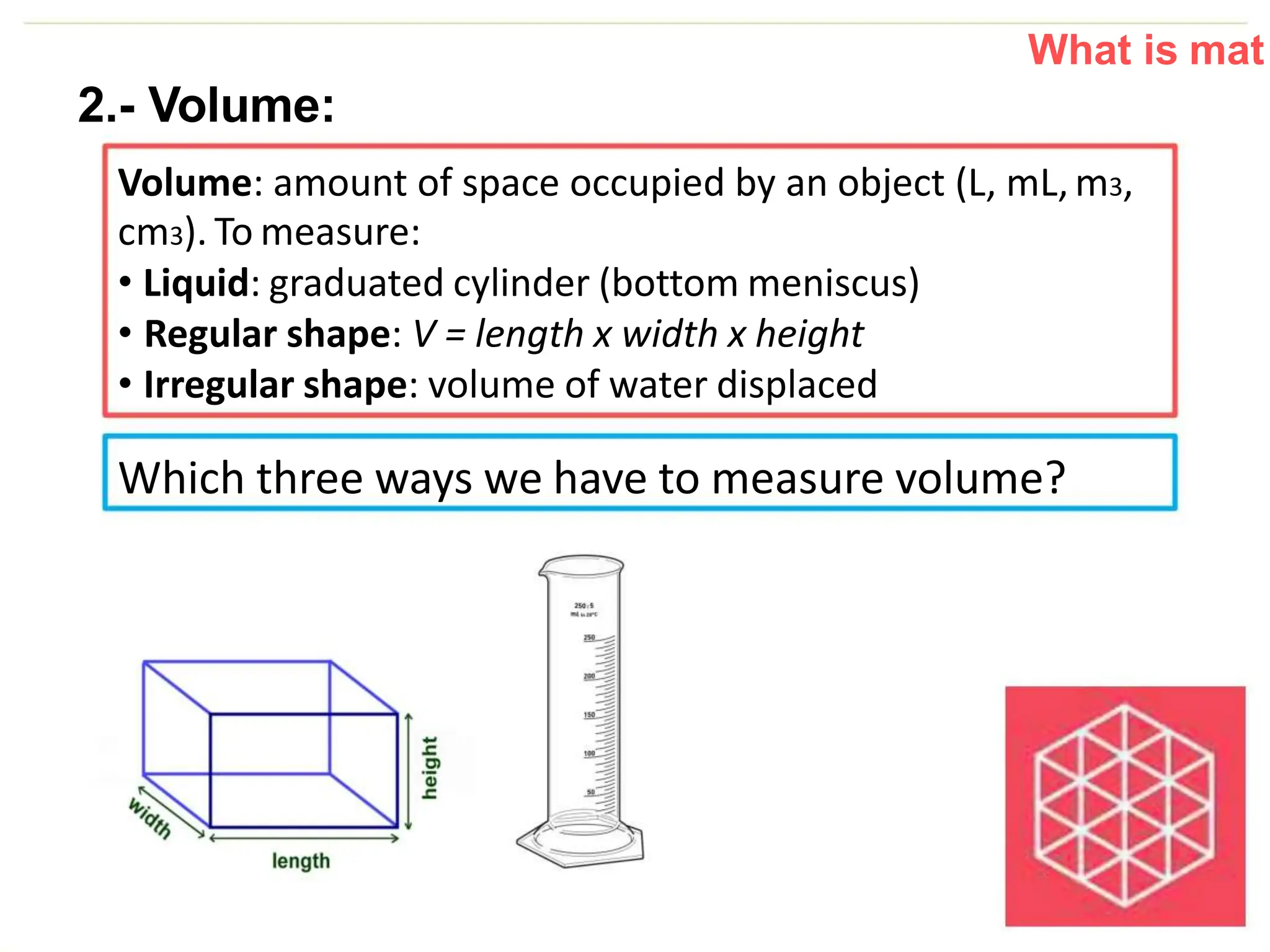
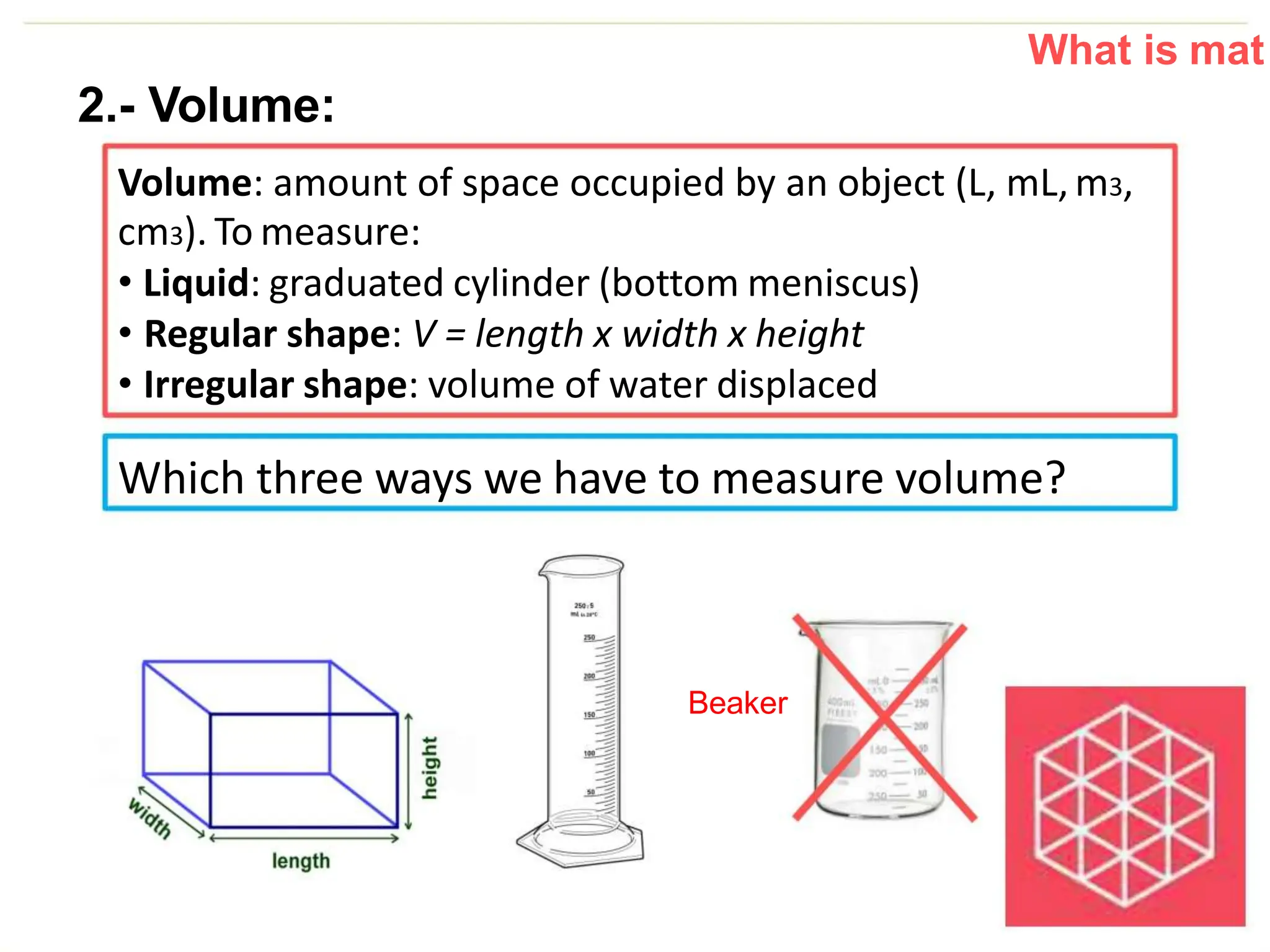
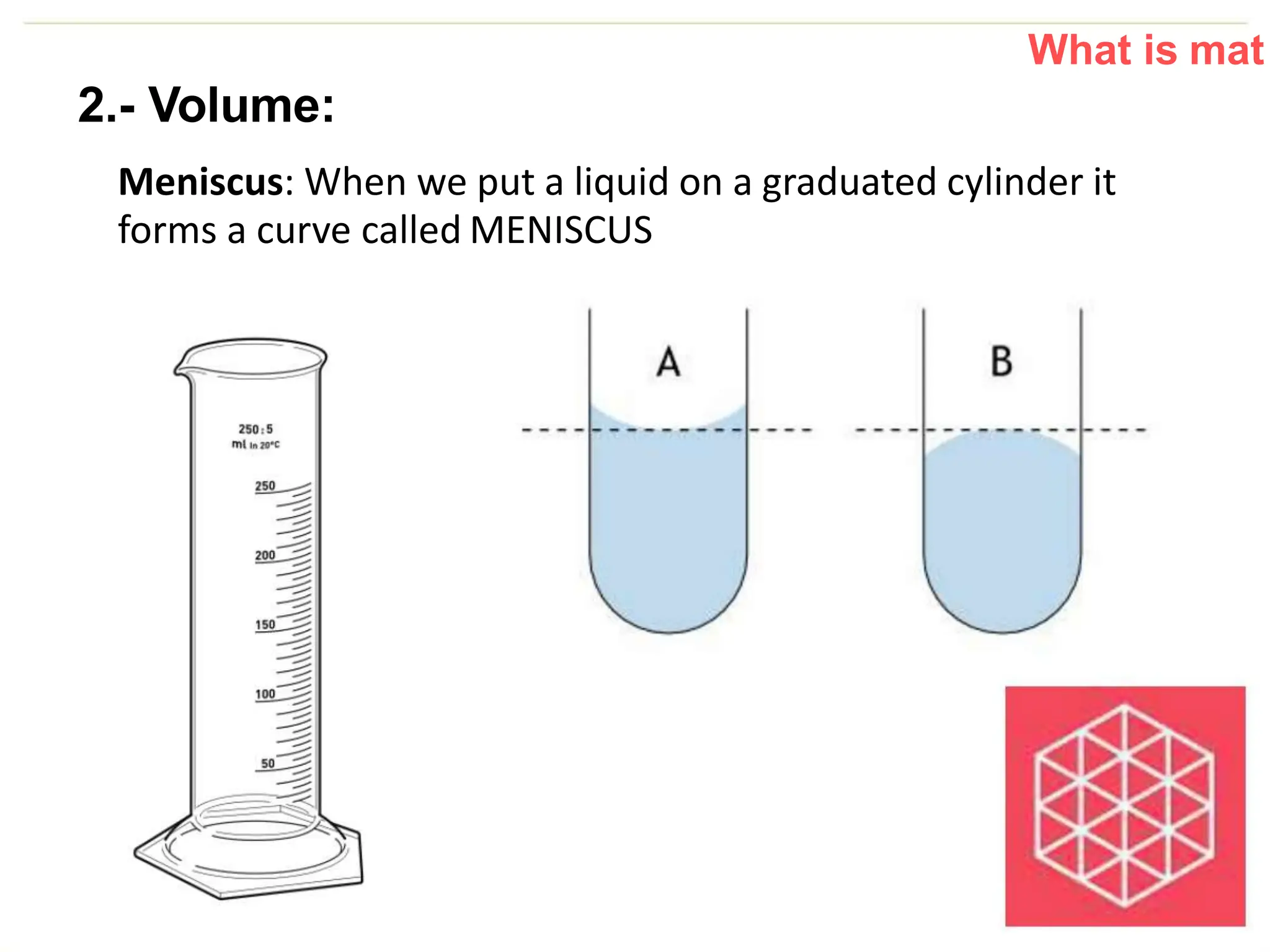
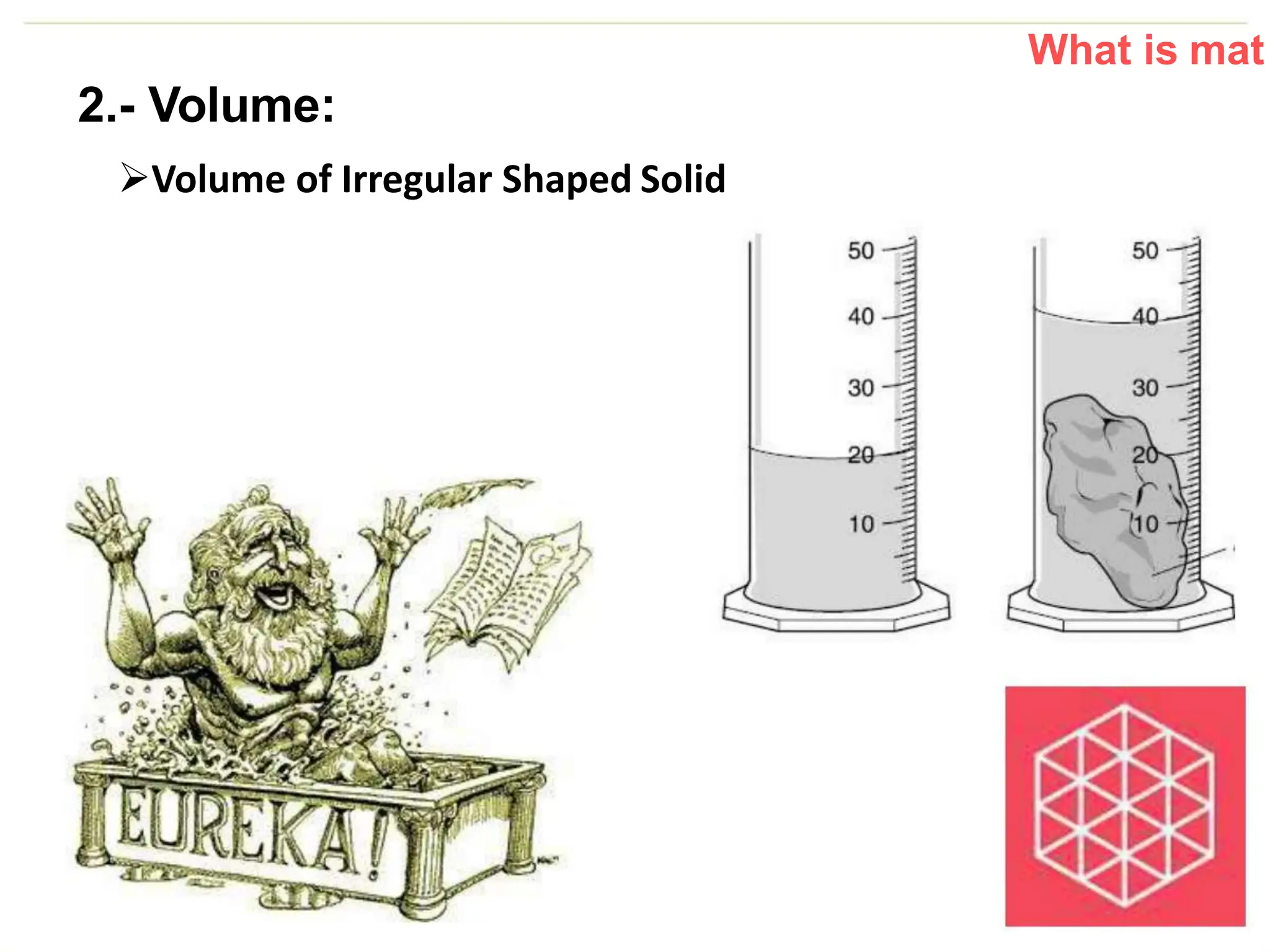
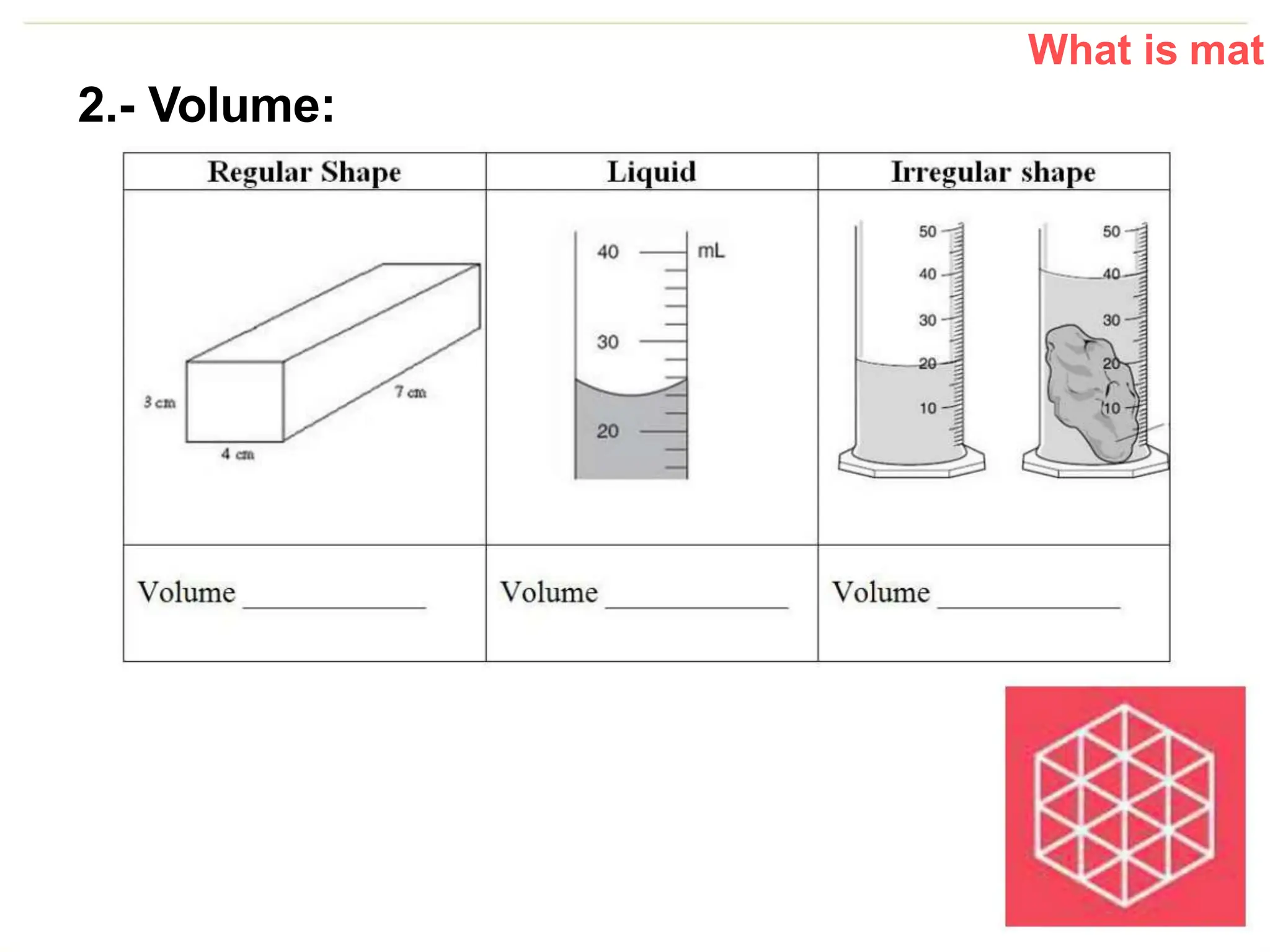
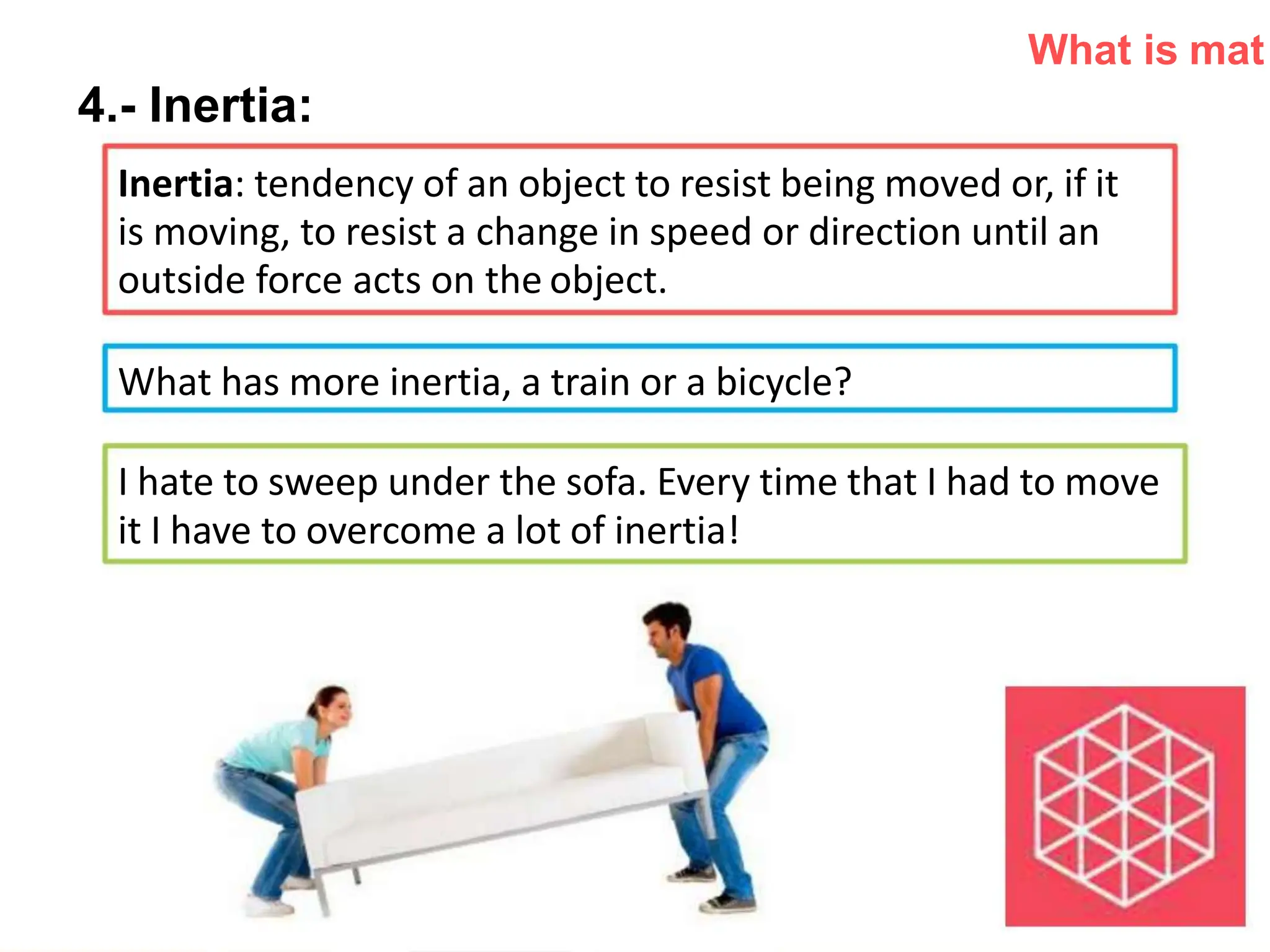
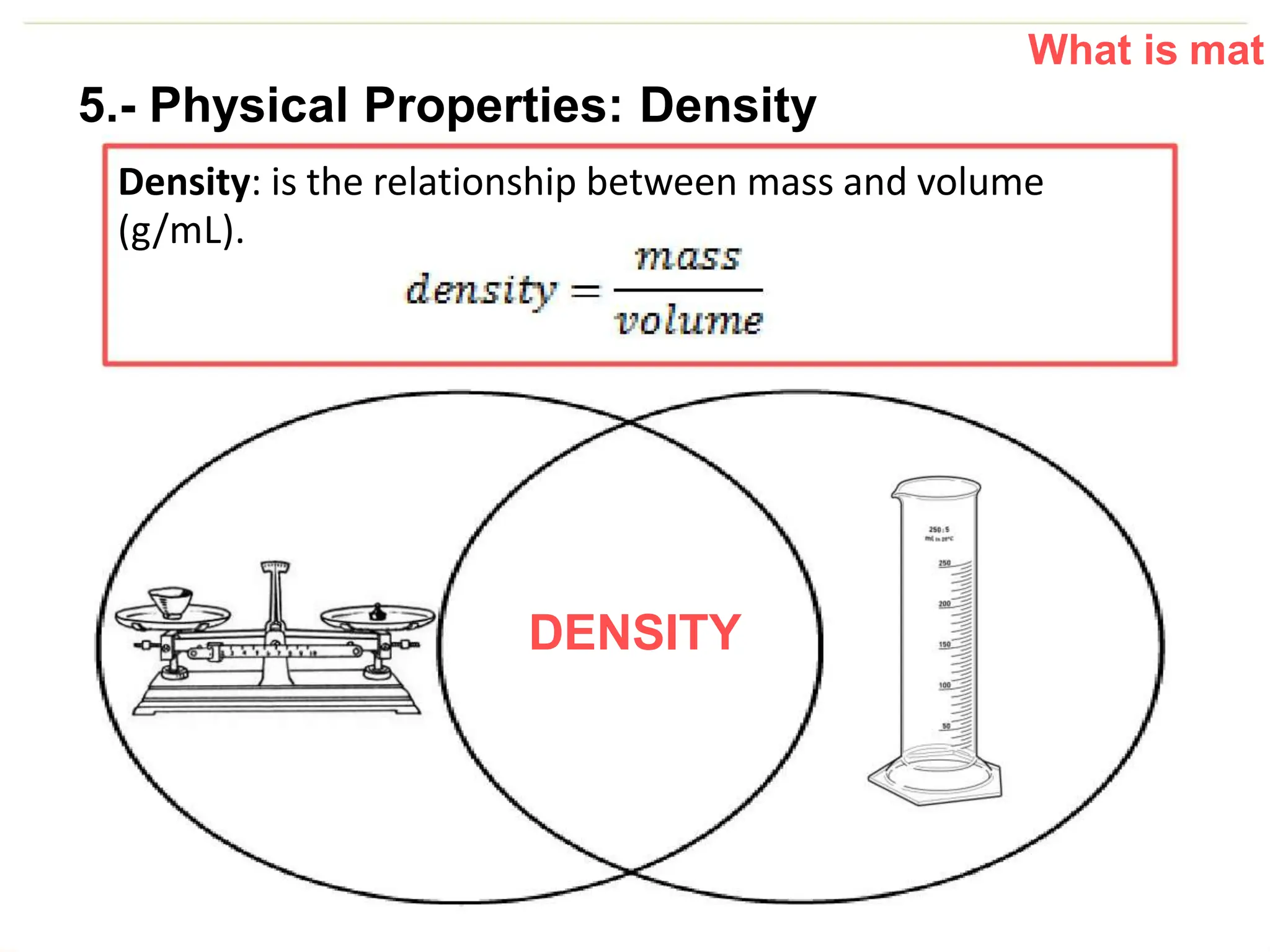
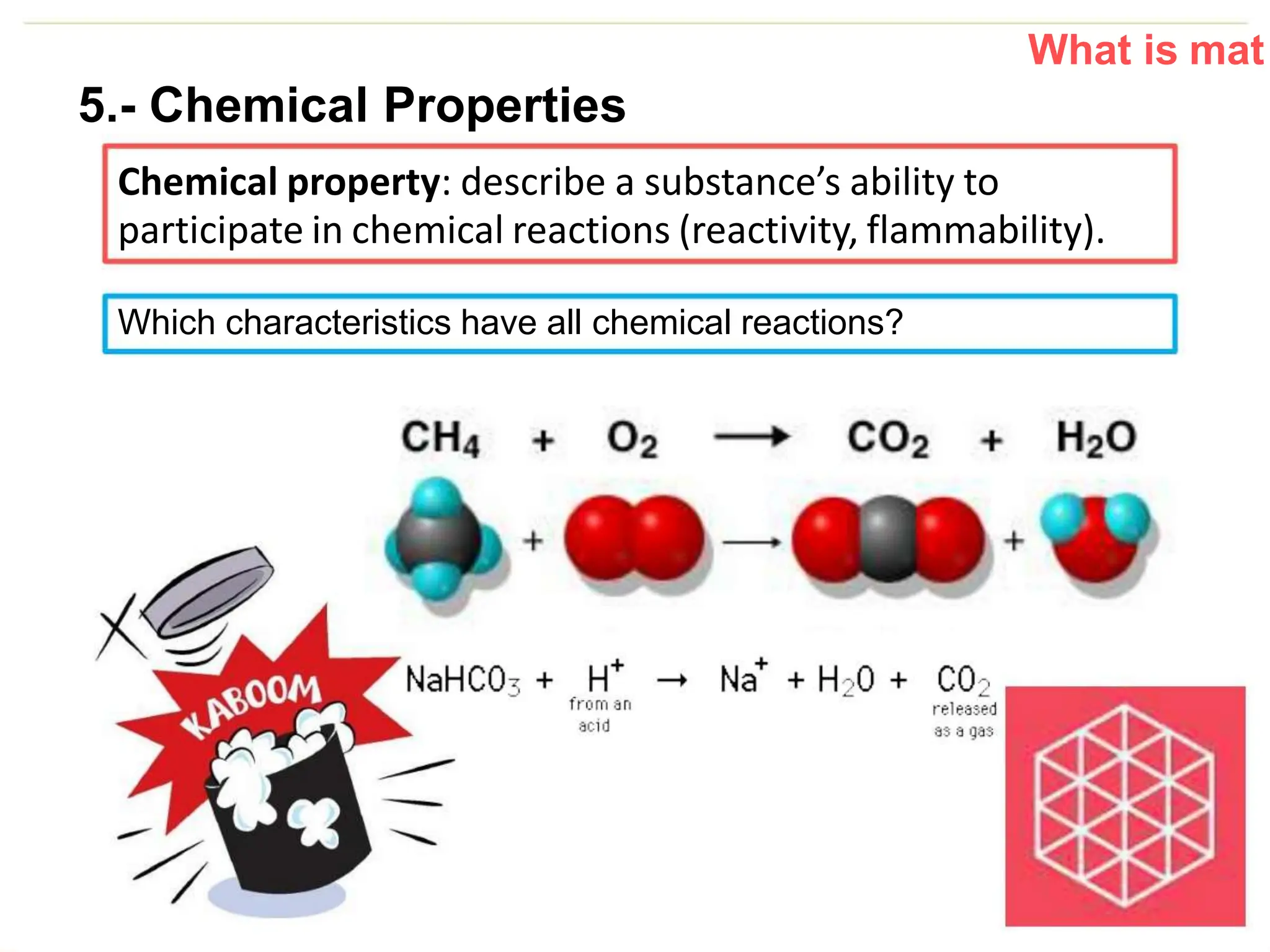
The document introduces the topic of matter and its key properties. It defines matter as anything that has mass and takes up space. It discusses various properties of matter including volume, mass, weight, inertia, physical properties like density, and types of changes including physical and chemical changes. As examples, it notes that volume can be measured in liquids using a graduated cylinder, and for irregular solids using water displacement. It also explains that mass is constant while weight depends on gravity.