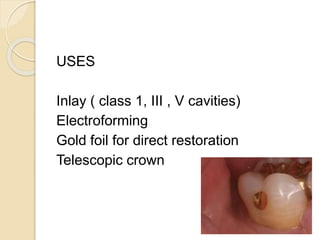
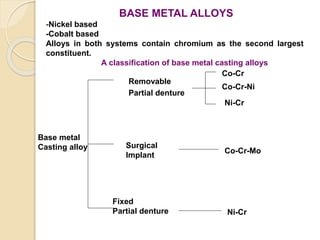
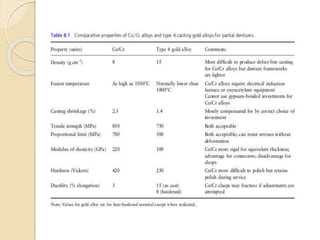
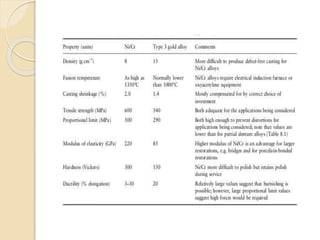
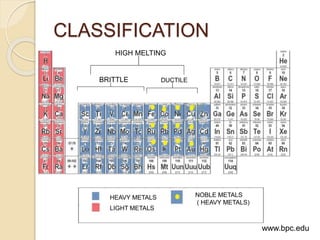
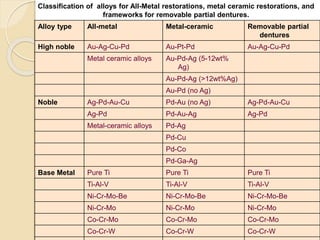
This document discusses metals and alloys used in dentistry. It defines key terms related to the mechanical properties of metals like ductility, hardness, strength and elasticity. It describes how alloys are classified based on their composition and properties. Common alloys used in dentistry include noble metal alloys like gold-based and palladium-based alloys, and base metal alloys like nickel-chromium and cobalt-chromium alloys. The document traces the history of alloy use and development in dentistry from gold to newer alloys that are stronger, cheaper and better suited for applications like metal-ceramic restorations and removable partial dentures.











![National Bureau of Standards
Type I (Soft, Vickers hardness number
[HV] between 50 and 90)
Type II (Medium, HV between 90 and
120)
Type III(Hard, HV between 120 and
150)and
Type IV (Extra Hard, HV ≥150).
APPLIED DENTAL MATERIALS –McCABBES AND WALLS](https://image.slidesharecdn.com/metalsandalloys-141031060317-conversion-gate01/85/Metals-and-alloys-13-320.jpg)
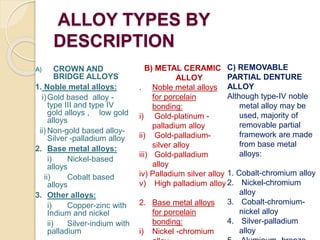
![ALLOY TYPE BY MAJOR ELEMENT: Gold-based, palladium-based,
silver-based, nickel-based, cobalt-based and titanium-based
.
ALLOY TYPE BY PRINCIPAL THREE ELEMENTS: Such as
Au-Pd-Ag, Pd-Ag-Sn, Ni-Cr-Be, Co-Cr-Mo, Ti-Al-V and Fe-Ni-
Cr.
(If two metals are present, a binary alloy is formed; if three
or four metals are present, ternary and quaternary alloys,
respectively, are produced and so on.)
ALLOY TYPE BY DOMINANT PHASE SYSTEM: Single
phase [isomorphous], eutectic, peritectic and intermetallic.](https://image.slidesharecdn.com/metalsandalloys-141031060317-conversion-gate01/85/Metals-and-alloys-16-320.jpg)