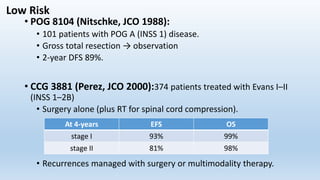
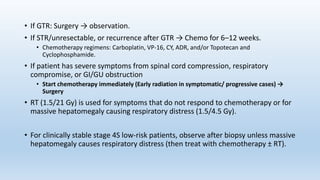
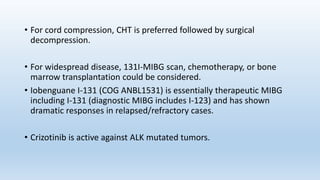
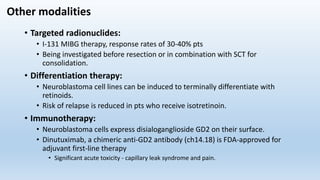
- Neuroblastoma is the most common extracranial solid tumor in children and accounts for 15% of pediatric cancer deaths.
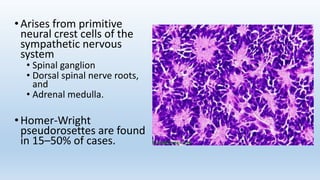
- It arises from primitive neural crest cells of the sympathetic nervous system and the abdomen is the most common primary site.
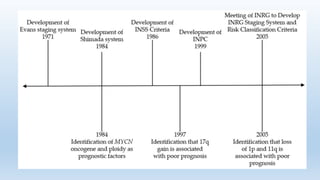
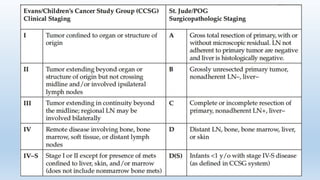
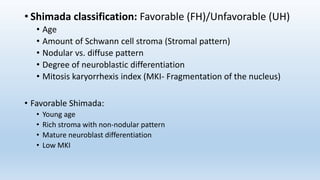
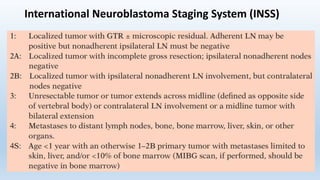
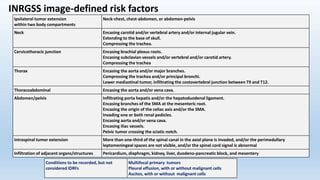
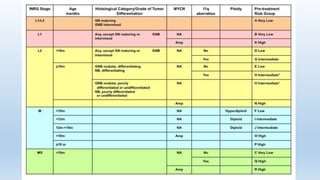
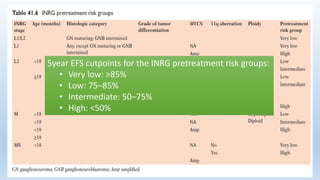
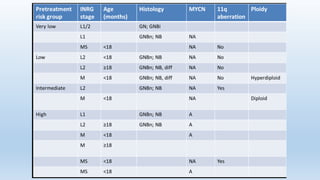
- Staging systems include the International Neuroblastoma Staging System and the International Neuroblastoma Risk Group Staging System, which classify disease as localized, metastatic, or multifocal based on imaging.
- Treatment involves chemotherapy, surgery, radiation therapy, stem cell transplant, and immunotherapy depending on risk stratification as low, intermediate, or high risk based on age, stage, biology, and other factors.










![Work-up
• H&P, Labs include urine catecholamines, vanillylmandelic acid, and homovanillic acid, CBC,
BUN/Cr, and LFTs.
Imaging: CT/MRI of primary, MIBG scan, and CXR.
• CXR/CT chest for metastases.
• Primary tumor is calcified on X-ray in 80–90% of cases (vs. 5–10% in Wilms’).
• MIBG Scan:
• Meta-iodobenzylguanidine scan uptake in 90 to 95% of all neuroblastomas (MIBG-avid) [Sensitivity 97% &
Specificity 92%]
• The mechanism is that MIBG is taken up by sympathetic neurons, and is a functioning analog of the
neurotransmitter norepinephrine.
• When it is radio-iodinated with I-131 or I-123, it is a very good radiopharmaceutical for diagnosis and
monitoring of response to treatment for this disease.
• Bone scan if primary tumor is not MIBG Avid.
• Biopsy the primary or involved nodes.
• All patients should have a bilateral bone marrow biopsy and aspirate.](https://image.slidesharecdn.com/neuroblastoma-191028120249/85/Neuroblastoma-14-320.jpg)