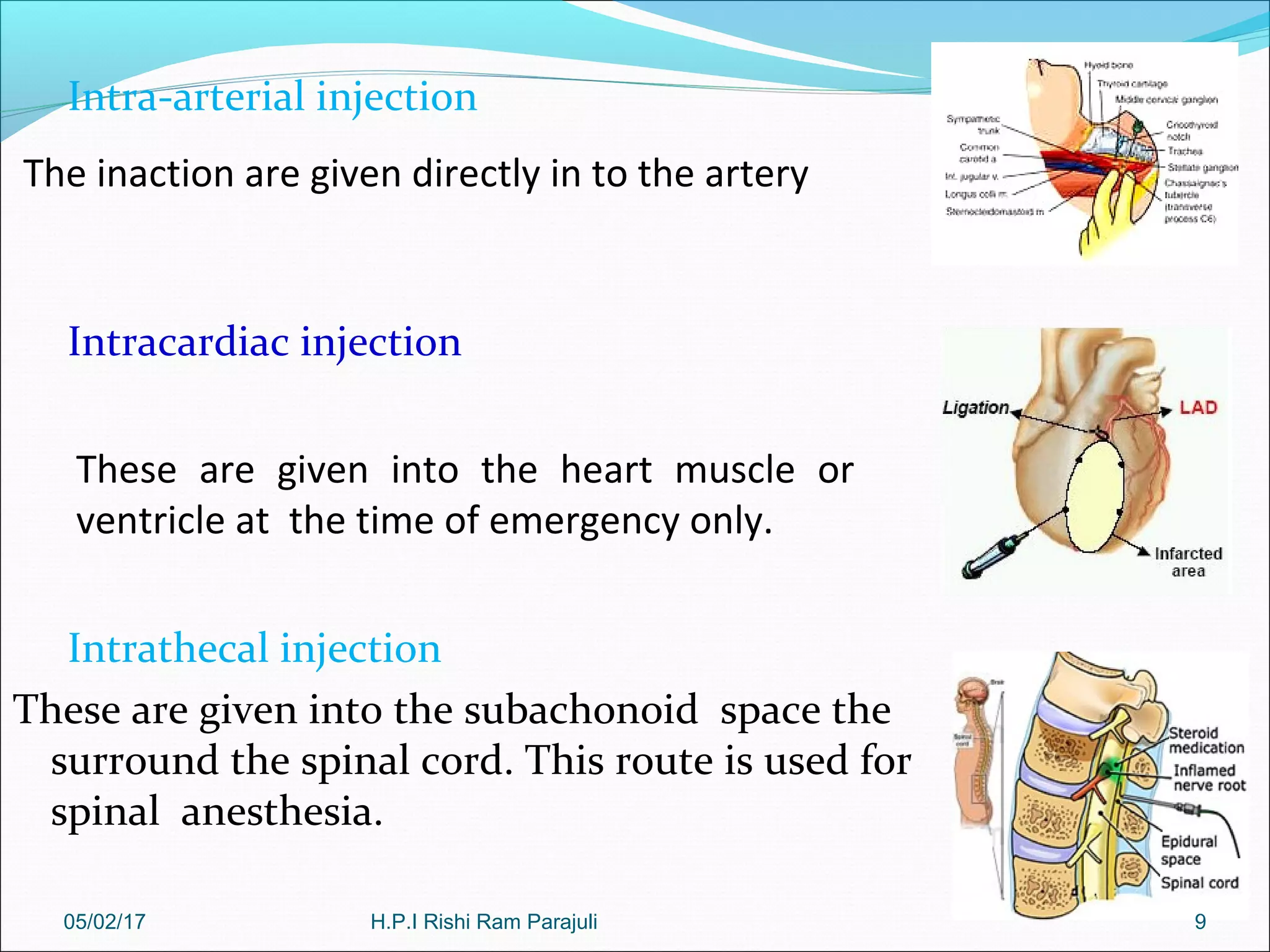
The document discusses parenteral products and their administration. It defines parenteral as referring to administration by injection rather than orally, bypassing the gastrointestinal tract. It then discusses the advantages and disadvantages of the parenteral route, including faster systemic delivery but also risks of infection. The document outlines various routes of parenteral injection and provides details on procedures like subcutaneous, intramuscular, and intravenous injection. It also discusses formulation, processing, and quality testing of parenteral products.