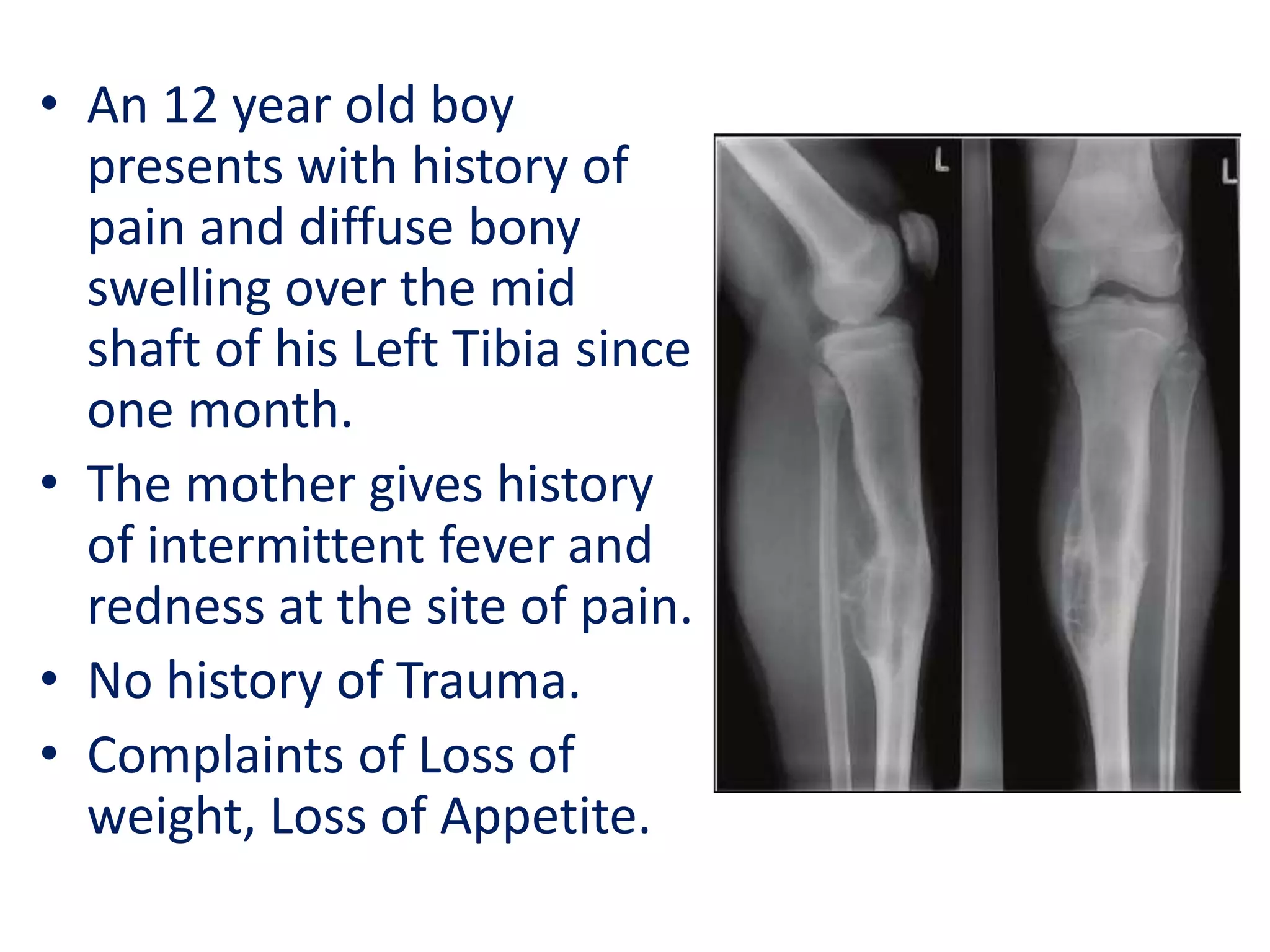
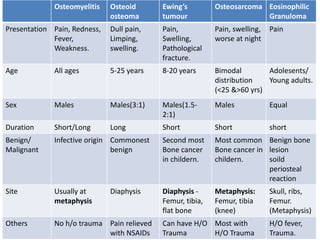
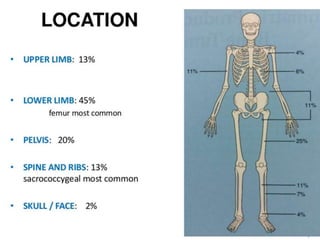
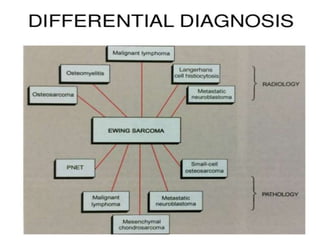
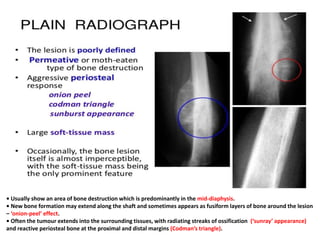
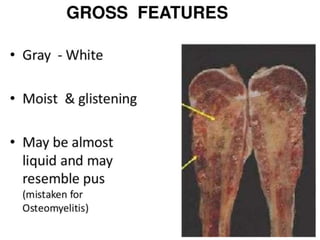
- A 12-year-old boy presented with pain and swelling in his left tibia for one month with a history of intermittent fever. Differential diagnoses included osteomyelitis, osteoid osteoma, Ewing's sarcoma, and osteosarcoma.
- Ewing's sarcoma most commonly affects children and young adults between 5-25 years old and presents with pain, swelling, and sometimes pathological fractures. Definitive diagnosis is based on histology, immunohistochemistry, and detection of specific gene fusions.
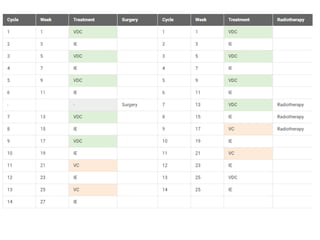
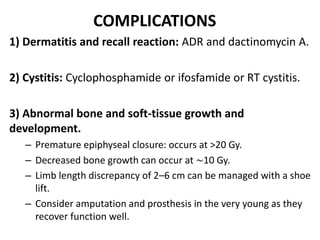
- Treatment involves chemotherapy with VACA/IE cycles alternating every 2-3 weeks for 17 cycles along with possible surgery and/or radiation therapy based on response and margins. The goal is






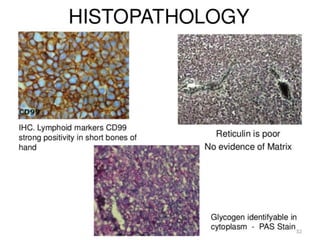
![• The characteristic immunostain is CD99, which diffusely marks
the cell membrane.
(CD99 is not specific for Ewing's sarcoma)
• The most common translocation, present in about 90% of Ewing
sarcoma cases, is t(11;22)(q24;q12)
• Generates an aberrant transcription factor through fusion of the
EWSR1 gene with the FLI1 gene.[FLI1(11): EWS(22)]
• Other minor translocations include:
• t(21;22), c-Myc proto-oncogene(chromosome 21) and EWS
gene(chromosome 22) in 10% of cases.
• t(7;22)](https://image.slidesharecdn.com/1-ewingssarcoma-190913144632/85/Ewing-s-sarcoma-16-320.jpg)


![Poor prognosis in EWS
• Male gender
• Age >15 yrs (>17 yrs in some)
• Pelvic/axial Site or rib origin
• Size (>8 cm per St. Jude or >100 cc per CESS-
81 [Cooperative Ewing Sarcoma Studies])
• Stage (presence/absence of metastatic Dz is
strongest prognostic factor)
• ↑LDH
• Poor response to chemo (>10% viable tumor)
• No surgery](https://image.slidesharecdn.com/1-ewingssarcoma-190913144632/85/Ewing-s-sarcoma-19-320.jpg)
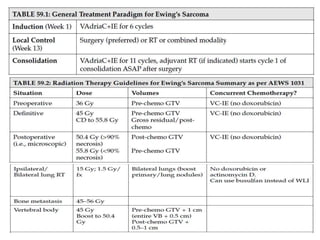


![• VAC/IE (vincristine + doxorubicin [Adriamycin] +
cyclophosphamide alternating with ifosfamide + etoposide)
• Alternating VAC and IE cycles.
• Repeat each cycle every 3 weeks for 17 cycles.
VAC cycles
• Day 1: Vincristine 2mg/m2 (max 2mg) IV + doxorubicin
75mg/m2 IV bolus + cyclophosphamide 1,200mg/m2 IV.
– Dactinomycin can be substituted for doxorubicin if there are
concerns regarding cardiotoxicity
– Dactinomycin 1.25mg/m2 IV can be substituted for doxorubicin
when a total doxorubicin dose of 375mg/m2 is reached.
IE cycles
• Days 1–5: Ifosfamide 1,800mg/m2 IV + mesna + etoposide
100mg/m2 IV.](https://image.slidesharecdn.com/1-ewingssarcoma-190913144632/85/Ewing-s-sarcoma-23-320.jpg)