This document outlines Health Canada's Good Manufacturing Practices (GMP) guidelines for drugs and medical devices. It provides the mandate, purpose, and scope of the GMP guidelines. The guidelines establish quality standards for facilities, equipment, personnel, manufacturing processes, quality control, packaging and labelling, and record keeping. Adherence to the GMP guidelines helps ensure drugs and medical devices in Canada are safe and effective.




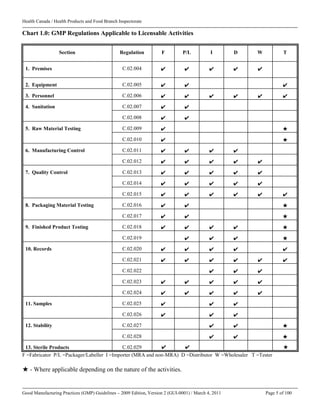





![Health Canada / Health Products and Food Branch Inspectorate
Good Manufacturing Practices (GMP) Guidelines – 2009 Edition, Version 2 (GUI-0001) / March 4, 2011 Page 11 of 100
3.6 Air quality is maintained through dust control, monitoring of pressure differentials between
production areas and periodic verification and replacement of air filters. The air handling
system is well defined, taking into consideration airflow volume, direction, and velocity. Air
handling systems are subject to periodic verification to ensure compliance with their design
specifications. Records are kept.
4. Temperature and humidity are controlled to the extent necessary to safeguard materials.
5. Rest, change, wash-up, and toilet facilities are well separated from production areas and are
sufficiently spacious, well ventilated, and of a type that permits good sanitary practices.
6. Premises layout is designed to avoid mix-ups and generally optimize the flow of personnel and
materials.
6.1 There is sufficient space for receiving and all production activities.
6.2 Working spaces allow the orderly and logical placement of equipment (including parts and
tools) and materials.
6.3 Where physical quarantine areas are used, they are well marked, with access restricted to
designated personnel. Where electronic quarantine is used, electronic access is restricted to
designated personnel.
6.4 A separate sampling area is provided for raw materials. If sampling is performed in the storage
area, it is conducted in such a way as to prevent contamination or cross-contamination.
6.5 Working areas are well lit.
7. Utilities and support systems [e.g., Heating, Ventilating, and Air Conditioning (HVAC), dust
collection, and supplies of purified water, steam, compressed air, nitrogen, etc.] for buildings in which
drugs are fabricated or packaged/labelled are qualified and are subject to periodic verification. Further
guidance is provided in Health Canada’s document entitled Validation Guidelines for Pharmaceutical
Dosage Forms (GUI-0029).( http://www.hc-sc.gc.ca/dhp-mps/compli-conform/gmp-
bpf/validation/gui_29-eng.php).
8. Outlets for liquids and gases used in the production of drugs are clearly identified as to their content.
9. Premises are maintained in a good state of repair. Repair and maintenance operations do not affect
drug quality.
10. Where necessary, separate rooms are provided and maintained to protect equipment and associated
control systems sensitive to vibration, electrical interference, and contact with excessive moisture or
other external factors.
11. Fabricators and packagers must demonstrate that the premises are designed in such a manner that the
risk of cross-contamination between products is minimized.](https://image.slidesharecdn.com/goodmanufacturingpracticesgmp-180220014221/85/Good-manufacturing-practices-gmp-11-320.jpg)

















































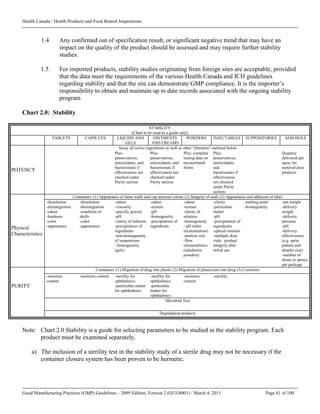



















![Health Canada / Health Products and Food Branch Inspectorate
Good Manufacturing Practices (GMP) Guidelines – 2009 Edition, Version 2 (GUI-0001) / March 4, 2011 Page 82 of 100
Appendix A1
Content of the Fabricator’s/Manufacturer’s Batch Certificate
for Drug/Medicinal Products Exported to Countries
under the Scope of a Mutual Recognition Agreement (MRA)
[Letterhead of Exporting Manufacturer]
1. Name of product.
Proprietary, brand, or trade name in the importing country.
2. Importing country.
3. Marketing authorization number.
The marketing authorization number of the product in the importing country should be provided.
4. Strength/Potency.
Identity (name) and amount per unit dose are required for all active ingredients/constituents.
5. Dosage form (pharmaceutical form).
6. Package size (contents of container) and type (e.g., vials, bottles, blisters).
7. Lot/batch number.
As related to the product.
8. Date of fabrication/manufacture.
In accordance with national (local) requirements.
9. Expiry date.
10. Name and address of fabricator(s)/manufacturer(s) - manufacturing recognized building(s).
All recognized buildings involved in the manufacture of the batch including packaging and quality
control of the batch, should be listed. The name(s) and address(es) given must correspond to the
information provided on the manufacturing authorization/establishment licence.
11. Number(s) of manufacturing authorization(s)/licence(s) or certificate(s) of GMP compliance
held by fabricator(s)/manufacturer(s).
A number should be given for each recognized building listed under Item 10.
12. Results of analysis.
Should include the approved specifications, describe all results obtained, and refer to the analytical
methods used (May refer to a separate certificate of analysis, which must be dated, signed, and
attached).
13. Comments/remarks.](https://image.slidesharecdn.com/goodmanufacturingpracticesgmp-180220014221/85/Good-manufacturing-practices-gmp-82-320.jpg)

















