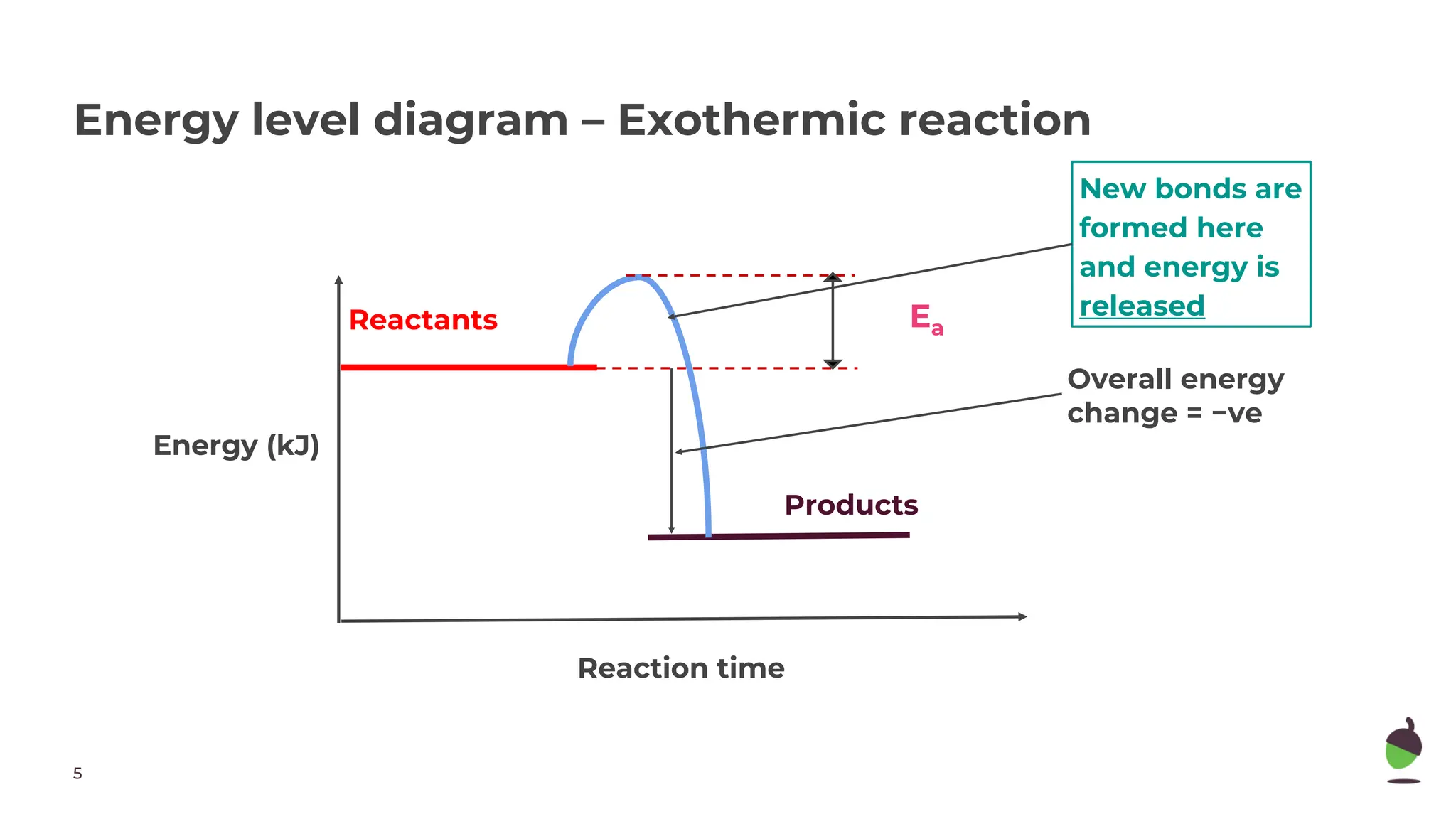
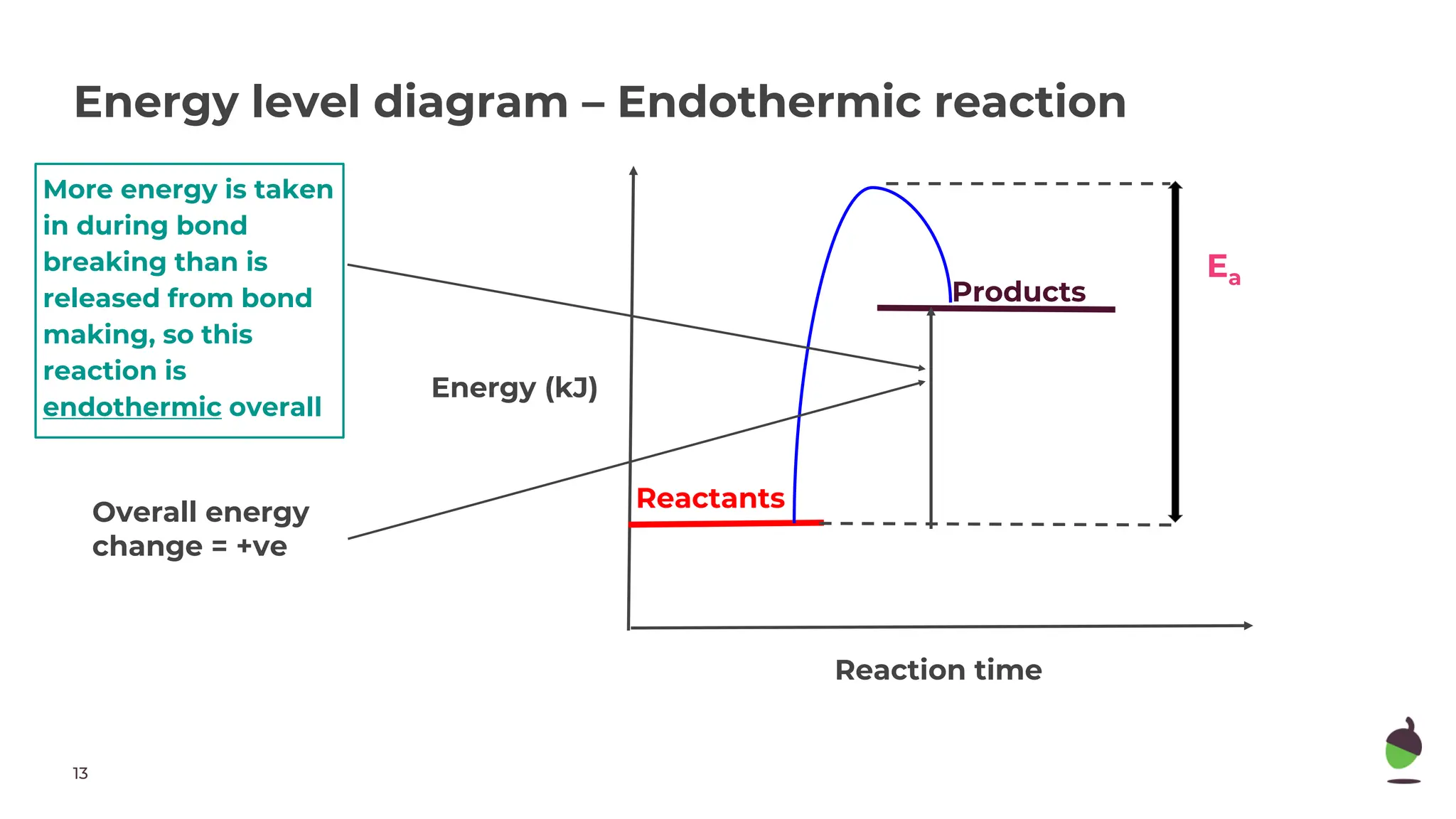
The document discusses exothermic and endothermic chemical reactions. Exothermic reactions release energy to the surroundings and feel hot, while endothermic reactions absorb energy from the surroundings and feel cold. A reaction is exothermic if more energy is released during bond making than used for bond breaking, and endothermic if more energy is used for bond breaking than released during bond making. Energy diagrams show exothermic reactions releasing energy overall and endothermic reactions absorbing energy overall.