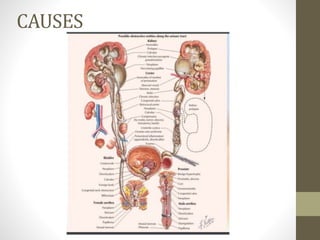
This document describes a case of obstructive uropathy in a 7-year-old male patient. It provides background on obstructive uropathy including definitions, prevalence, causes, pathophysiology, effects on renal function and structure, management, and diagnostic imaging techniques. Key details include unilateral right kidney hydronephrosis due to vesicoureteric junction obstruction, multiple stent placements and surgeries, and the use of CT scans, renography, and cystoscopy to diagnose and manage the patient's condition over time.