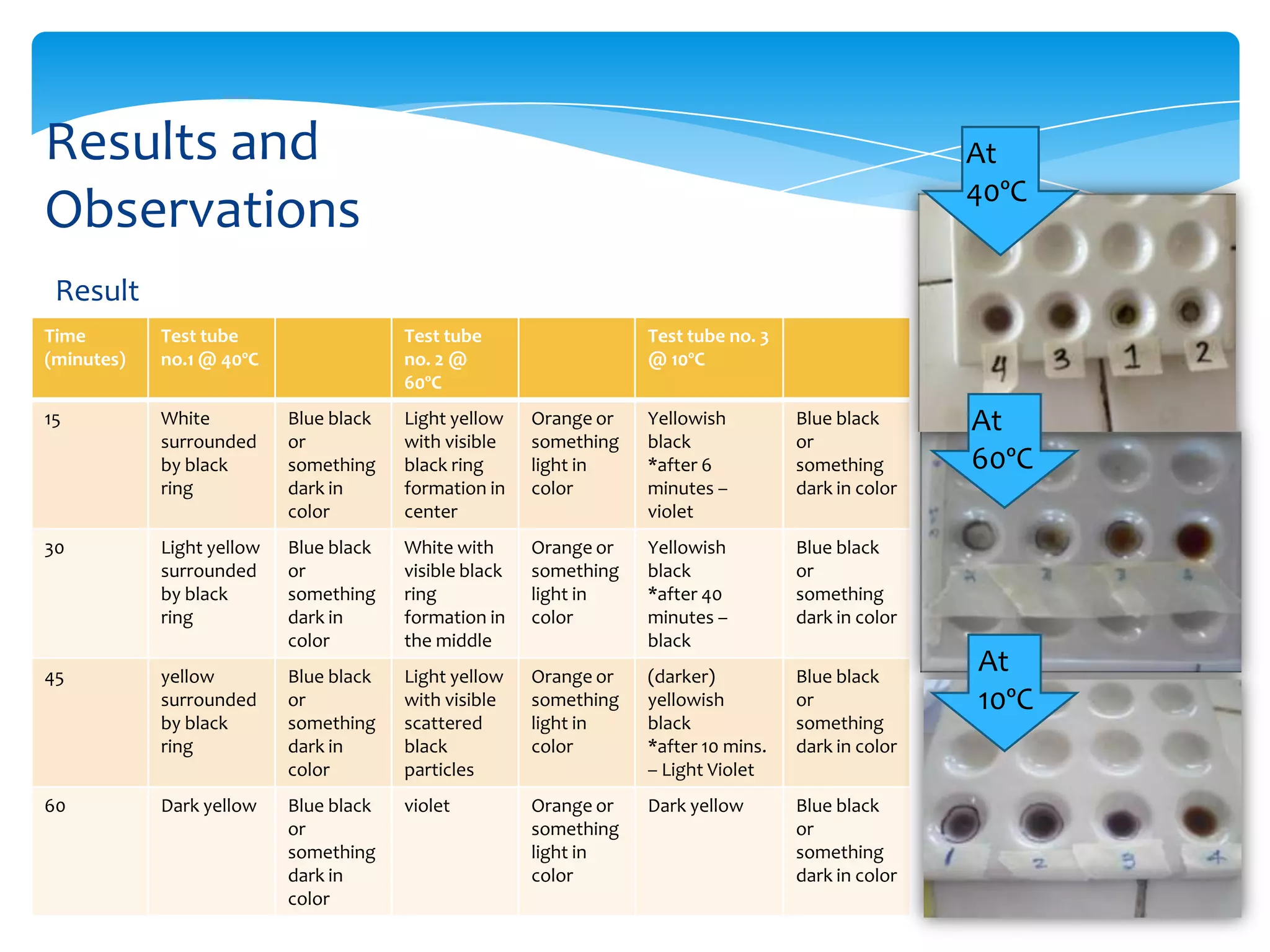
This document describes experiments that test how temperature and pH affect enzyme activity. The temperature experiment uses salivary amylase and starch solution in three test tubes held at 10°C, 40°C, and 60°C. Iodine tests show that activity is highest at 40°C and stops at 60°C. The pH experiment uses pepsin, pancreatin, egg white, HCl, and Na2CO3 to create acidic and basic conditions. Biuret tests find that enzyme activity occurs under both acidic and basic conditions.