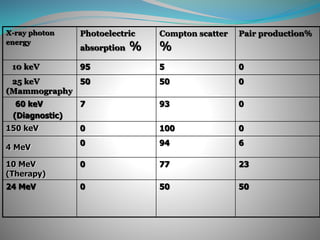
This document discusses the dual wave-particle nature of X-rays and their interaction with matter. It notes that X-rays can behave as both waves that propagate through space, as well as particles called photons. The two main interactions discussed are the photoelectric effect and Compton scattering.
The photoelectric effect occurs when a photon ejects an inner shell electron from an atom. This produces characteristic radiation, a photoelectron, and an ionized atom. It is most likely with low-energy photons and high atomic number elements. Compton scattering occurs when a high-energy photon ejects a loosely bound outer shell electron. This produces a recoil electron and scattered photon, with energy distributed between the two. Scattered photons are