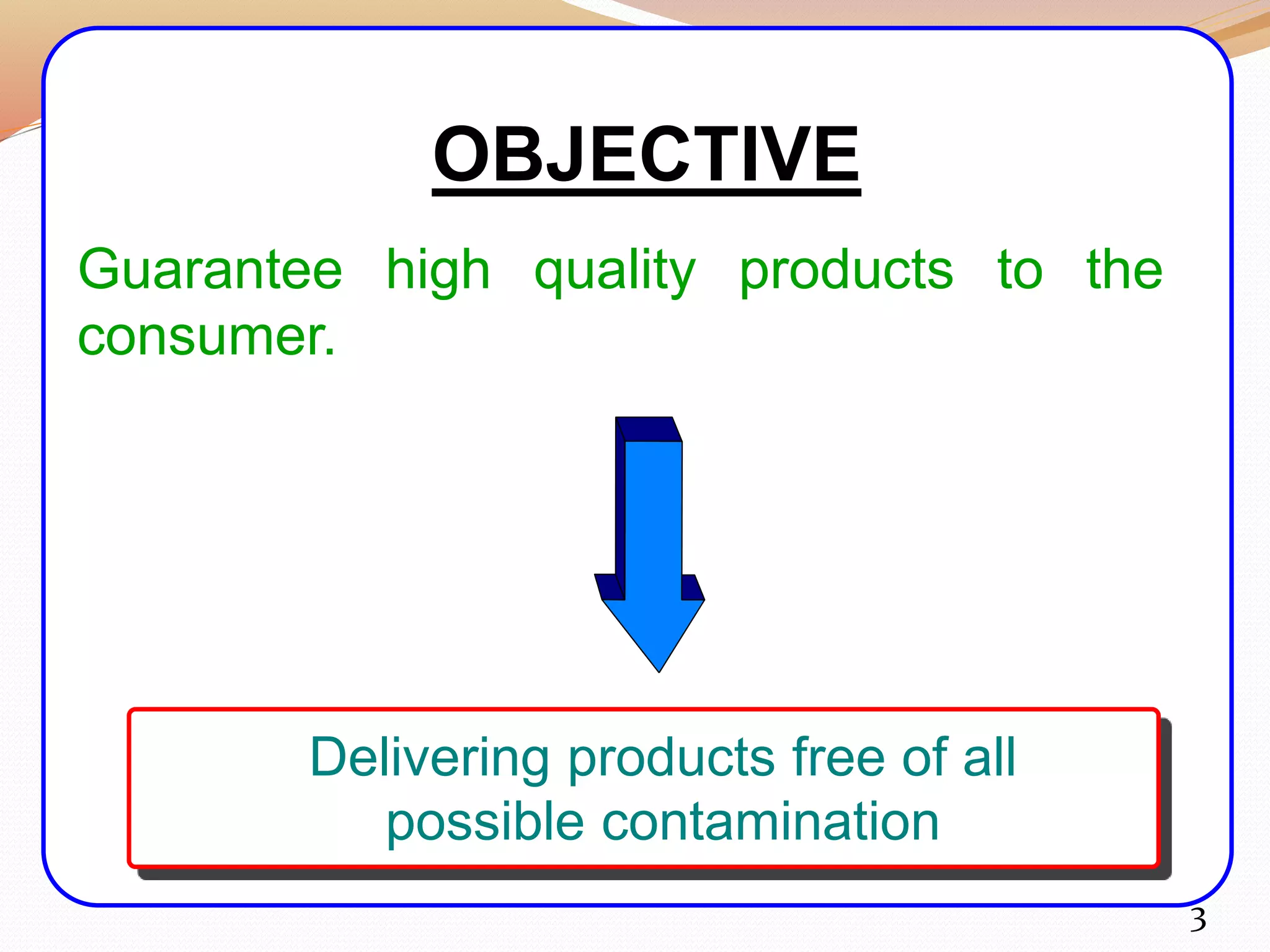
Good Manufacturing Practice (GMP) encompasses regulations and guidelines aimed at ensuring the quality of drug substances, medical devices, and food products. Key principles include proper facility design, rigorous documentation, monitoring, and employee training to prevent contamination and ensure product safety. Adhering to GMP is crucial for the pharmaceutical industry to deliver safe and effective products, mitigating risks such as contamination and inaccurate labeling.
![GOOD MANUFACTURING PRACTICE
Mr. Sagar Kishor Savale
[Department of Pharmaceutics]
avengersagar16@gmail.com
2015-2016
Department of Pharmacy (Pharmaceutics) | Sagar savale](https://image.slidesharecdn.com/goodmanufacturingpracticepritesh-160516130050/75/Good-manufacturing-practice-GMP-1-2048.jpg)