This document provides an introduction to nuclear magnetic resonance (NMR) spectroscopy. It discusses:
1) How NMR spectroscopy uses radio waves and magnetic fields to determine the structure of organic molecules. The two most common types are 1H NMR and 13C NMR.
2) In an NMR experiment, nuclei such as 1H and 13C can absorb energy and "flip" their spin when radio waves match their energy difference in magnetic fields.
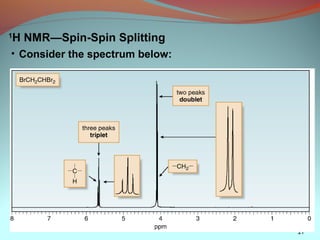
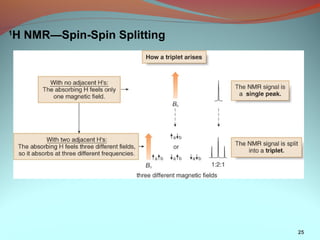
3) NMR spectra provide information on the number, position, intensity, and splitting of peaks which reveal details about a molecule's carbon-hydrogen framework.