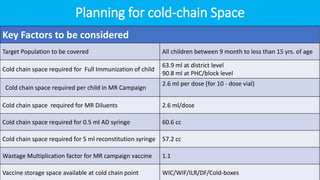
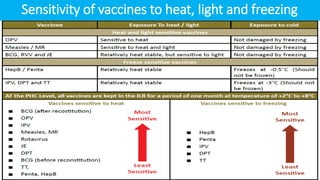
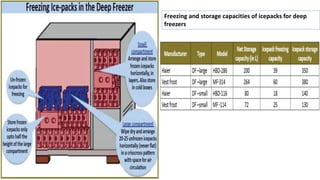
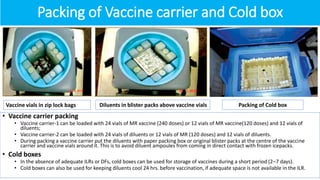
Cold chain management is crucial for the successful implementation of measles rubella vaccination campaigns. Key steps include reviewing existing cold chain equipment and capacity, estimating vaccine and logistical needs, ensuring adequate storage space for vaccines and diluents, planning for ice pack freezing, and developing contingency plans. Proper packing and transportation of vaccines while maintaining the cold chain is also important both during distribution and at vaccination sessions. Calculating cold chain space requirements and monitoring equipment is essential to reduce vaccine wastage.