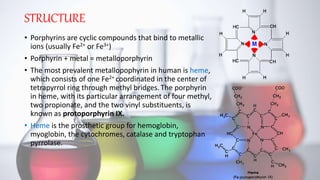
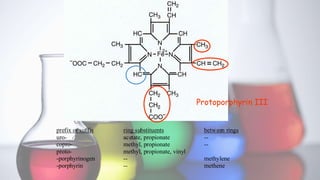
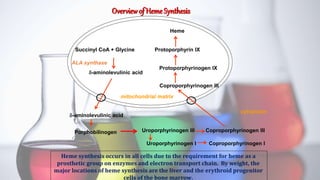
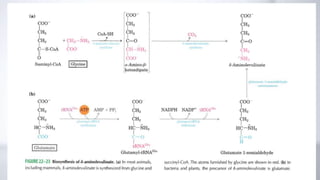
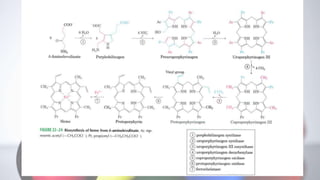
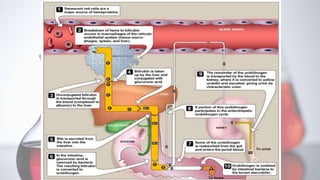
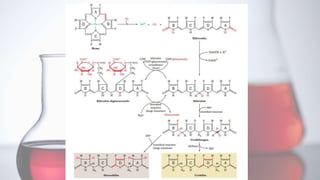
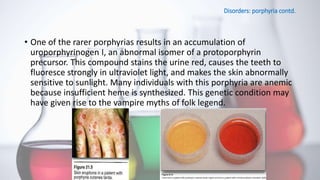
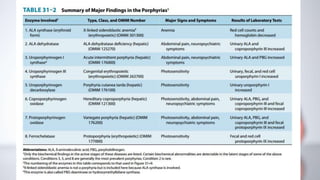
The document discusses porphyrin metabolism, focusing on heme synthesis, regulation, and catabolism. It details the pathways for heme synthesis, regulatory mechanisms in liver and erythroid cells, and the processes leading to bilirubin formation and excretion. It also covers disorders related to heme metabolism, including porphyrias and jaundice, highlighting their causes and symptoms.