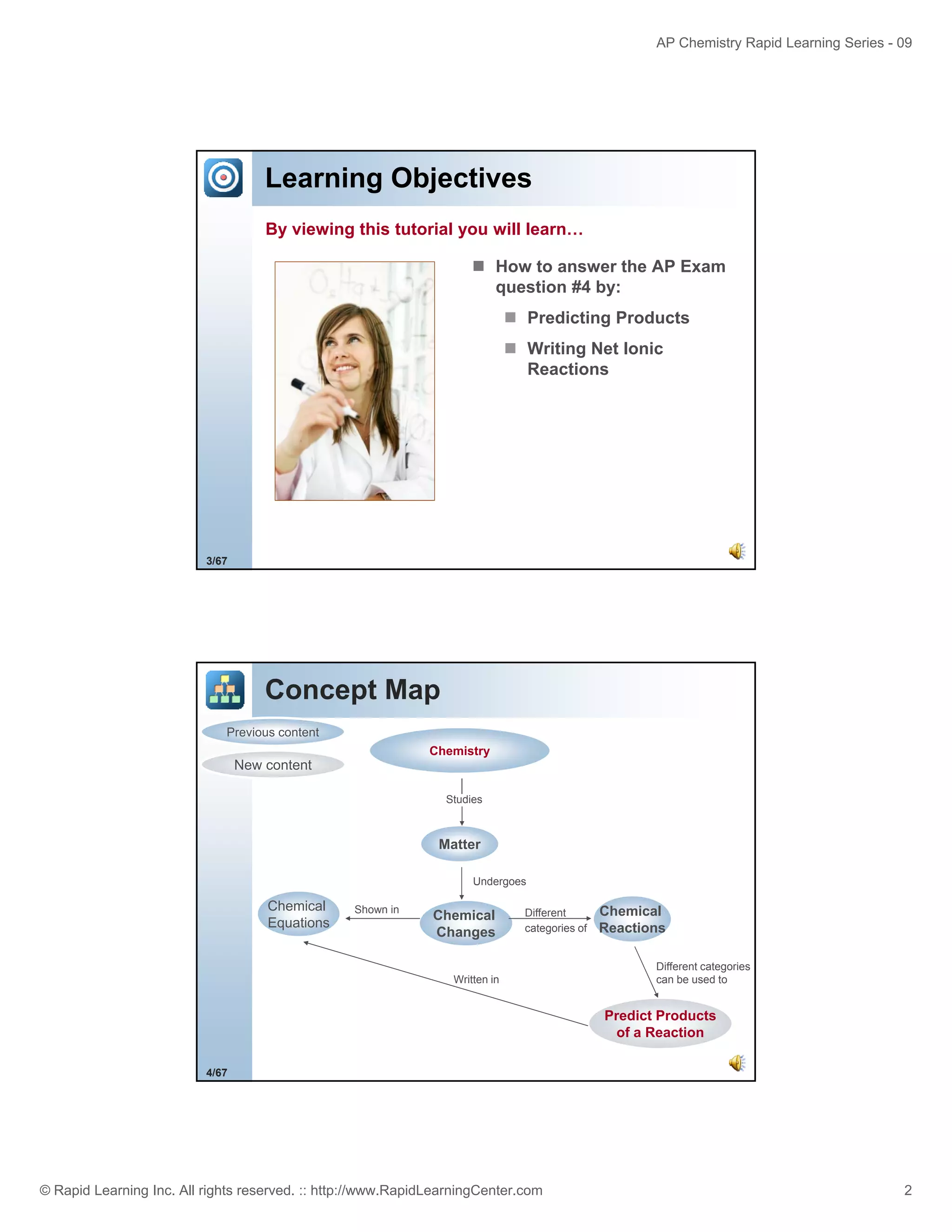
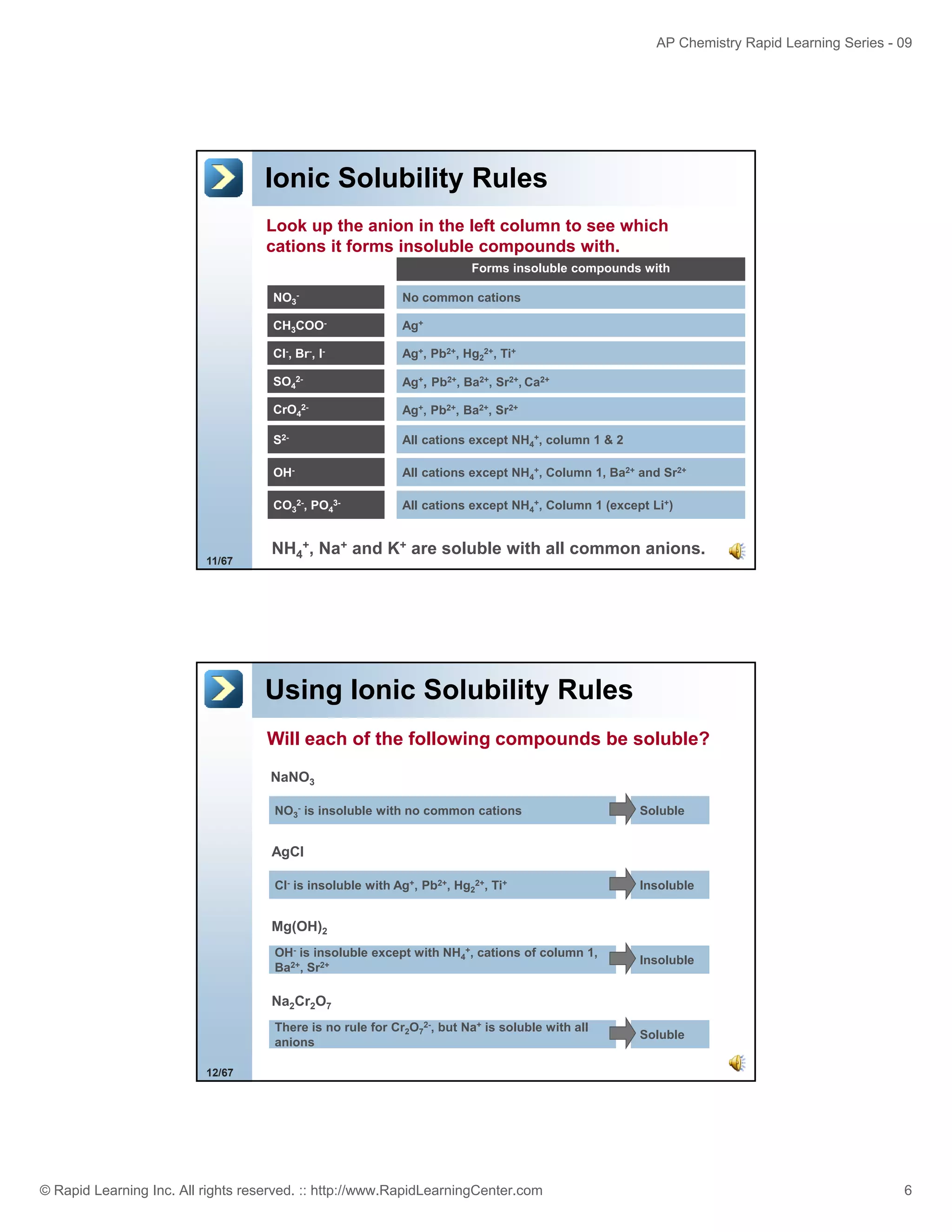
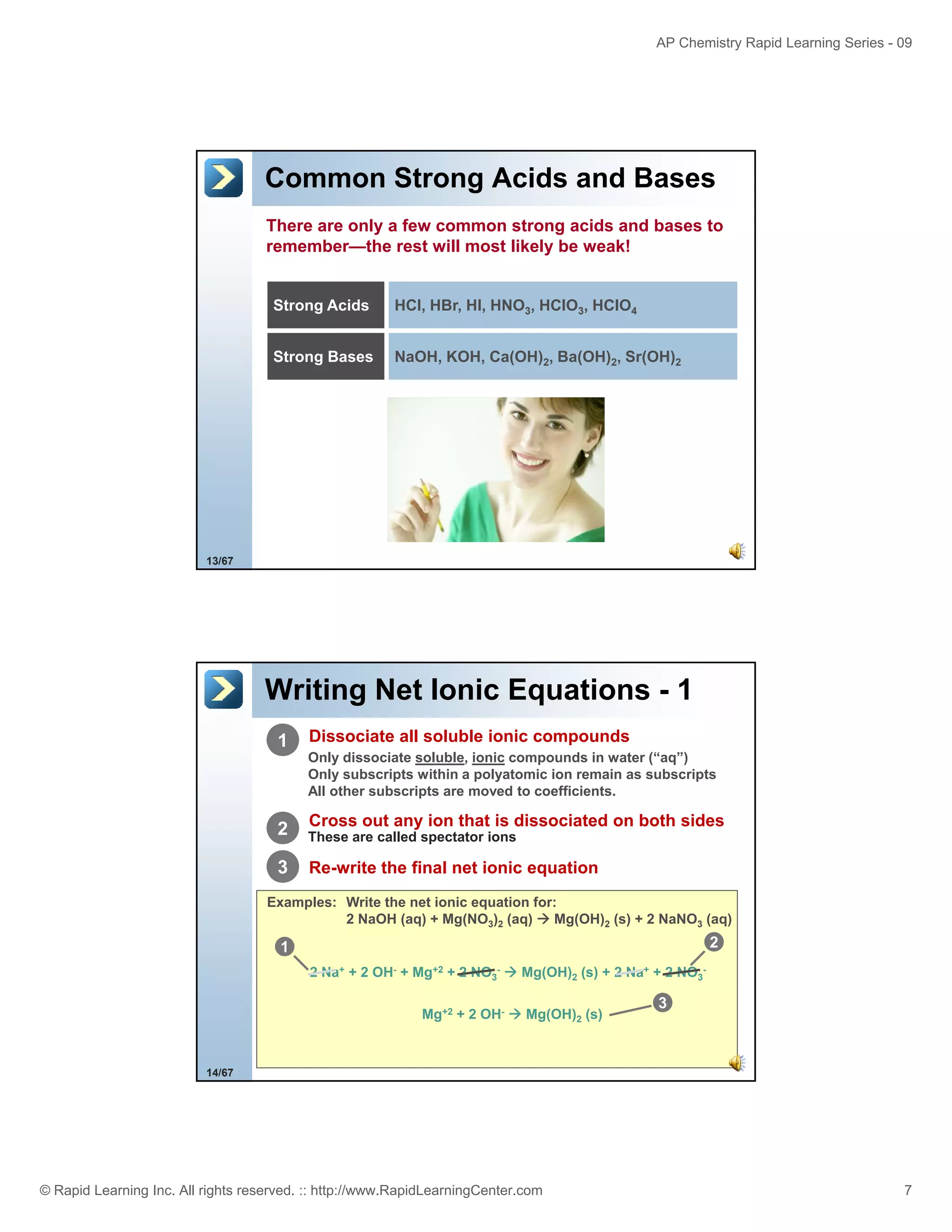
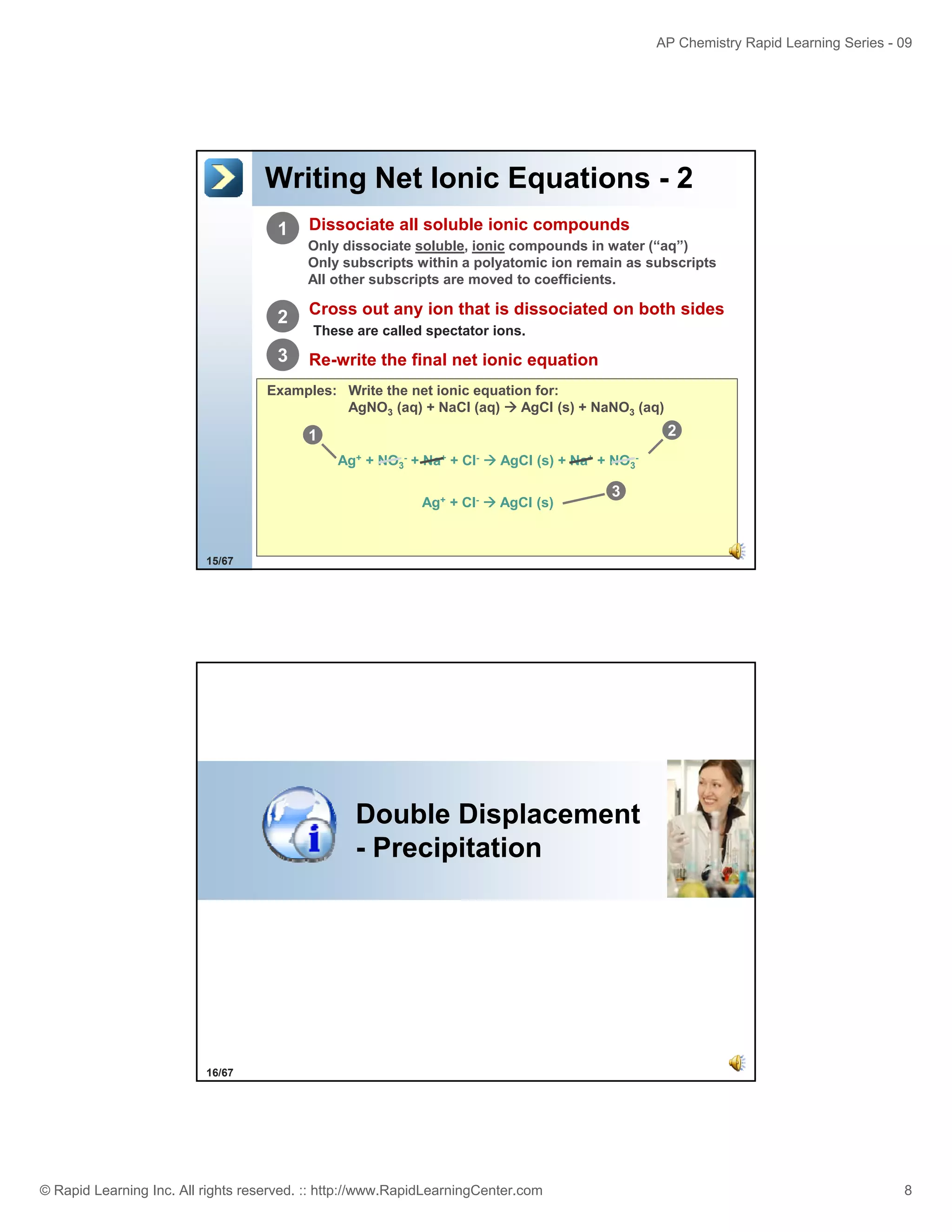
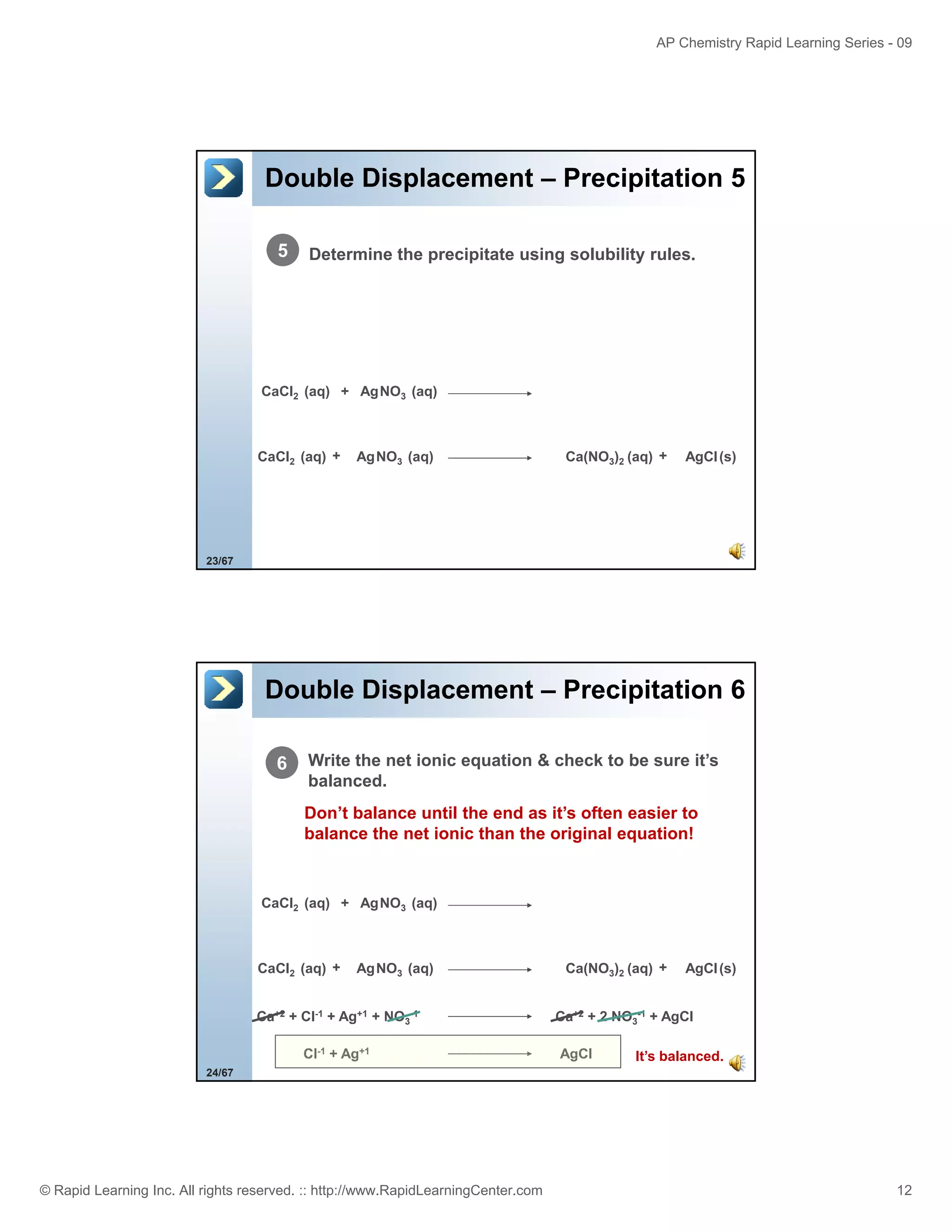
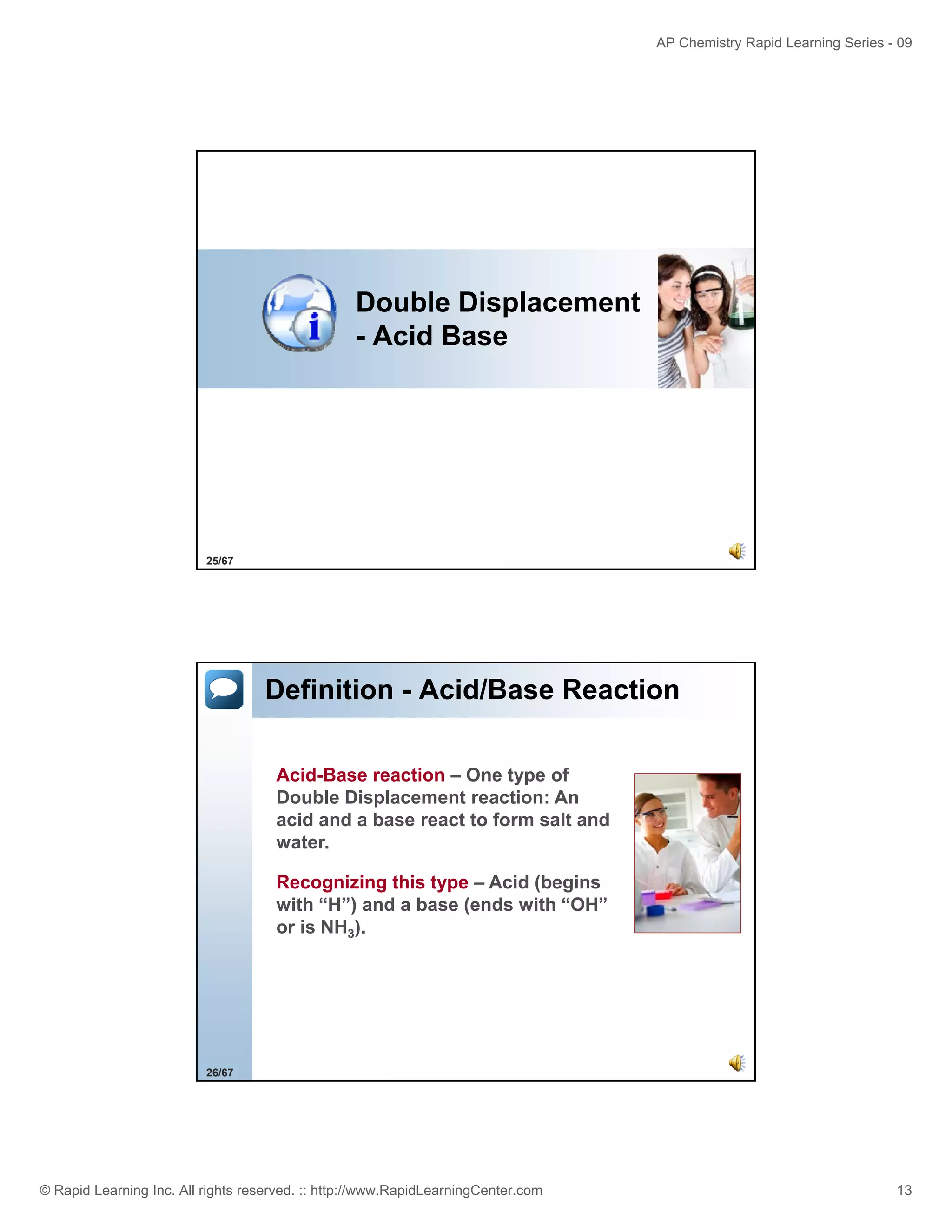
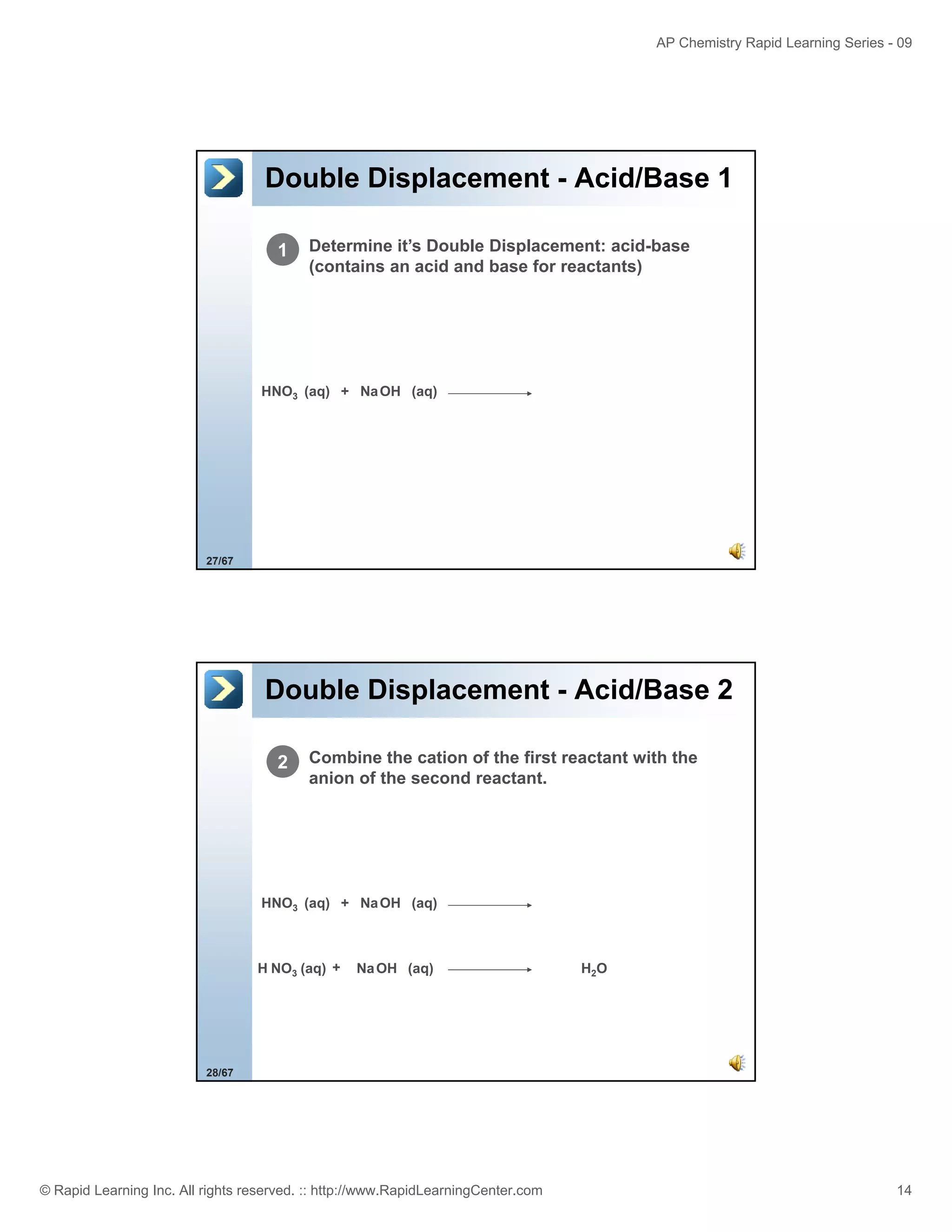
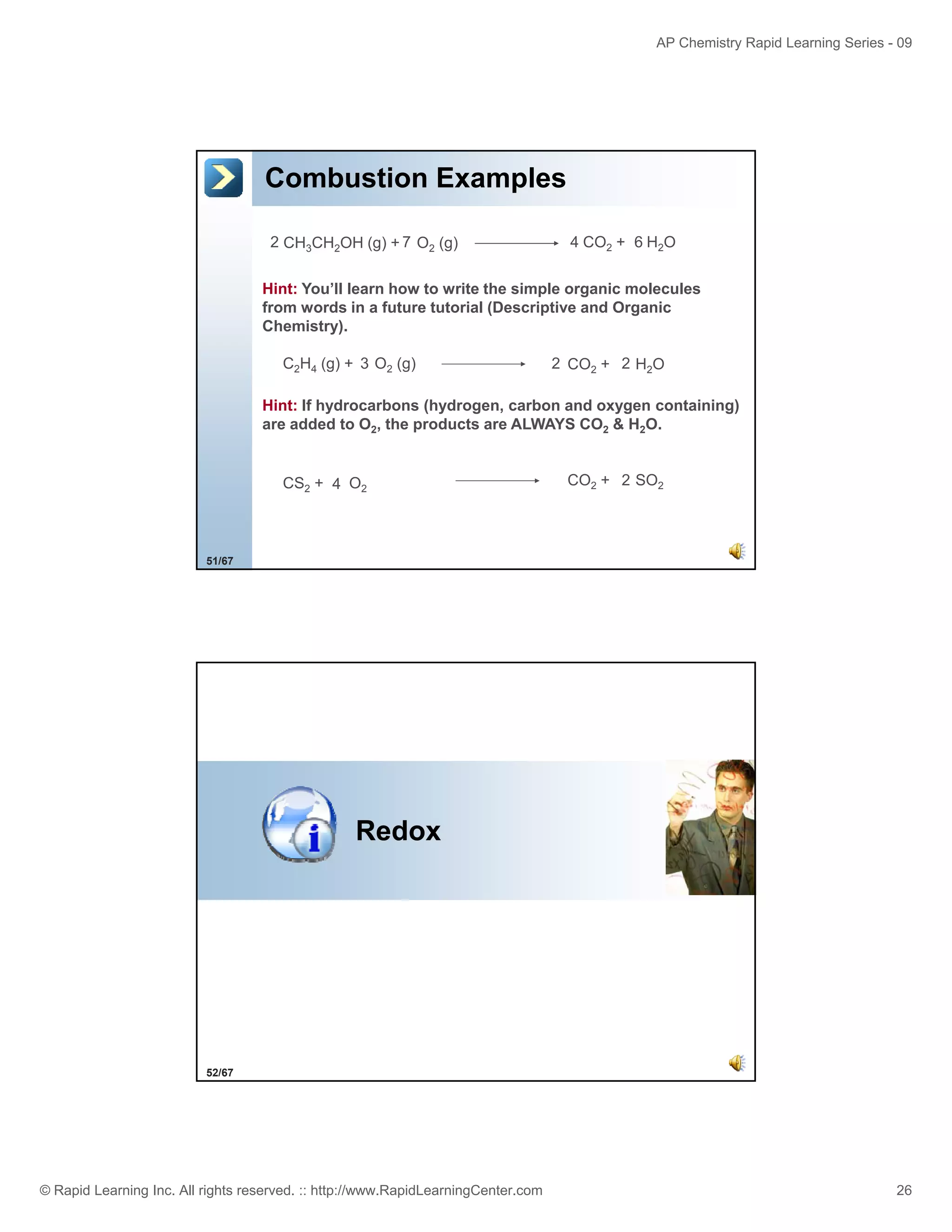
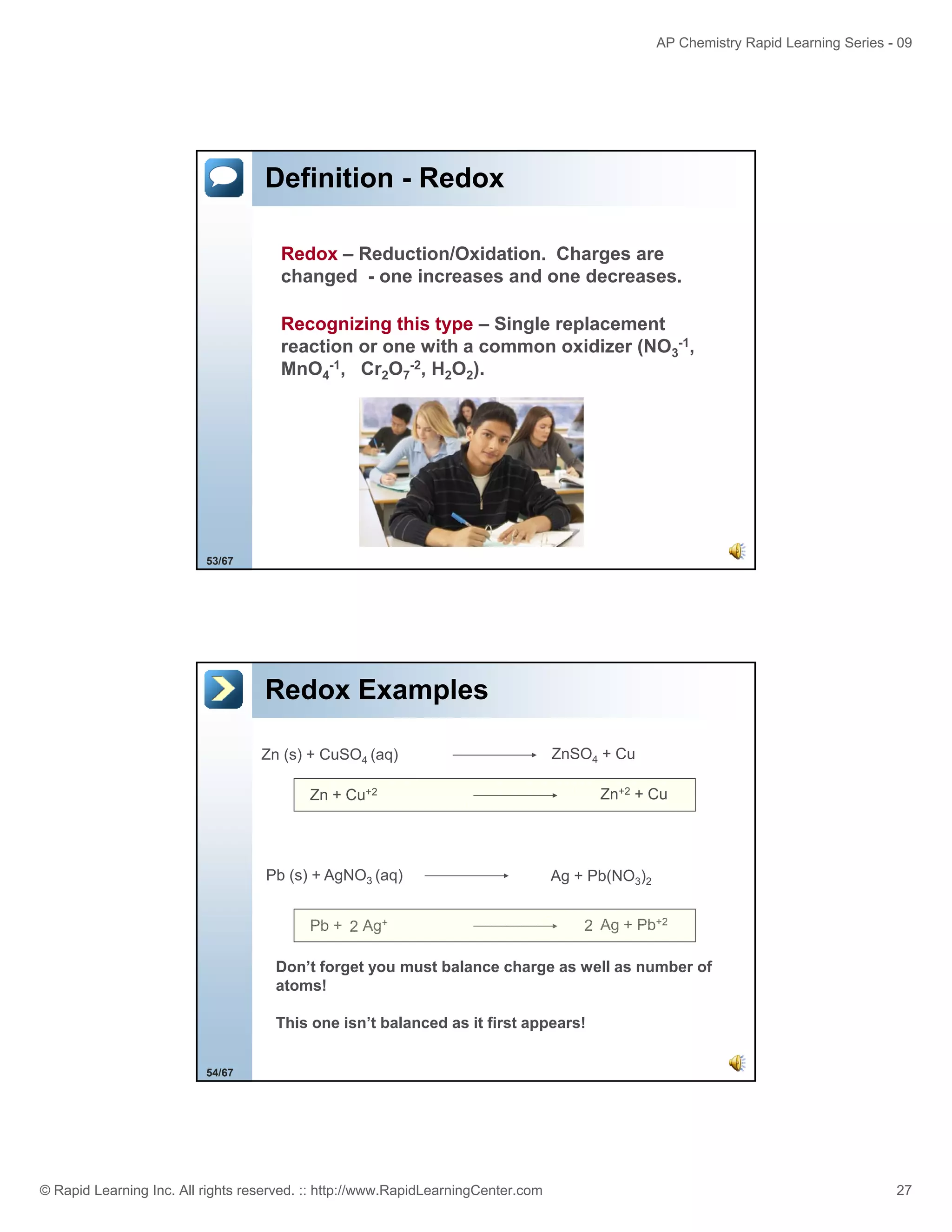
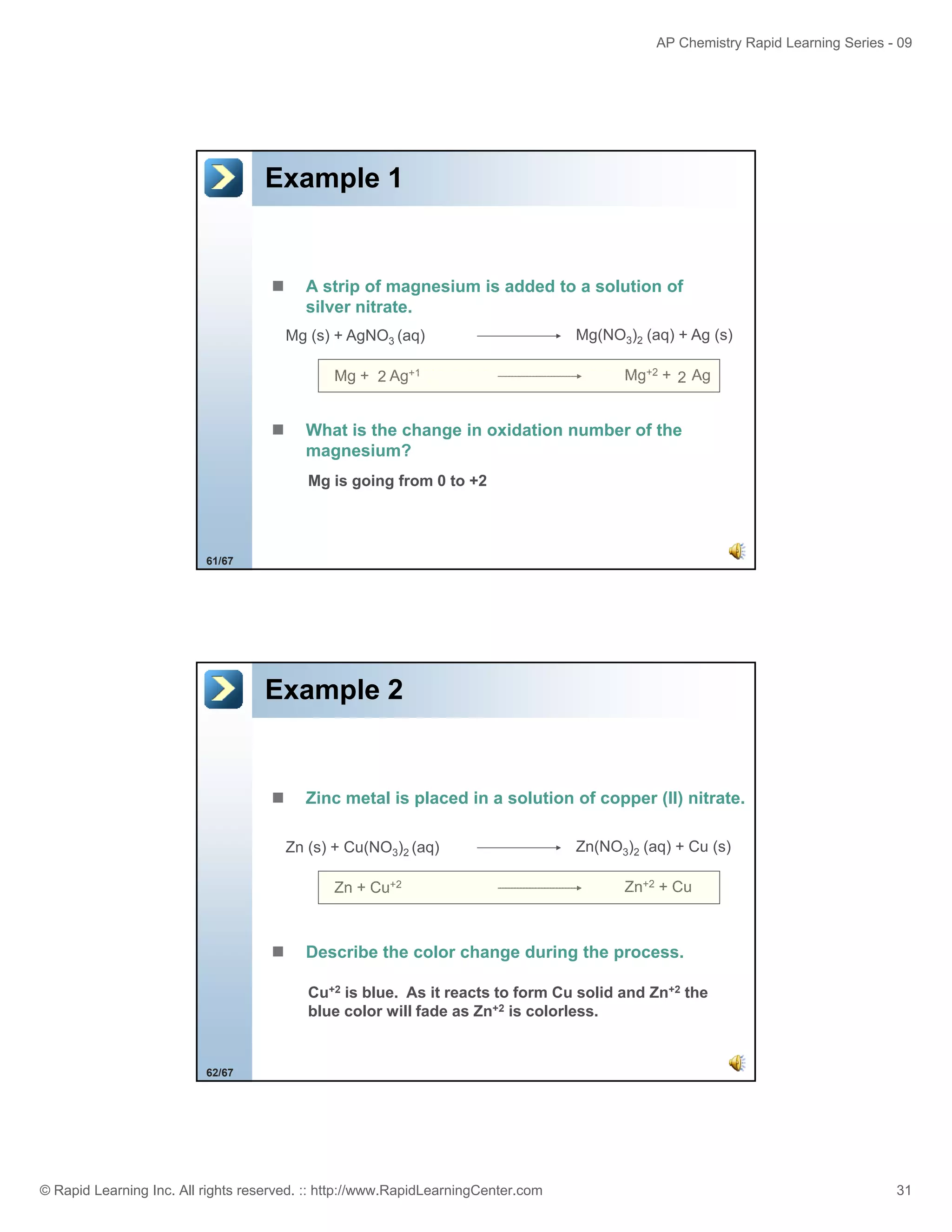
This document provides an overview of predicting products for the AP Chemistry exam question #4. It begins by explaining that question #4 asks students to write balanced net ionic equations for 3 given chemical reactions. It then reviews how to write net ionic equations, including dissociating ionic compounds and identifying spectator ions. The document outlines the steps to predict products for double displacement reactions, including precipitation reactions where an insoluble compound forms, and acid-base reactions where a salt and water form. It provides examples of writing net ionic equations for different reaction types that could appear in the AP question. The goal is to teach students the process for systematically predicting products to answer question #4.