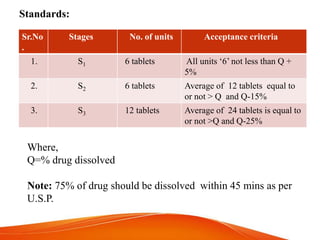
Quality control tests are important to ensure tablets meet standards for safety, efficacy and patient acceptability. Key tests include weight variation, hardness, friability, disintegration and dissolution. Weight variation tests if individual tablet weights match the average weight. Hardness ensures tablets can withstand manufacturing and handling stresses. Friability tests surface strength and disintegration confirms how quickly tablets break down in fluid. Dissolution determines the rate of drug release.







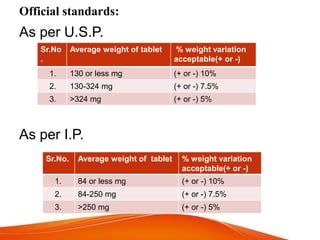

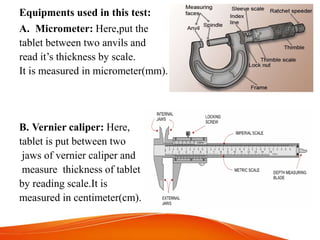








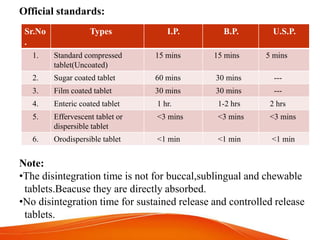

![Equipments used in this test:
[1] Rotating basket apparatus:
Assembly consists of:
• A covered vessel
• Metallic drive shaft
• Cylindrical basket
• Motor
• Sample port
• Water bath holding temperature
37◦C ± 0.5◦C
• Acidic media or alkaline media](https://image.slidesharecdn.com/qcoftablet-190211144802/85/Quality-control-tests-of-tablet-21-320.jpg)
![[2]Rotating paddle apparatus:
Assembly consists of:
• A covered vessel
• Metallic drive shaft
• Stainless steel paddle
• Motor
• Sinker to prevent floating of
tablet
• Water bath holding tempe
-rature at 37◦C ± 0.5◦C
• Acidic media or alkaline
media](https://image.slidesharecdn.com/qcoftablet-190211144802/85/Quality-control-tests-of-tablet-22-320.jpg)