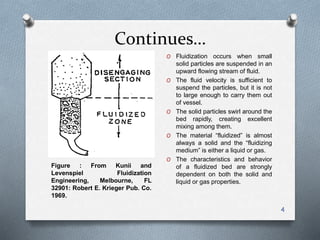
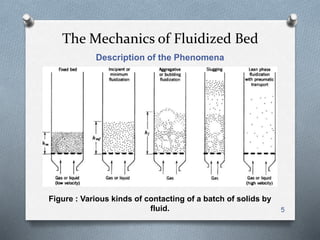
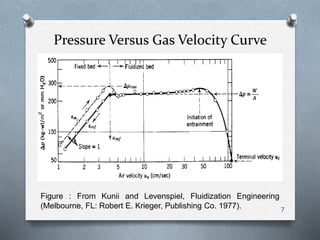
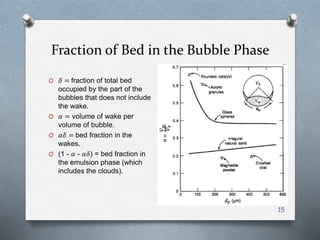
The document provides an overview of fluidized bed reactors (FBR), including their characteristics, operational mechanics, and applications within chemical engineering. It discusses key concepts such as mass transfer between phases, reaction behavior in fluidized beds, and the balance of materials in different phases including bubbles and emulsions. Additionally, the document highlights advantages and disadvantages of FBRs, and presents the mathematical models that describe their performance and behavior.




























![References
1. nptel.ac.in/courses/103103026/module2/lec18/1.html
2. D. Kunii and O. Levenspiel, Fluidization Engineering (New York:
Wiley, 1968).
3. H. S. Fogler and L. F. Brown [Reactors, ACS Symposium
Series, vol.168, p. 31 1981, H. S. Fogler ed.]
4. T.E. Broadhurst and H.A. Becker, AIChE J., 21, 238 (1975).
5. J. F. Davidson and D. Harrison, Fluidized Particles (New York:
Cambridge University Press, 1963).
6. S. Mori and C. Y. Wen, AIChE J., 21, 109 (1975).
7. J. Werther, ACS Symposium Series., 72, D. Luss & V. W.
Weekman, eds. (1978).
8. https://en.wikipedia.org/wiki/Fluidized_bed_reactor
9. http://faculty.washington.edu/finlayso/Fluidized_Bed/FBR_Intro/
uses_scroll.htm
34](https://image.slidesharecdn.com/fluidizedbedreactoranoverview-161106192106/85/Fluidized-bed-reactor-an-overview-34-320.jpg)
