Embed presentation
Downloaded 48 times



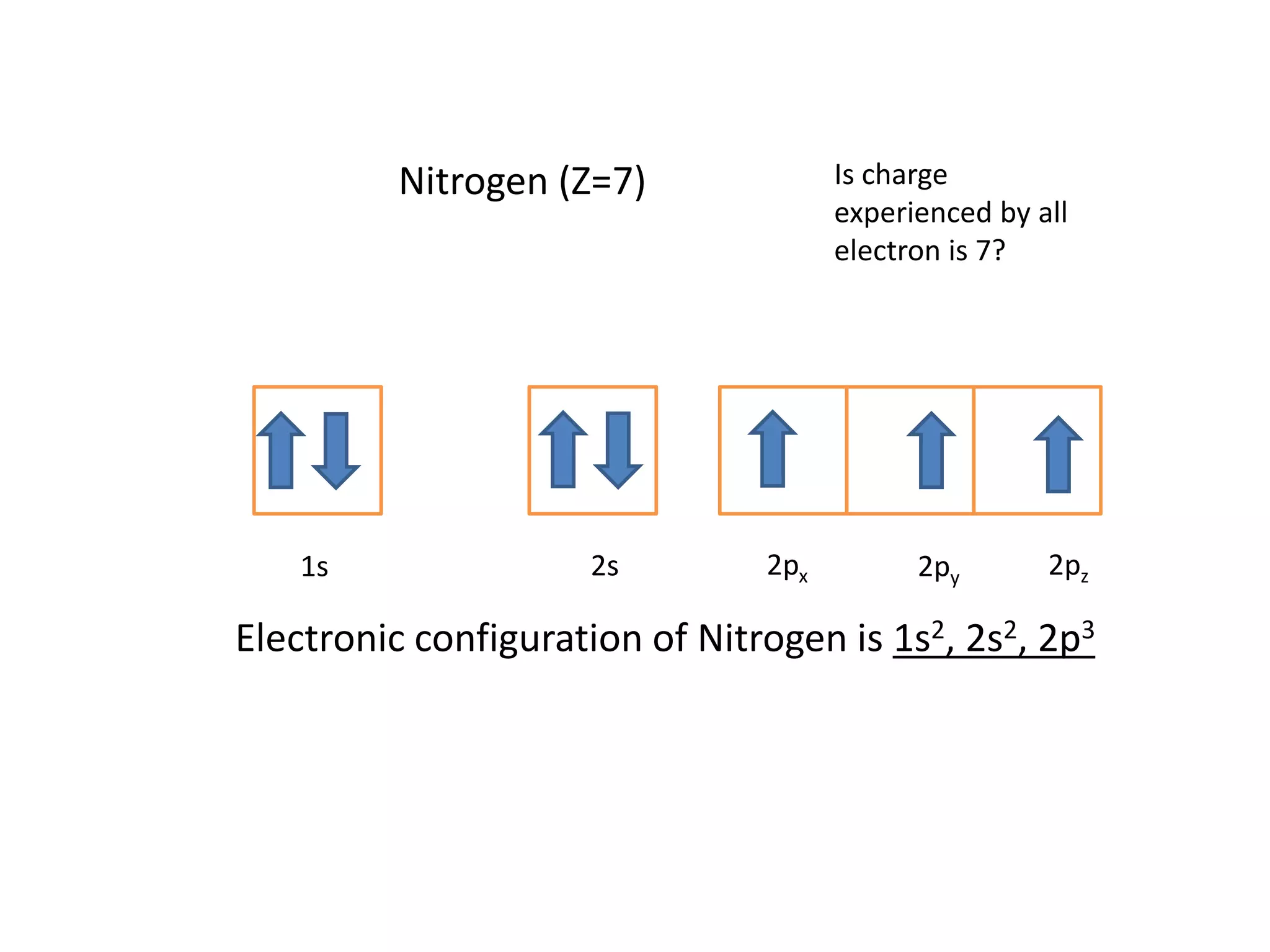



Slater's rules provide a method to estimate the shielding of electrons and the effective nuclear charge experienced by electrons in an atom. The rules involve writing the electron configuration, ignoring higher energy level electrons, and applying shielding constants of 0.35 for electrons in the same subshell and 0.85 for electrons in the previous subshell. As an example, the rules are used to calculate that the effective nuclear charge experienced by the valence electrons of nitrogen is 3.9 instead of the actual nuclear charge of 7.






