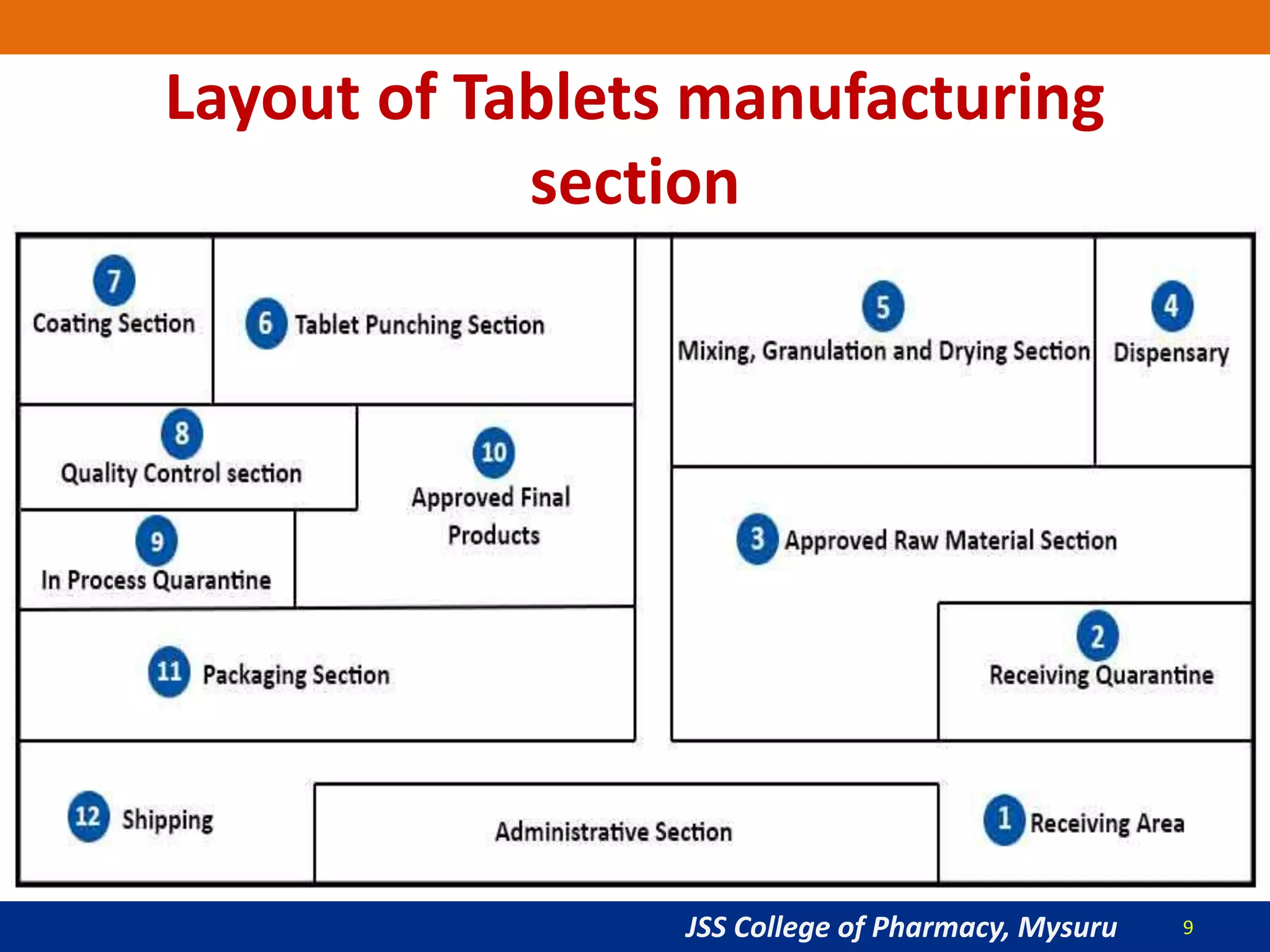
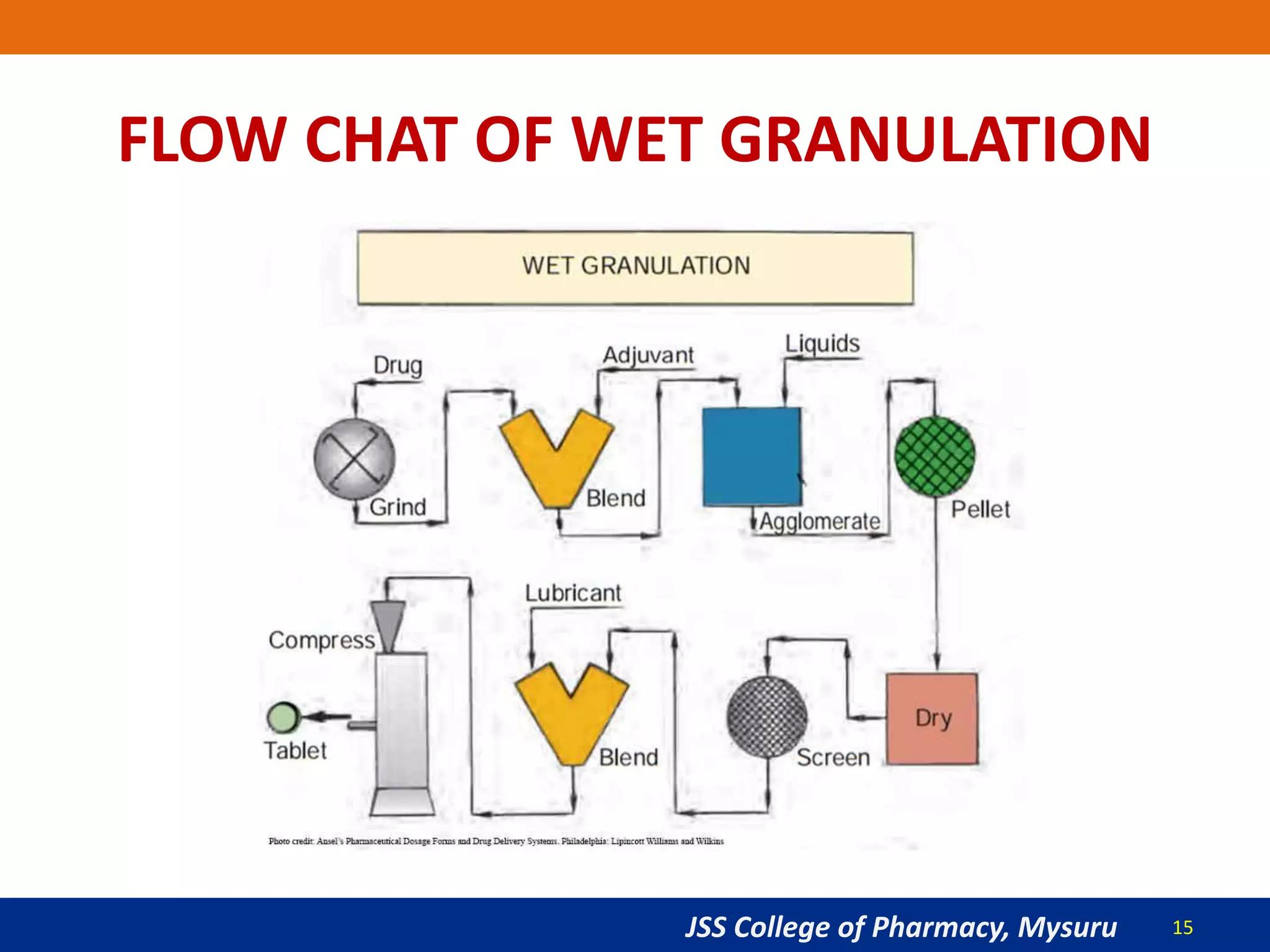
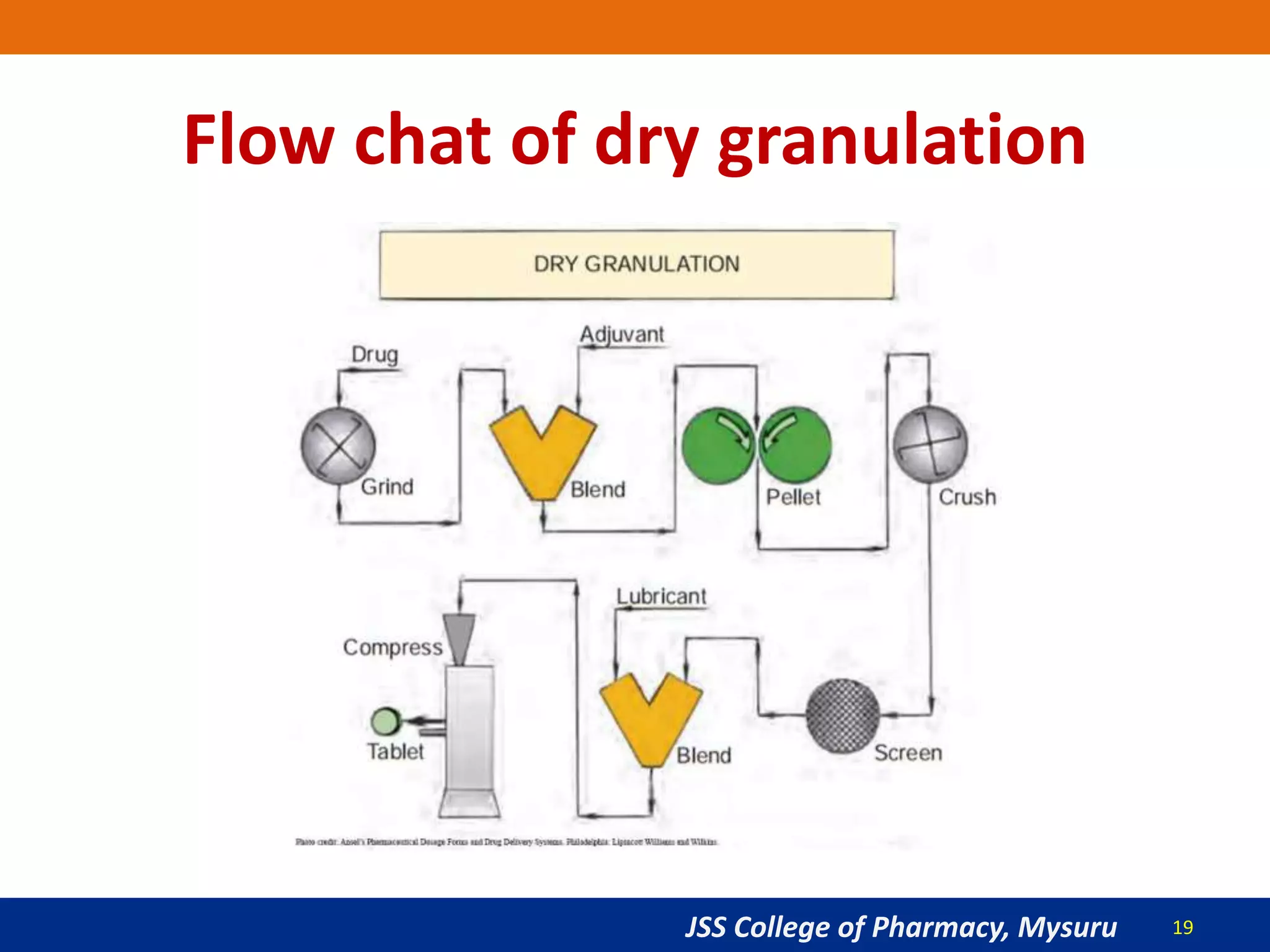
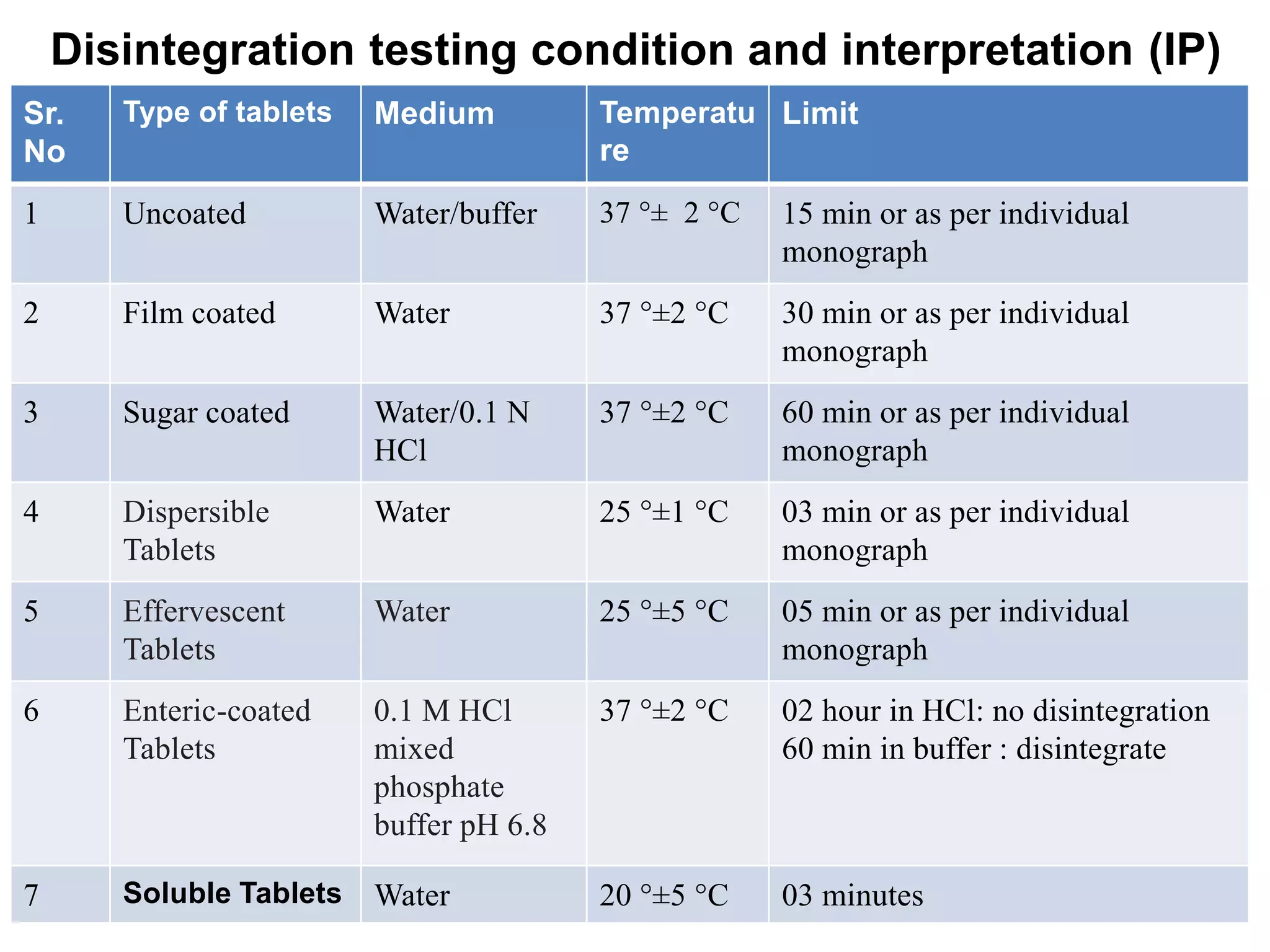
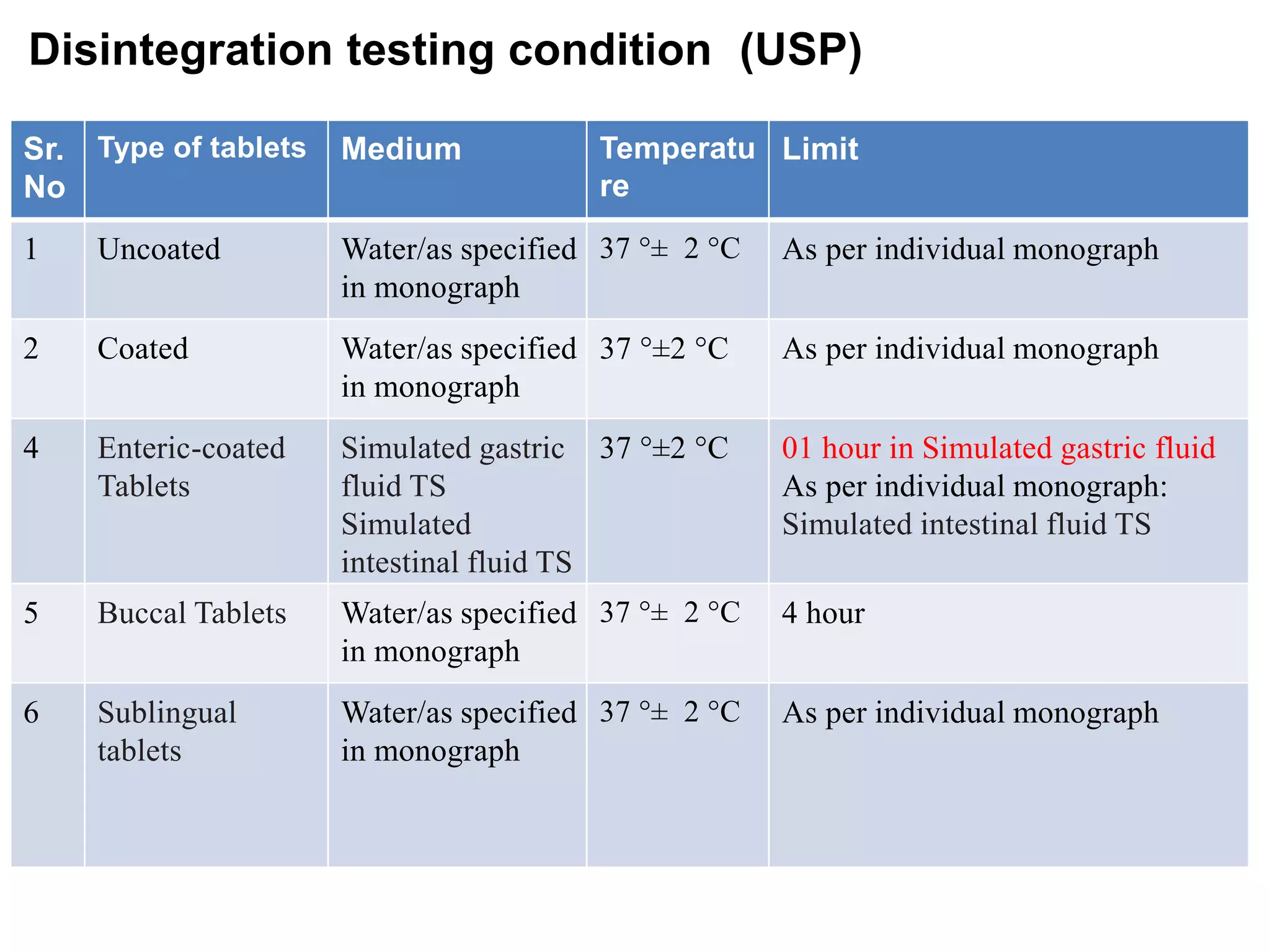
The document summarizes the key steps in the tablet manufacturing process, including weighing, milling, mixing, granulation, drying, compression, coating, and packaging. It describes the main methods of tablet production as wet granulation, dry granulation, and direct compression. Quality control tests mentioned are content uniformity, disintegration testing, and dissolution testing. Personnel required are production pharmacists, manufacturing chemists, analytical chemists, and machine operators. Common equipment used includes mills, mixers, granulators, dryers, tablet presses, and coating pans.