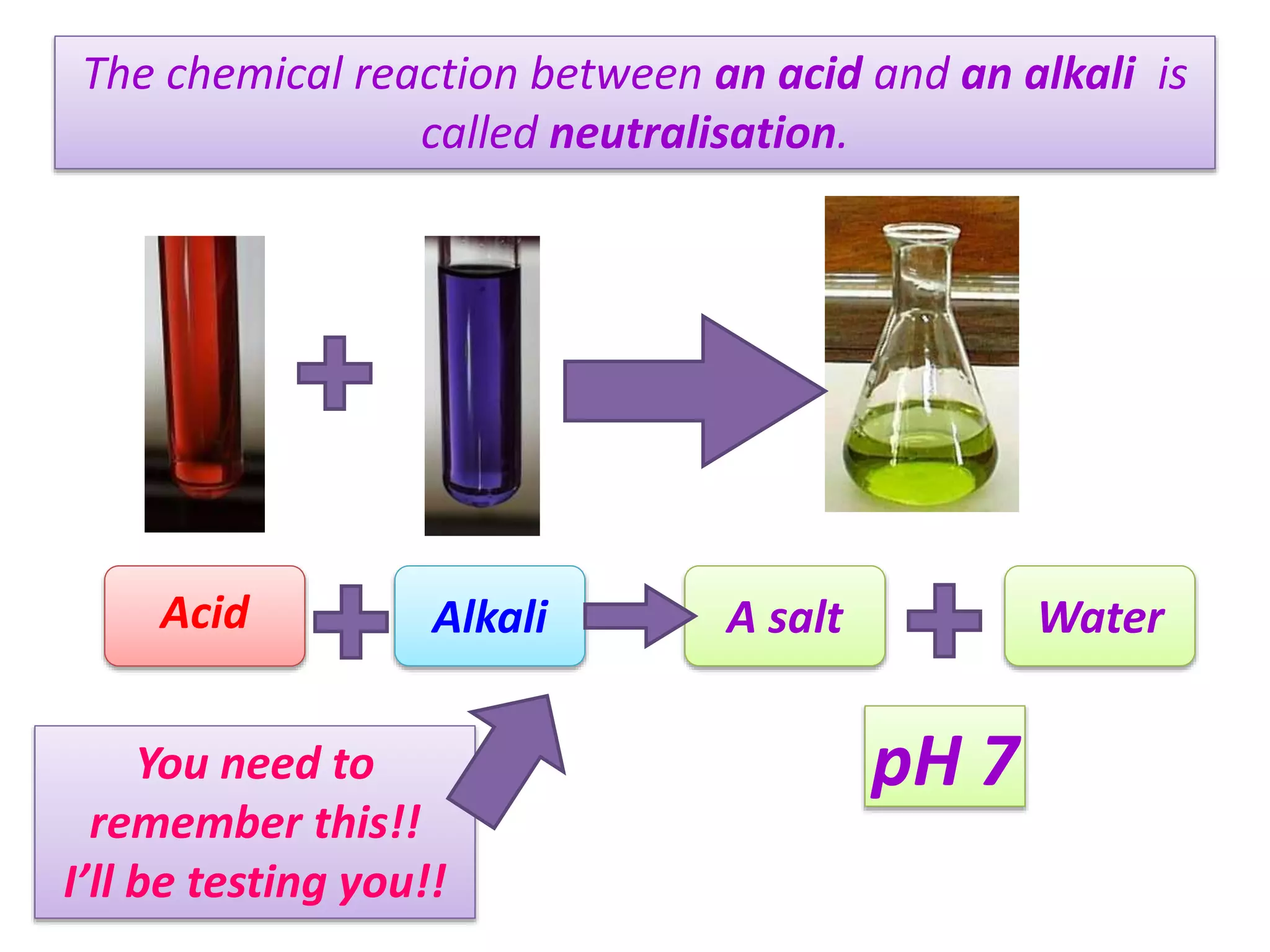
The document discusses the concept of neutralization, a chemical reaction between an acid and an alkali that results in the formation of salt and water, with a neutral pH of 7. It includes practical exercises and examples, such as using vinegar for wasp stings and laboratory instructions for creating a neutral solution. Additionally, it emphasizes the importance of word equations and real-life applications of neutralization, along with a scenario involving a chemical spill.