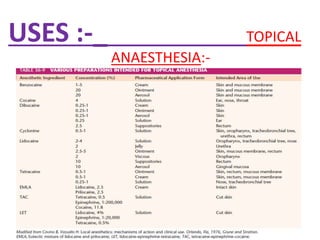
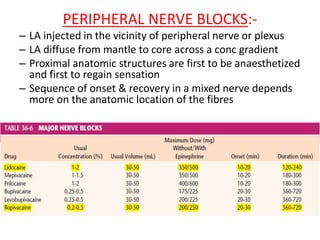
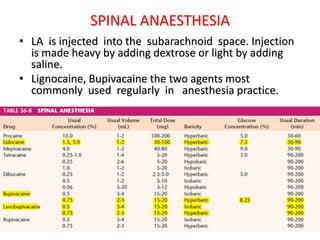
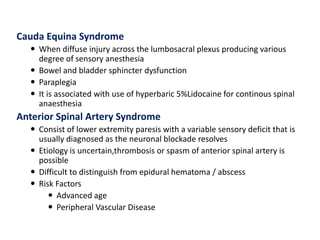
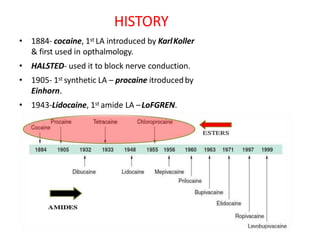
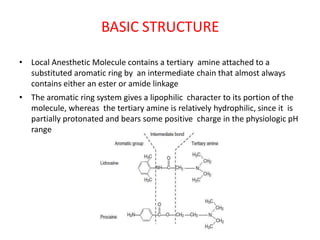
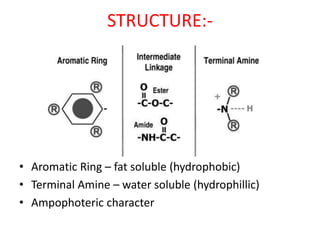
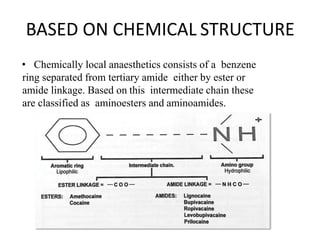
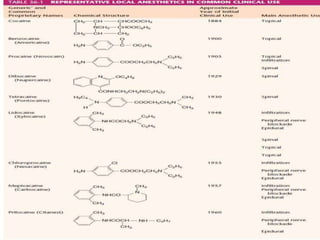
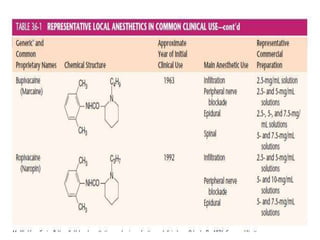
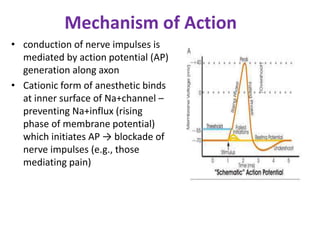
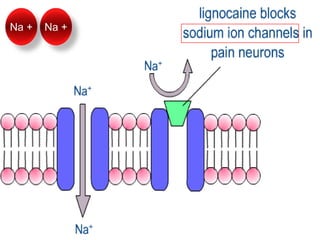
The document discusses local anesthetics, covering their introduction, classification, mechanism of action, and metabolism. It details properties of ideal local anesthetics, the historical context of their development, and specific agents like lignocaine and bupivacaine, emphasizing their pharmacokinetics and potential systemic effects. Additionally, it addresses factors influencing anesthetic effectiveness, such as ionization and binding characteristics, and provides clinical applications and considerations for safety and efficacy.








































![Additives and modifiers
of LA activity
• Increasing dose: ↓latency of onset; ↑duration, ↑block
success, ↑[LA]
• Vasoconstrictors: ↑duration, ↑block success, ↓[LA]
• α2 agonists: ↑duration,↑[LA]
• Opioids: ↑duration; permit ↓LA dose
• Alkalinization (usually NaHCO3): ↓latency of onset,
↑potency
• Pregnancy: ↑dermatomal spread, ↑LA potency, ↑free
blood ( doses should be decreased in patients in all
stages of pregnancy.)](https://image.slidesharecdn.com/localanaesthetics-191224143949/85/Local-anaesthetics-54-320.jpg)