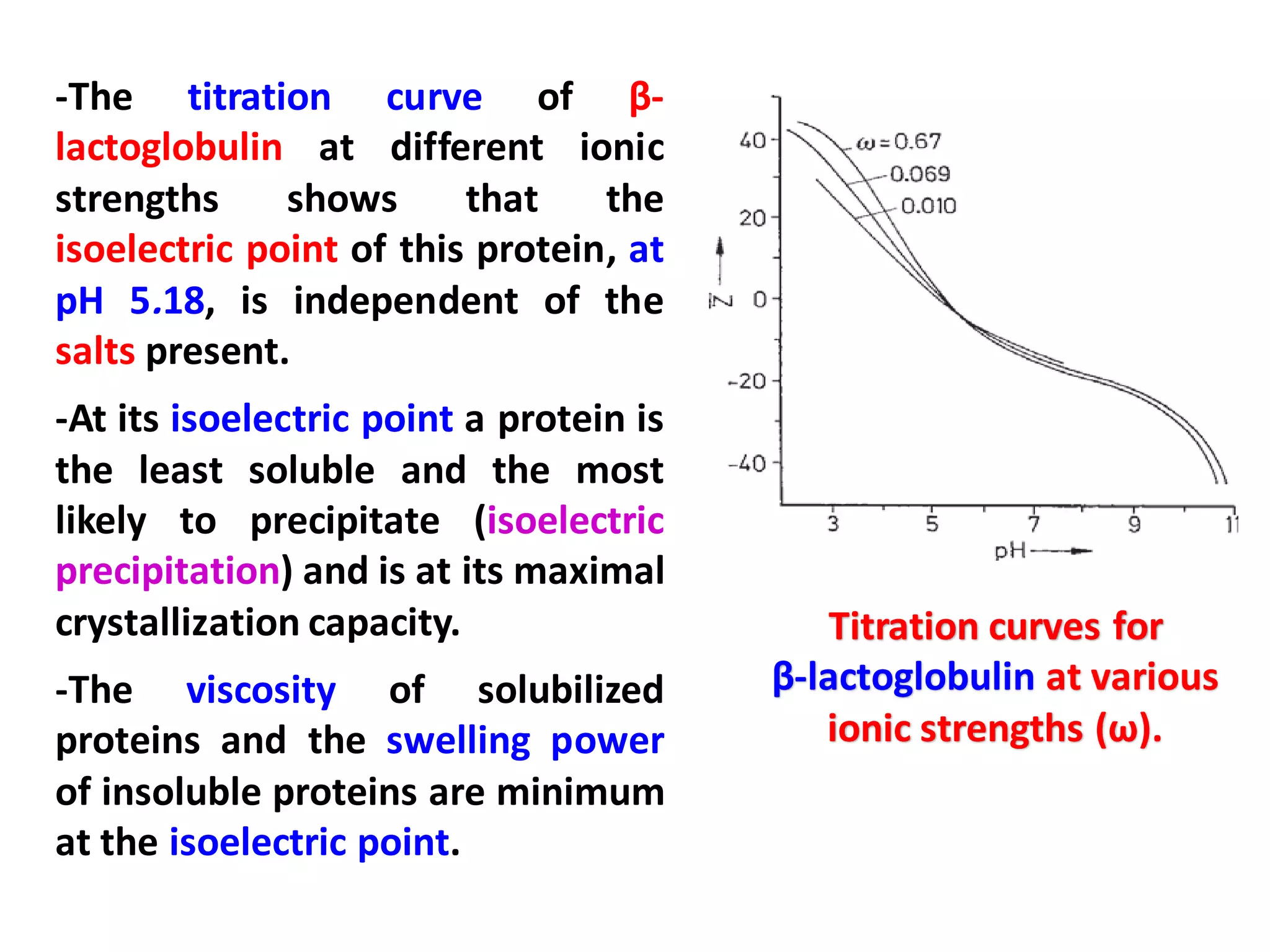
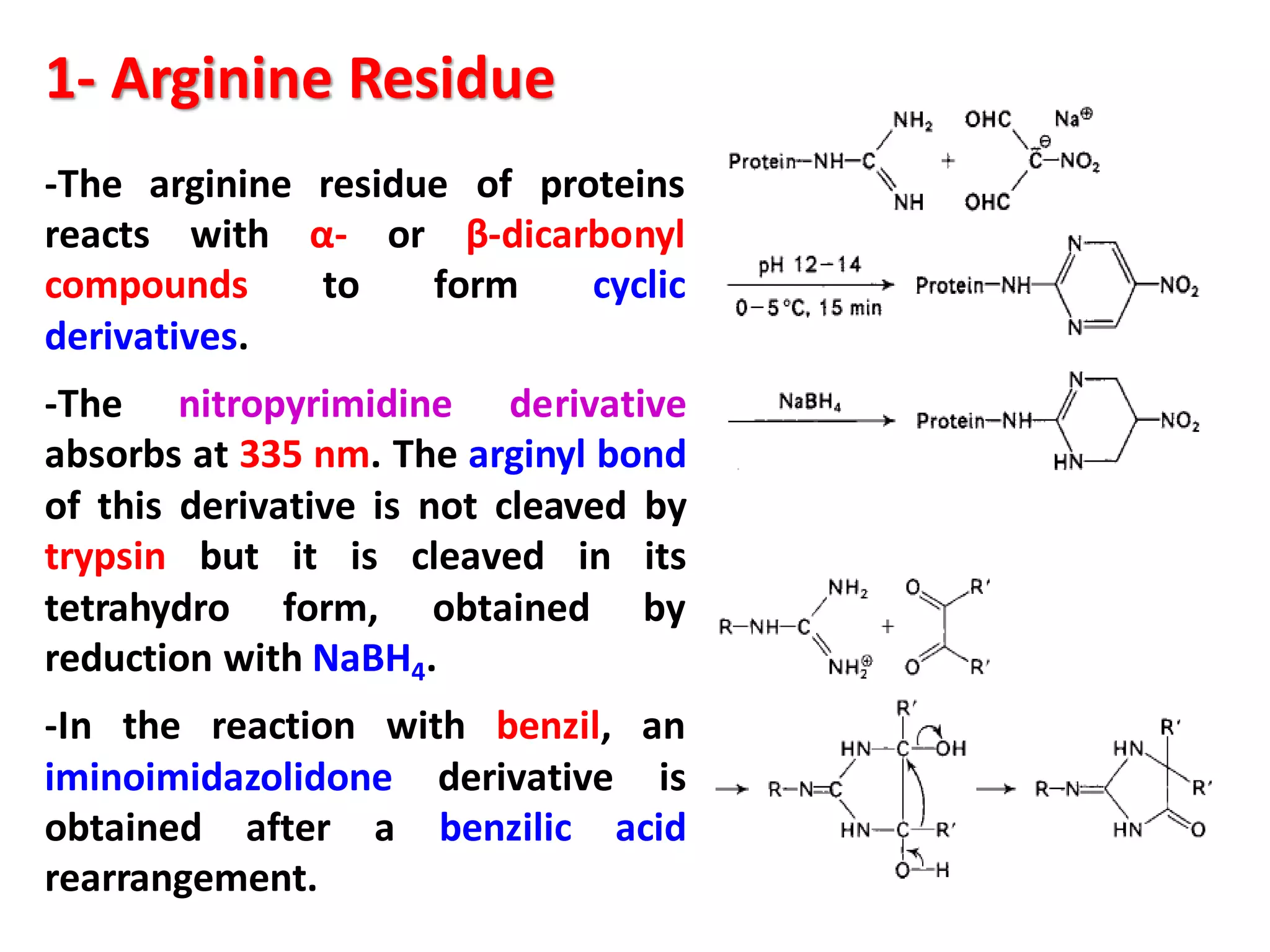
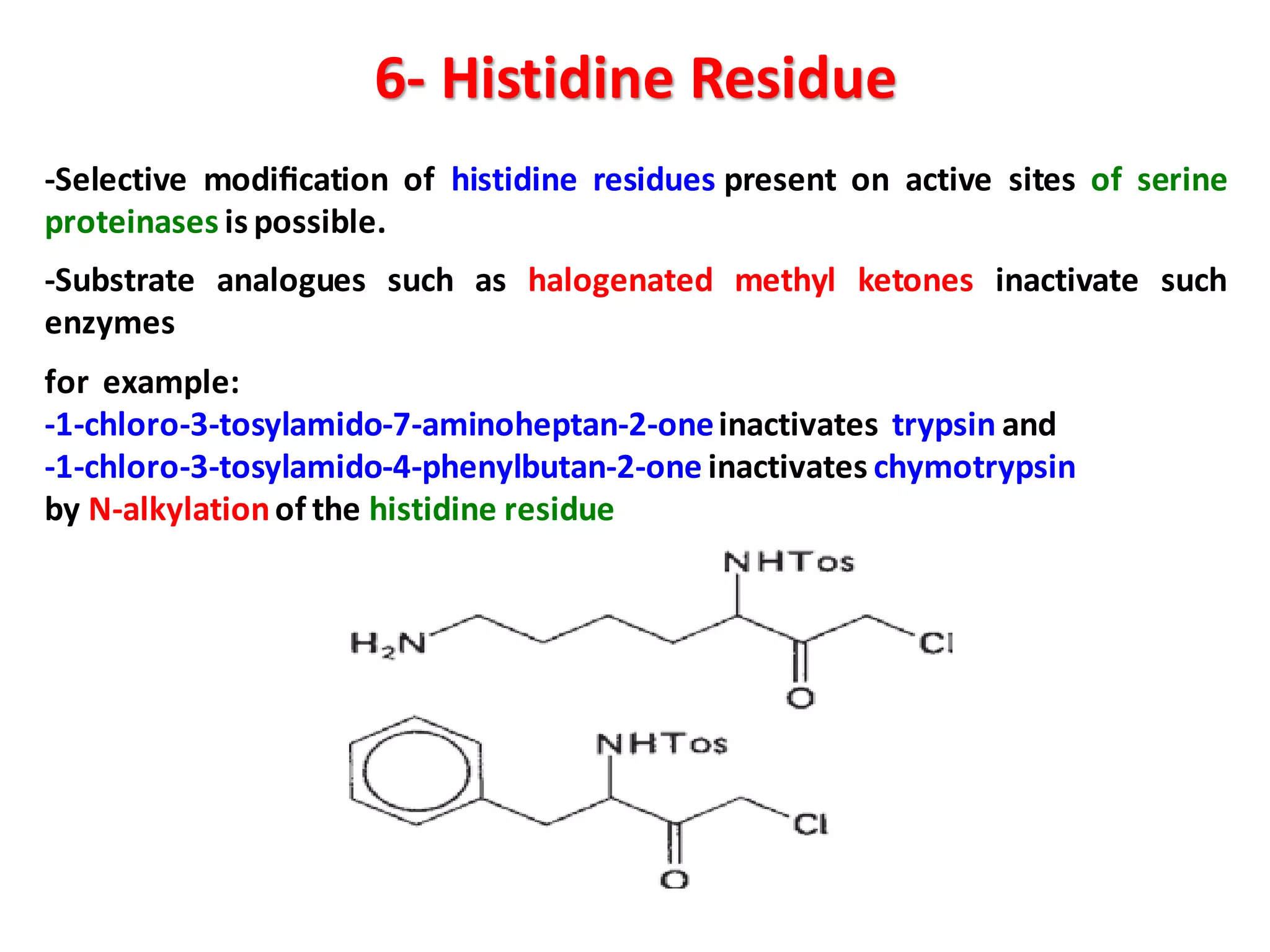
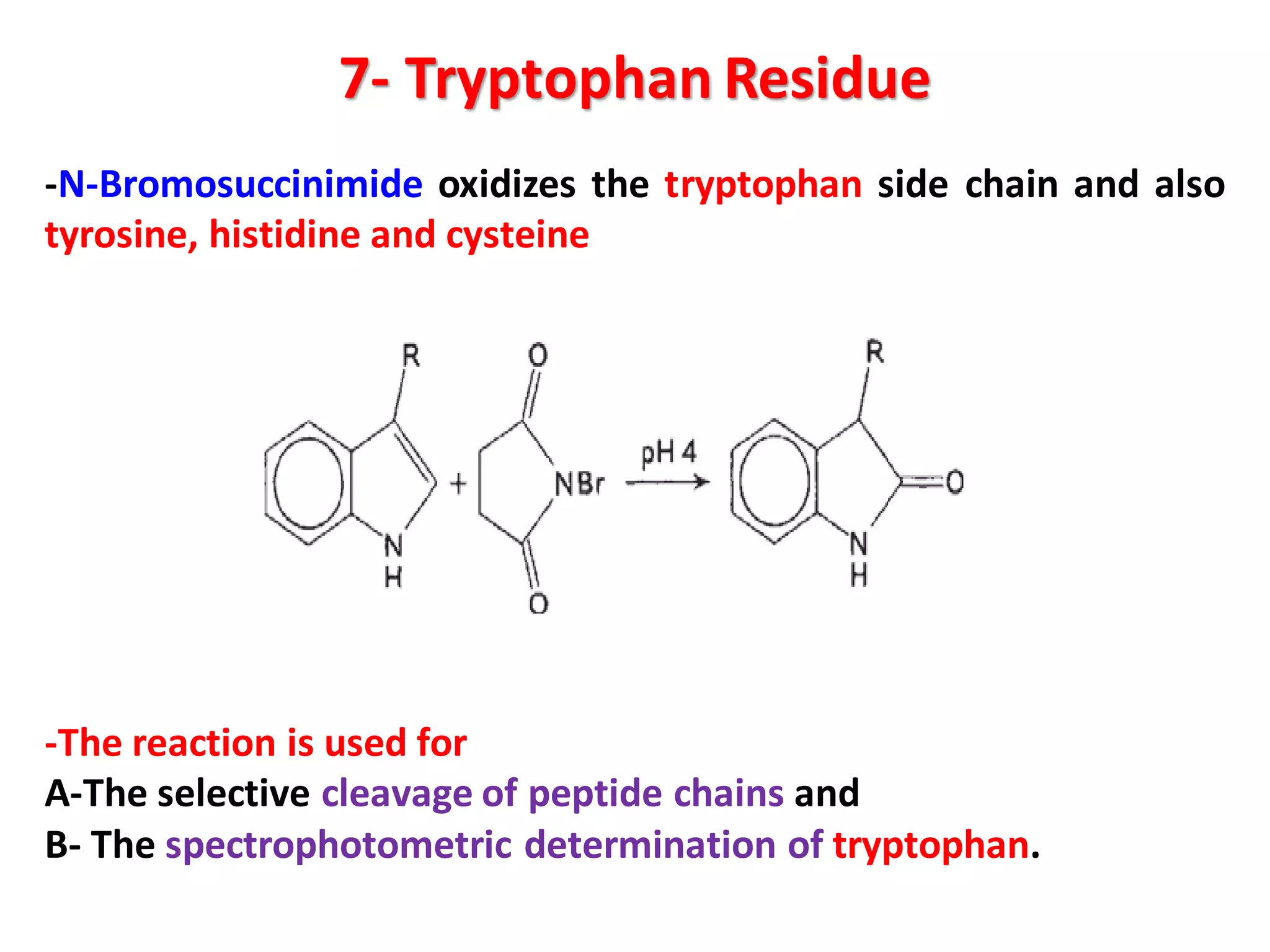
This document discusses the physico-chemical properties of proteins, including their physical and chemical properties. It describes proteins' ability to dissociate depending on pH, their optical activity, solubility which depends on factors like pH and salt concentration, and their ability to form foams and stabilize foams through adsorption at gas-liquid interfaces. It also discusses proteins' hydration, swelling power in water, and ability to form gels through polymeric networks or aggregate dispersions.