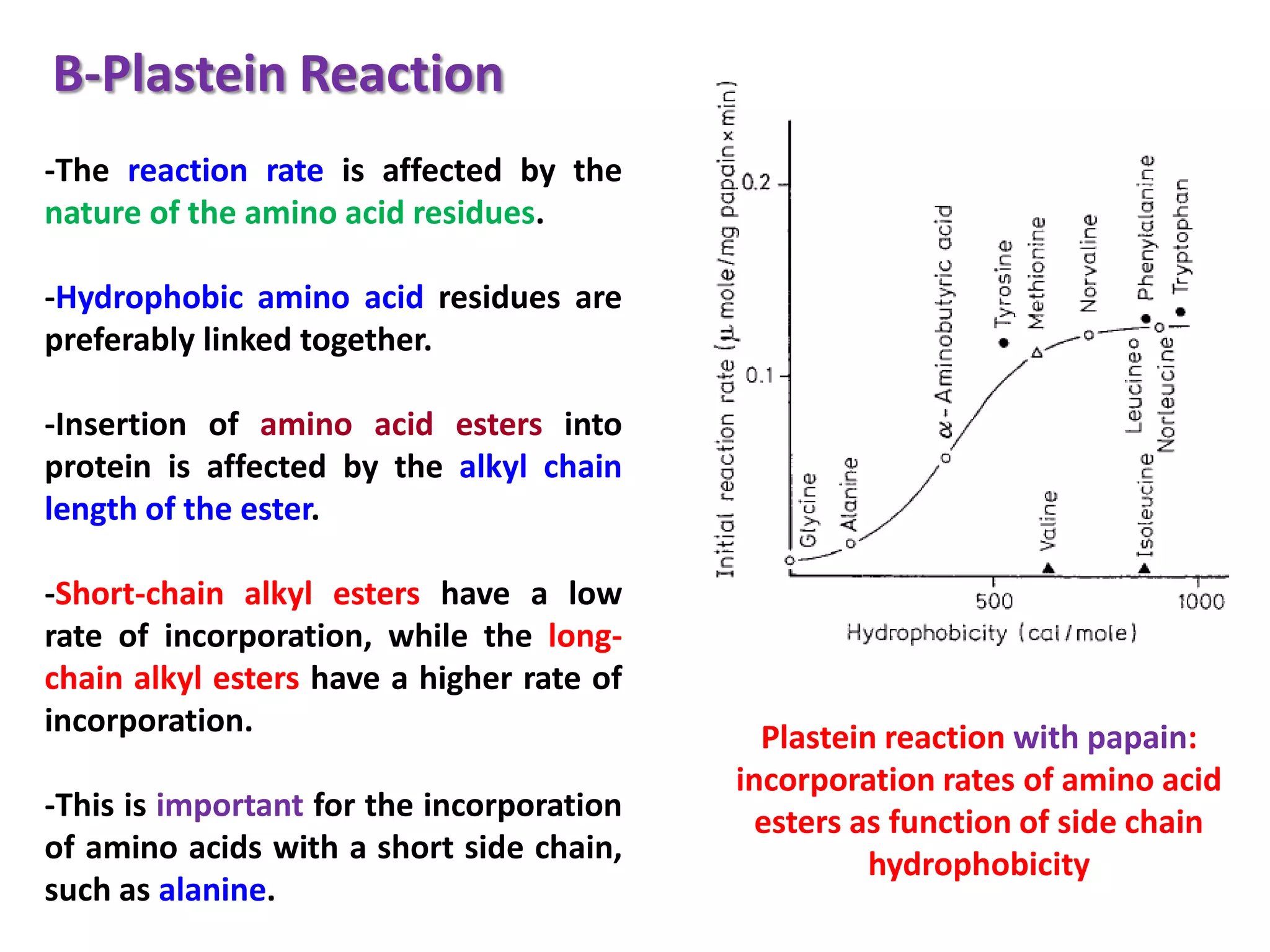
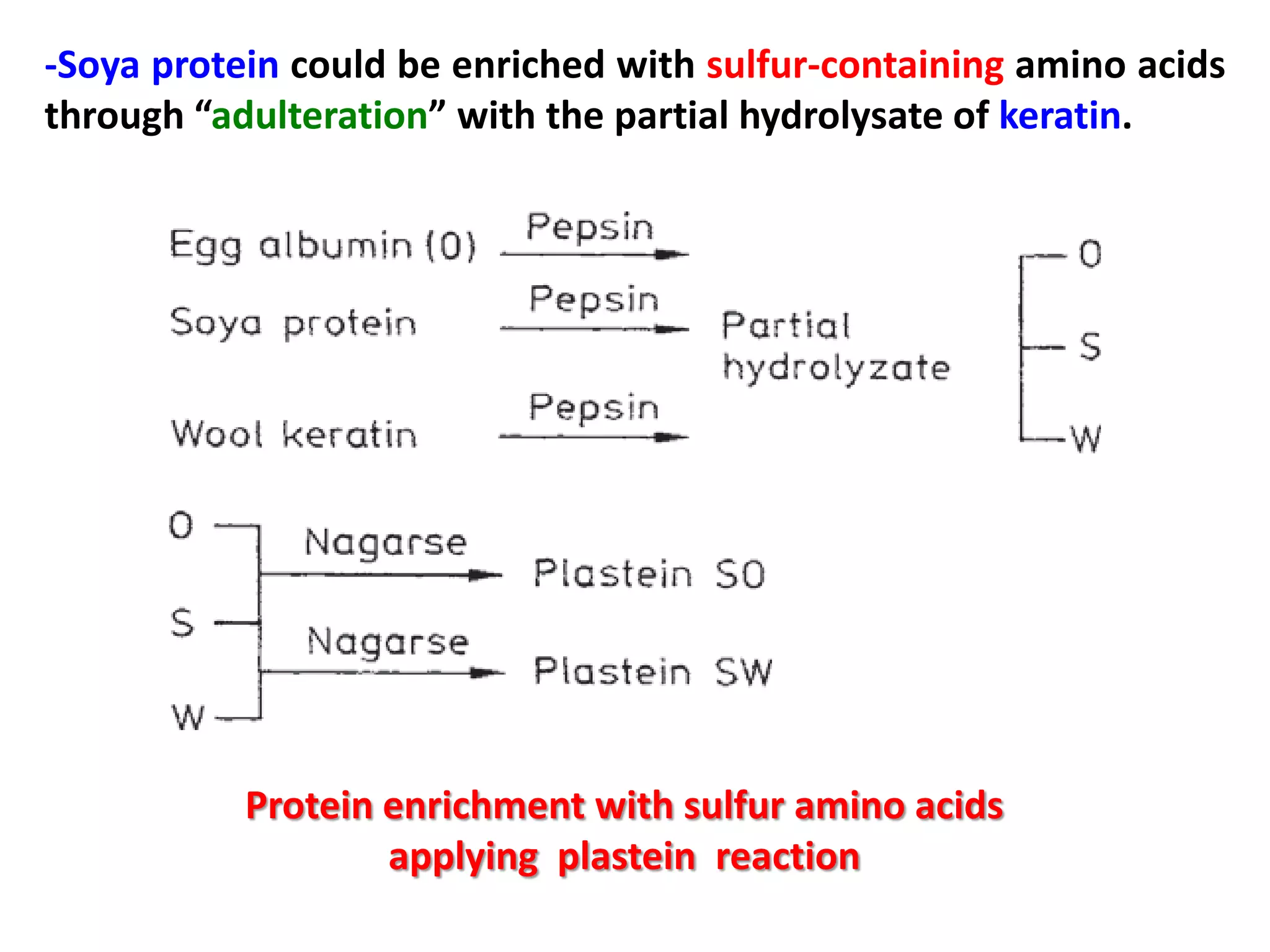
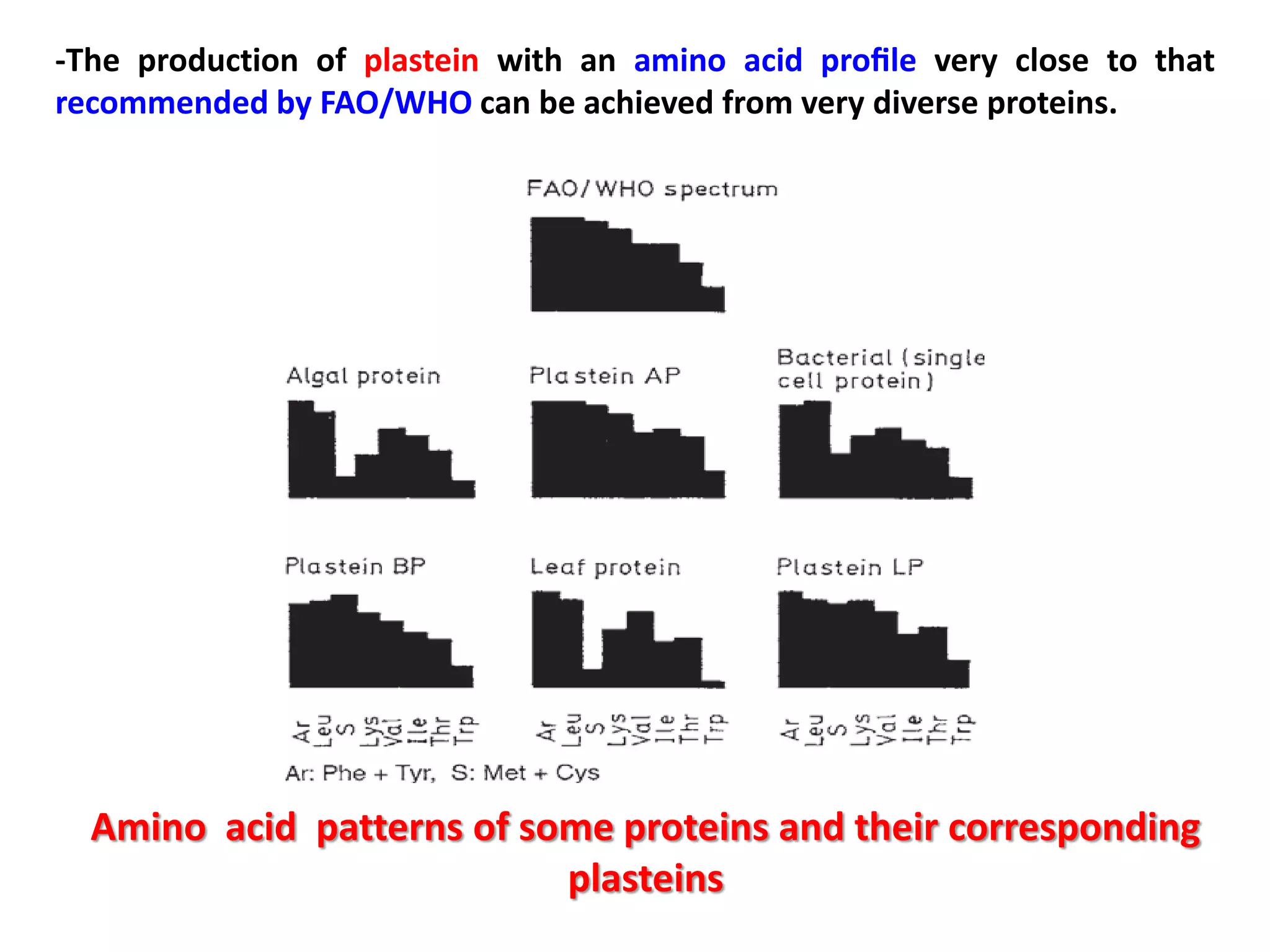
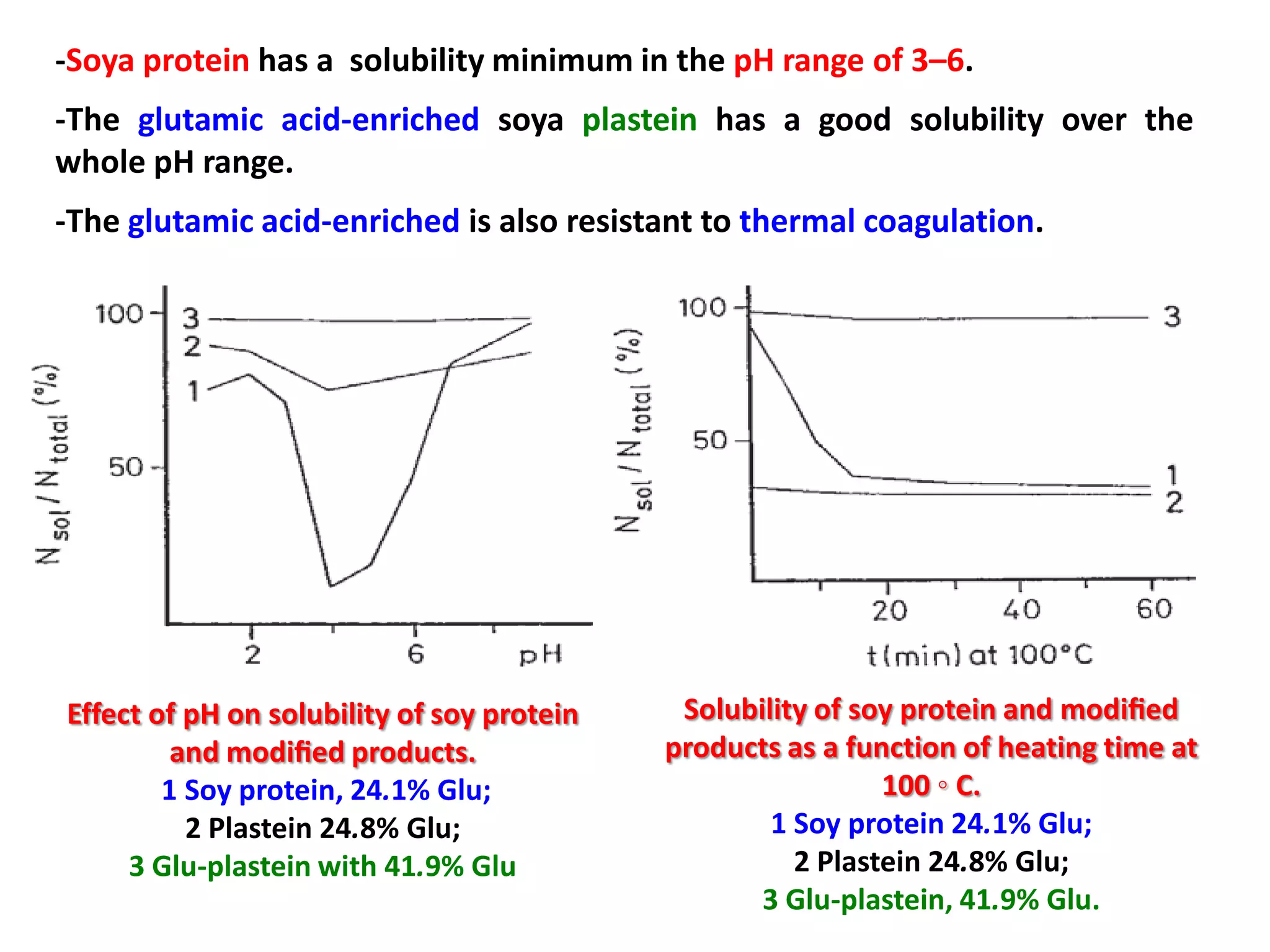
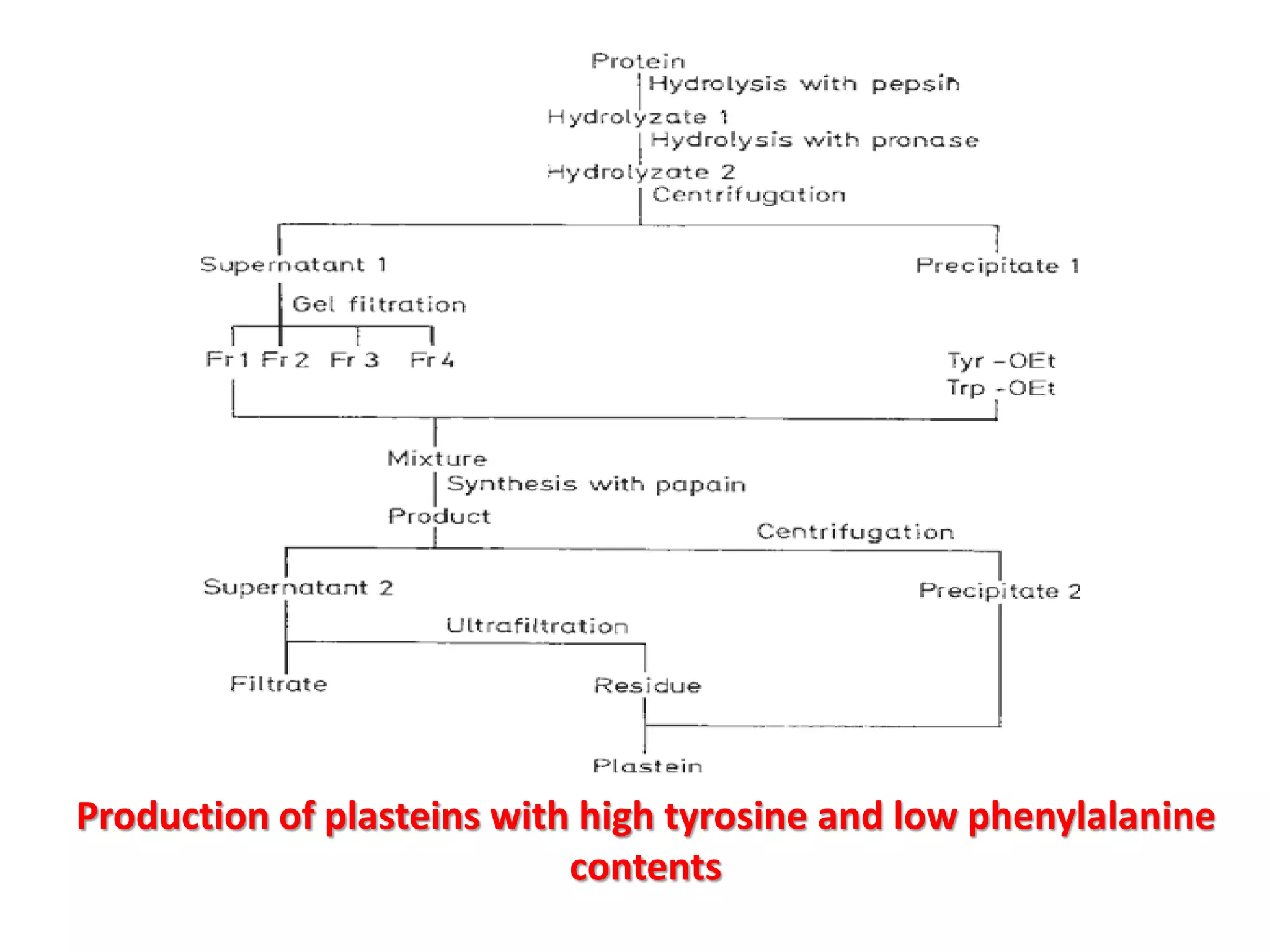
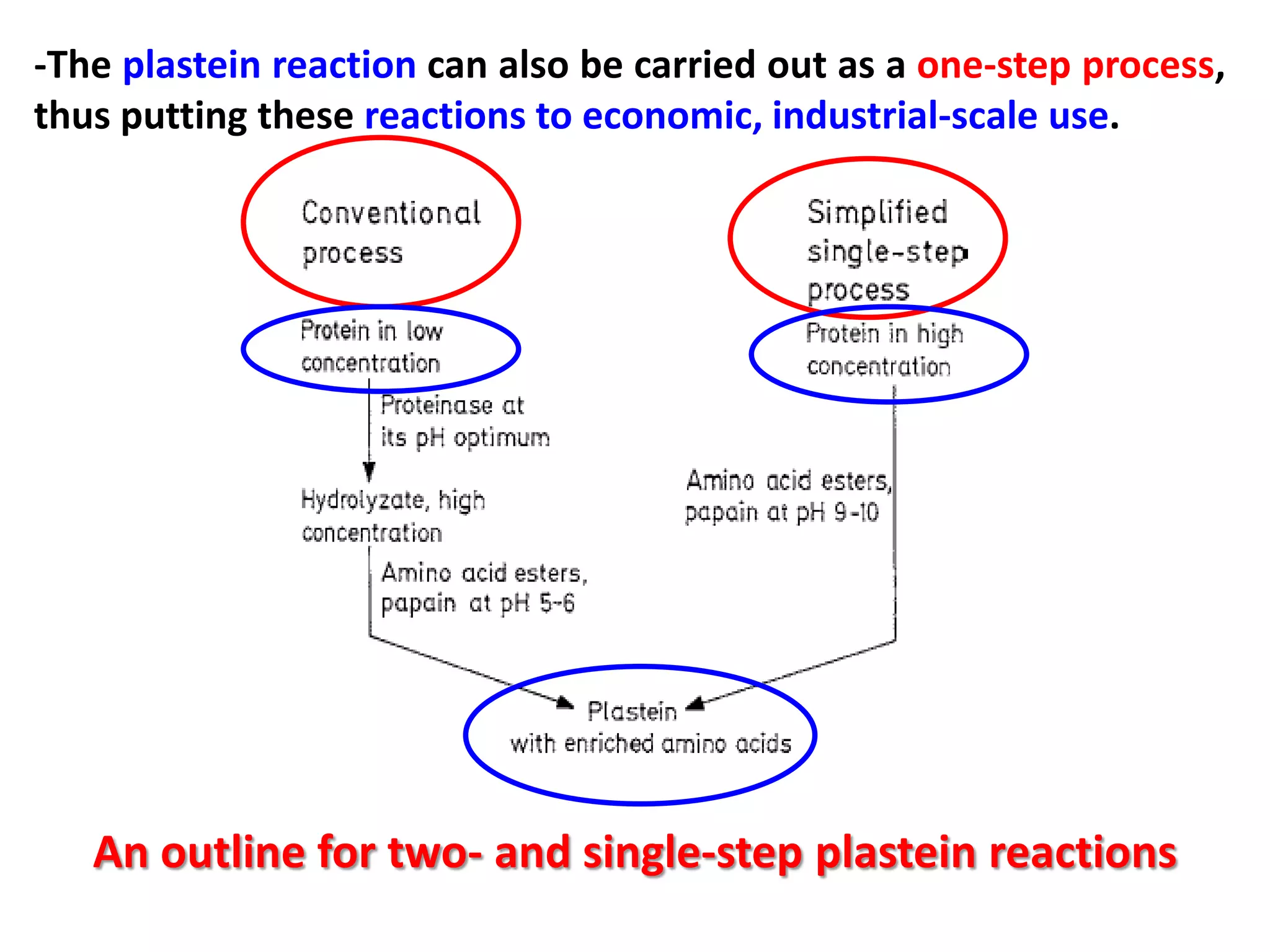
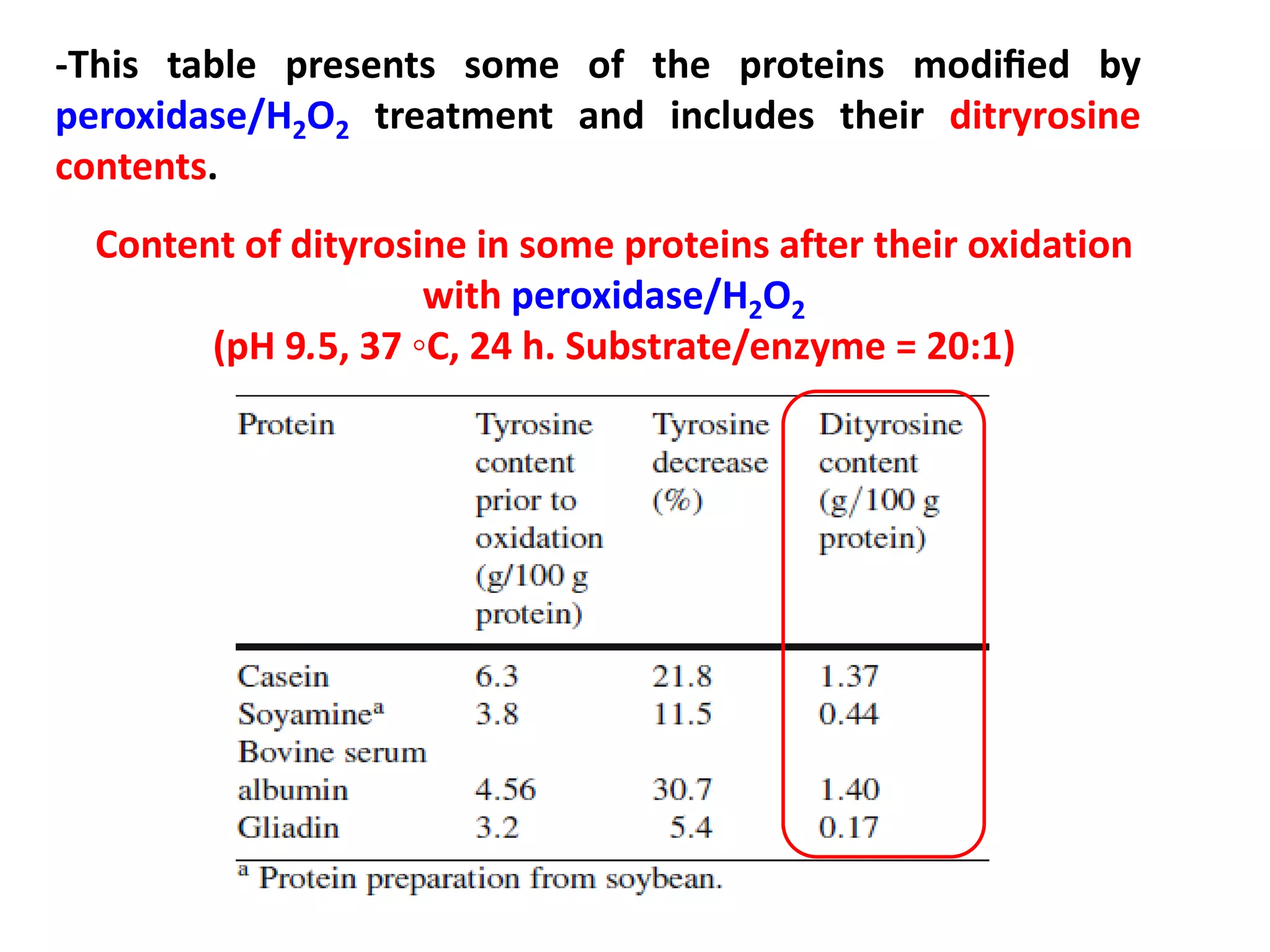
This document discusses reactions of proteins involved in food processing. It describes various enzyme-catalyzed reactions like those involving serine, cysteine, metallo and aspartic endopeptidases. It also discusses chemical and enzymatic modifications of proteins for food processing, including succinylation, reductive methylation, and disulfide bond reduction/reoxidation. The enzymatic plastein reaction is described which joins peptide fragments through peptide bonds. Overall, the document provides an overview of reactions and modifications that can change protein properties for uses in food processing.