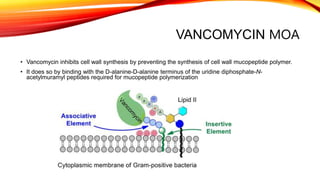
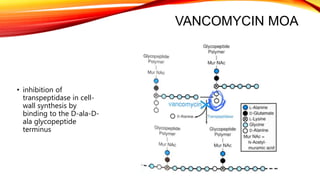
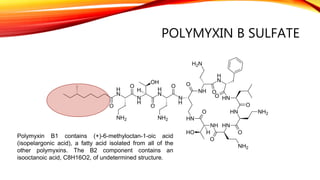
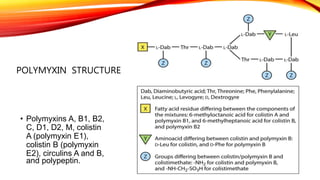
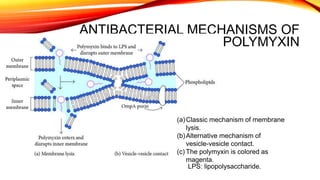
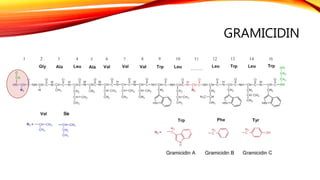
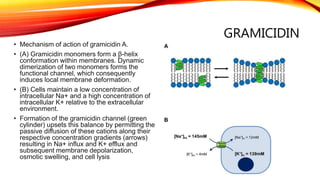
Polypeptide antibiotics, including vancomycin and teicoplanin, are primarily derived from bacterial sources and are effective against various gram-positive bacteria, although they have limitations such as renal toxicity and poor oral bioavailability. Vancomycin works by inhibiting cell wall synthesis and is administered intravenously, while teicoplanin offers advantages like greater lipid solubility and reduced toxicity. Other antibiotics in this class, including bacitracin and polymyxin B, also target bacterial cell walls and membranes but vary in their usage and mechanisms of action.