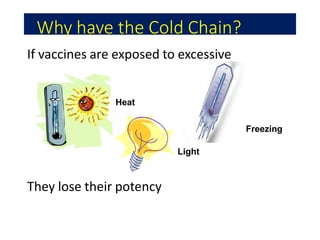
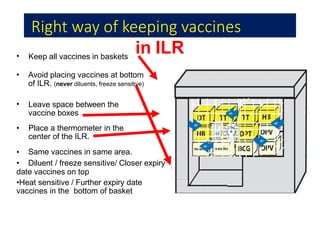
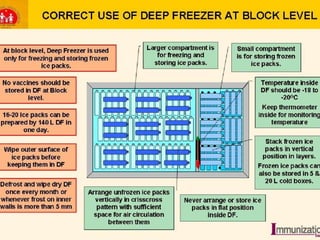
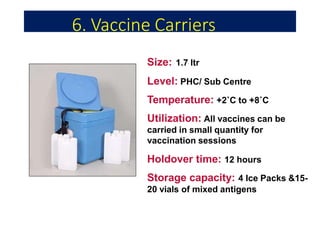
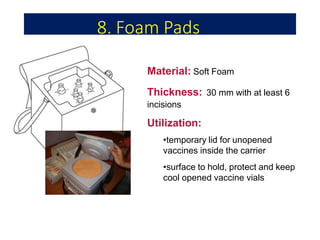
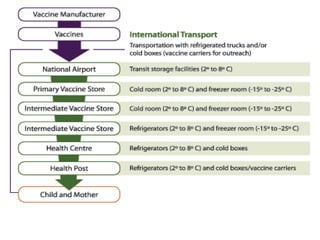
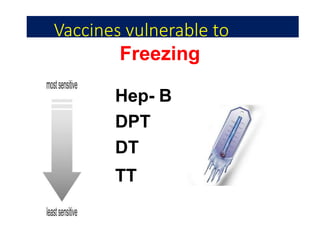
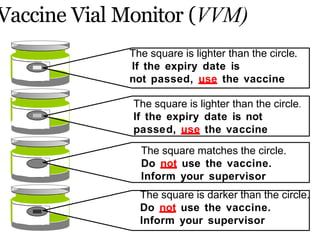
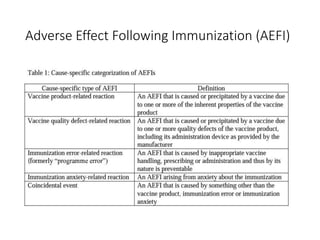
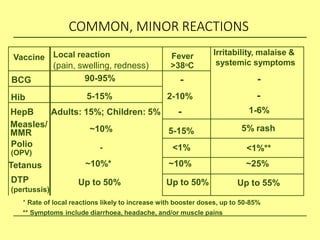
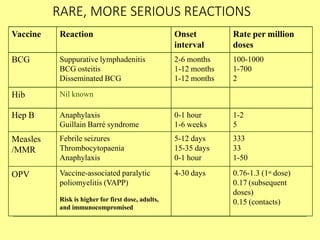
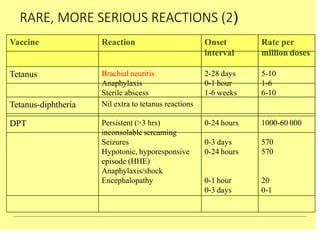
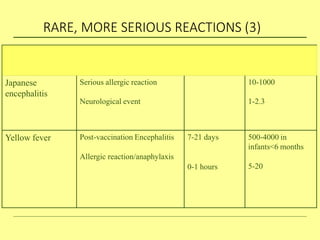
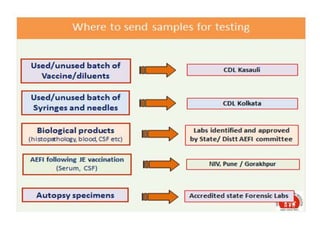
The document discusses India's cold chain system for vaccine storage and transportation. It describes the various levels of the cold chain from the state down to primary health centers and sub-centers. Key equipment used includes walk-in freezers and coolers, refrigerated trucks, ice-lined refrigerators, deep freezers, cold boxes, vaccine carriers, and ice packs. Proper use and maintenance of the cold chain is important to maintain vaccine potency as vaccines can be damaged by excessive heat, freezing, or light exposure. Failure to properly store and transport vaccines may lead to vaccine failure and adverse events following immunization.