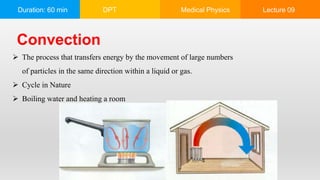
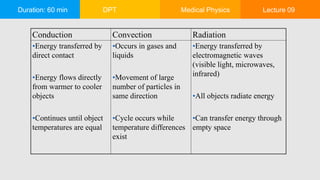
This document summarizes a lecture on heat and temperature. It defines heat as the flow of energy due to temperature differences and explains that all matter is made up of atoms that are constantly moving. Temperature is defined as the measure of the average kinetic energy of particles in an object. Heat transfer occurs through conduction, convection and radiation. Conduction involves the direct transfer of energy between touching objects. Convection refers to the transfer of energy by particle movement within fluids like gases and liquids. Radiation involves the transfer of energy through electromagnetic waves. The lecture also discusses thermal expansion, specific heat and uses examples to explain these concepts of heat transfer.